Bio 112 - Proteins, Sugars, Lipids, Nucleic Acids, & Energy
1/95
Earn XP
Description and Tags
Basically everything that makes up things
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
96 Terms
Covalent bond → interaction & strength?
2 atoms sharing an electron → strong(est)
Ionic bond → interaction & strength?
One atom gives up electron to another - not as strong as covalent but stronger then hydrogen
Hydrogen bond → interaction & strength?
Sharing of H+ ions/ attraction of opposite charges → weakest
Cation vs. Anion
Cation: gives electron → release/give energy
Anion: takes electron → take energy
Acid vs Base
Acid (pH = 0-7) gives H+; Base (pH = 7-14) takes H+
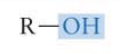
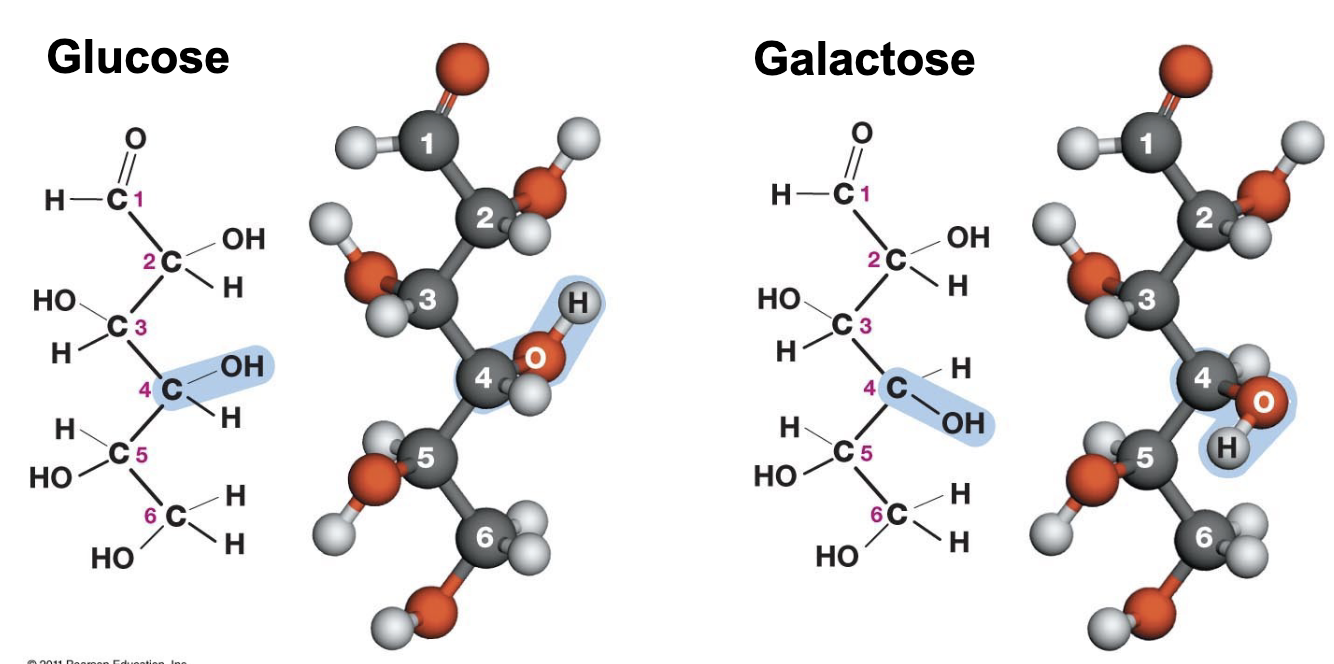
What functional group is this? Is it polar?
Hydroxyl group* → very polar so therefore makes compounds more soluble
*Important to know!
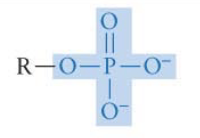
What functional group is this? Is it polar?
