Magnetic Properties of Complexes
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
what is paramagnetism and how do molecules have it.
The electrons inside a molecule may be regarded as miniature magnets.
when there are unpaired electrons, the magnetic moments will align themselves with an externally applied magnetic field.
Para-magnetic complexes are attracted into magnetic fields; the magnitude of the attraction depends on the number of unpaired electrons
How do we calculate the effective magnetic moment of complexes of first row elements can be calculated by using the following ‘spin-only’ equation.
(n is the number of unpaired electrons)
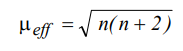
What are the units of μeff
μeff is usually expressed in units of Bohr magneton (μB), or BM, the magnetic moment associated with a single electron in the H atom:

What are two practical consequences regarding weight that is caused by para-magnetism
When weighed in the absence of an applied field and subsequently in the presence of a magnetic field, paramagnetic substances show: an increase in weight
From the weight difference, μeff can be calculated and: the number of unpaired electrons can be determined
can find out whether a complex is high or low spin
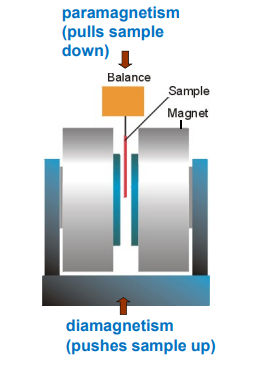
How do we weigh paramagnets
The weighing's are carried out in a Gouy balance:
The magnet is an electromagnet
Calibrations are carried out against samples of precisely known composition corrected for diamagnetism
What are Diamagnets
Complexes without unpaired electrons: external magnetic field still disturbs the movement of the electrons around the nuclei
(effect is small)
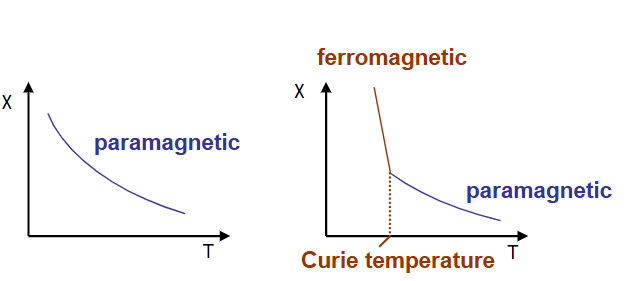
What is Ferromagnetism
Substances that exhibit extremely strong attraction to magnetic fields and can become permanently magnetized
Detail the temperature dependence of magnets
Above the critical temperature, the thermal energy is high enough to prevent alignment.
χ: magnetic susceptibility, degree of magnetisation in response to an applied field