Phosphate group → polar
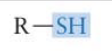
What functional group is this? Is it polar?
Sulfhydryl group → polar
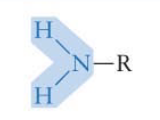
What functional group is this? Is it polar?
Amino group* → polar
*important to know!
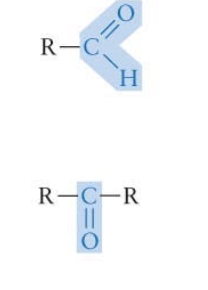
What functional group is this? Is it polar?
(top = found at end, bottom = found in middle)
Carbonyl group - polar
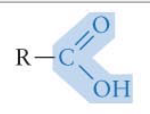
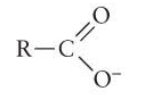
What functional group is this? Is it polar?
Carboxyl group* (-COOH) - polar
*important to know - acts as an acid (tends to loose a proton in a solution to form pictured formula
Condensation reaction vs Hydrolysis
Monomer & Water → in or out?
Use or give energy → reaction type?
Condensation: Monomer comes in, Water goes out; uses energy → anabolic /// Hydrolysis: Monomer goes out, Water comes in; gives energy → catabolic
Polypeptide (protein) chain is made of __ joined by __ bonds
Amino acids joined by peptide bonds
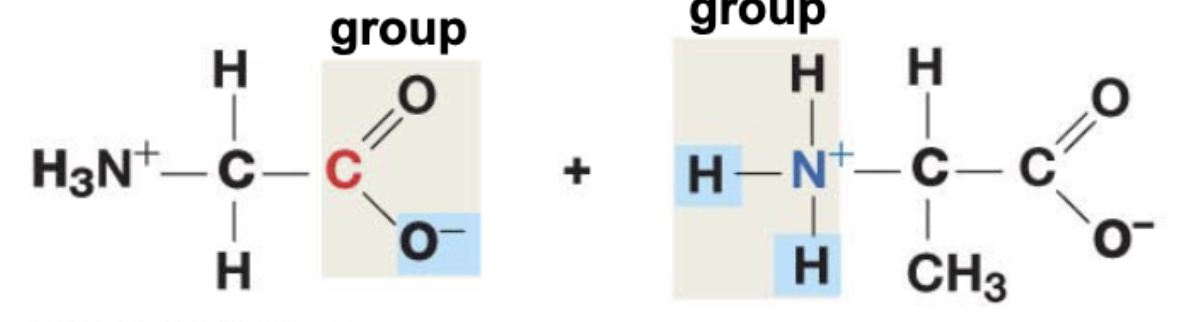
Peptide bonds are between the (atom) from the (functional) and the (atom) from the (functional group); Can this bond rotate freely?
Carbon (C) from the Carboxyl group and the Nitrogen (N) from the amino group
Peptide bond’s CANT rotate freely - electron sharing makes them similar to double bonds, which also can’t move freely
What do buffers do?
Make a solution resistant to pH change
Why are buffers important?
Human body has pH of 7 → amino acids are ionized (charged) at this pH → buffers keep pH stable/constant → if they didn’t, proteins would become non-charged & get destroyed
Primary structure of a protein refers to?
The order of the monomers/amino acids
What does secondary structure of a protein refer to?
The way the protein folds/formation of α-helices & β pleated sheets
What determines how a protein can fold? (2 things)
Polarity of side-chains (2 neg charged AA’s would repel each other → be on opposite sides)
size of side chains (bulky side chain wants to be on it’s own)
What does tertiary structure of a protein refer to?
The overall 3D shape of a polypeptide/protein
What determines tertiary structure in a protein? *what’s the most important?
Side chain interactions - basically all of the “weak” interactions
*hydrophobic interactions are the most important
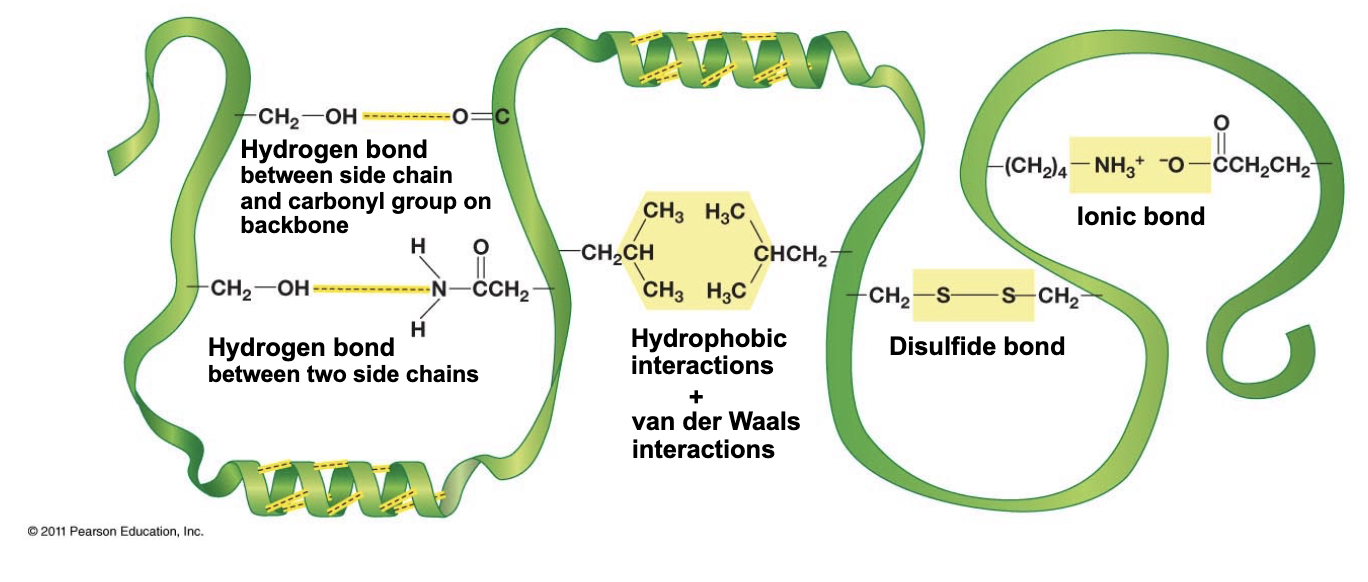
Tertiary structure - hydrogen bonds btwn the side chains of?
polar amino acids
Tertiary structure - hydrophobic or van der Waals interactions are btwn the side chains of?
non-polar amino acids
Tertiary structure - ionic bonds are btwn side the chains of
charged amino acids
Tertiary structure - disulfide bonds (what’s special/needed)
The only covalent bond within proteins - happens VERY occasionally
“basically” need 2 cysteines in close physical proximity (secondary structure; during folding)
redox reaction occurs and disulfide bridge is formed
often found in proteins that fulfill structural roles
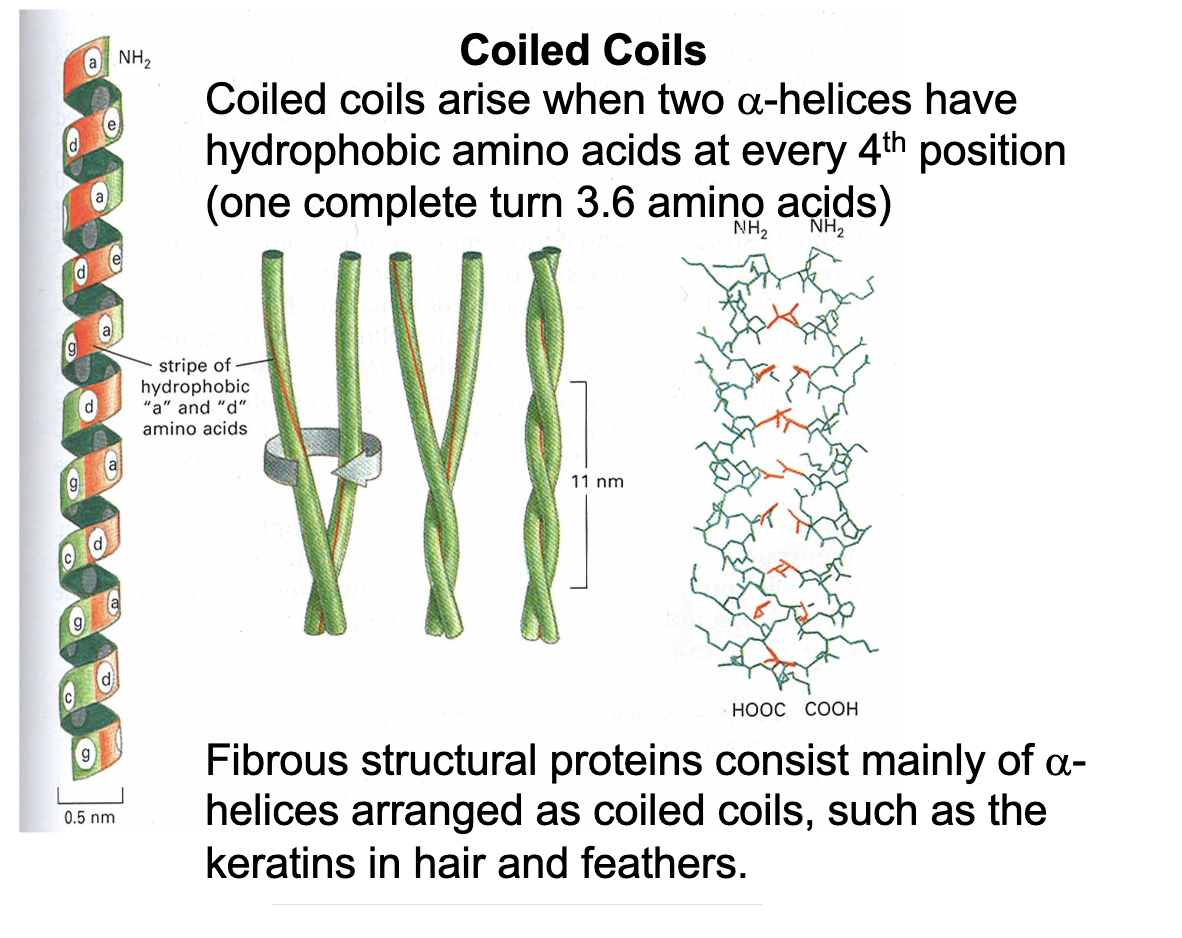
Fibrous structural proteins (like keratins in hair and feathers) consist of mainly…
Coiled coils
Coiled coils - what, how, & why
Two α-helices have hydrophobic amino acids at every 4th position (which are facing towards each other)
thermodynamically favorable because it “minimally’ inter-fears with the hydrogen bonds in the surrounding the protein in a (watery) solution - so its just the “most favorable”
What is a Quaternary structure? What determines if there even is one?
The shape produced by combinations of polypeptides (and therefore combinations of tertiary structures); primary structure ultimately determines if there’s a Quaternary structure - maybe 10% of proteins
What percent of proteins have Quaternary structure?
10%
General formula for a carbohydrate monomer =
Multiples of CH2O
What functional groups are found in a sugar?
one carbonyl group + several hydroxyl groups
What’s an isomer?
Molecules that share the same formula but have different arrangements of atoms in space
All isomers differ in their __?
Covalent bonds - organization, aka “what is connected to what”
In many isomers, a group is attached to different (element) atoms
Carbon (ex: CH3-CHOH-CH3 vs CH3-CH2-CH2OH)
Optical isomers are _ _ of each other, and occur whenever a carbon have (number) different atoms/groups attached to it
Mirror images; 4
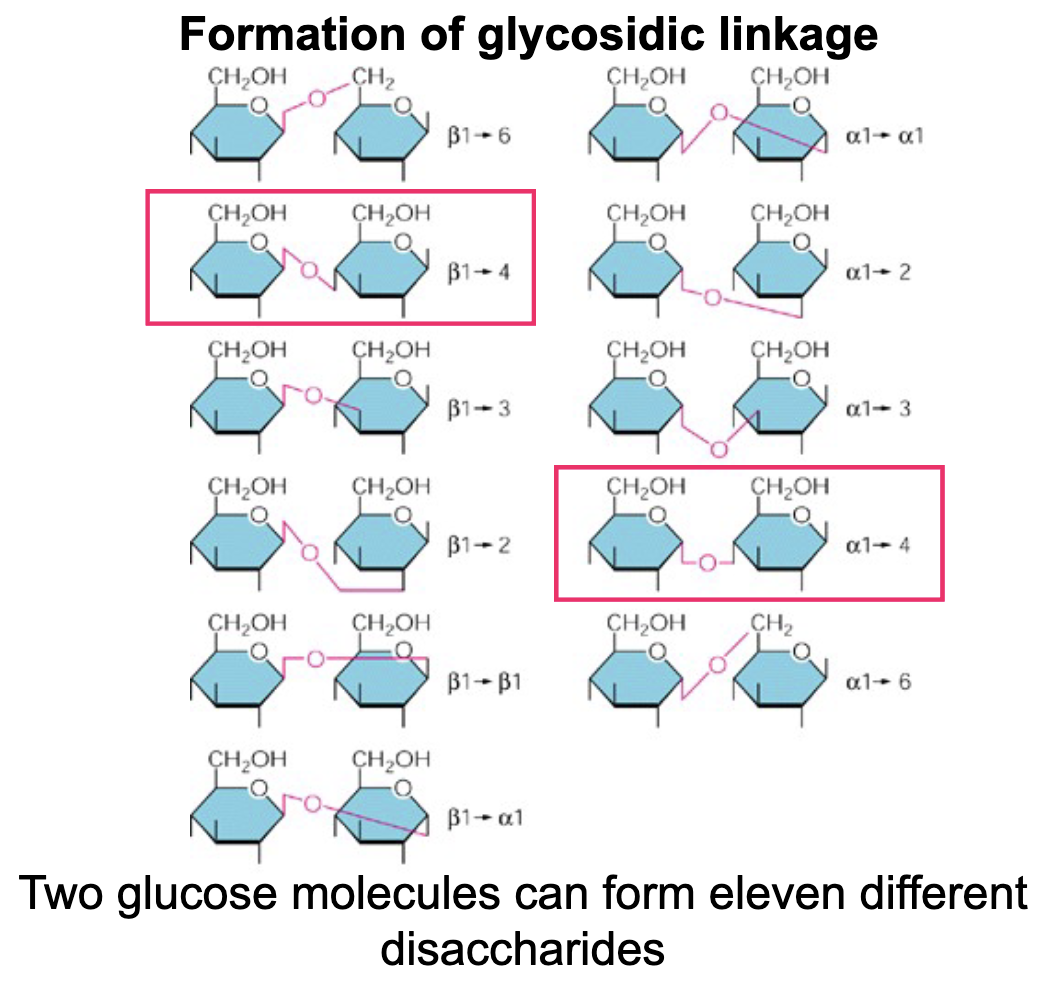
Polysaccharides (sugars) are formed by (covalent/ionic) bonds called __ __
Covalent; glycosidic linkages
Glycosidic linkages in polysaccharides are between __ and __
The C-1 of one sugar and any OH-group of the second sugar
Starches are in the shape of a spiral because of what
Bulky side-chains (CH2OH) are all on the same side, and they bend the polymer
Generally what are lipids/fats for?
Long term (energy) storage
Are lipids soluble in water?
No!
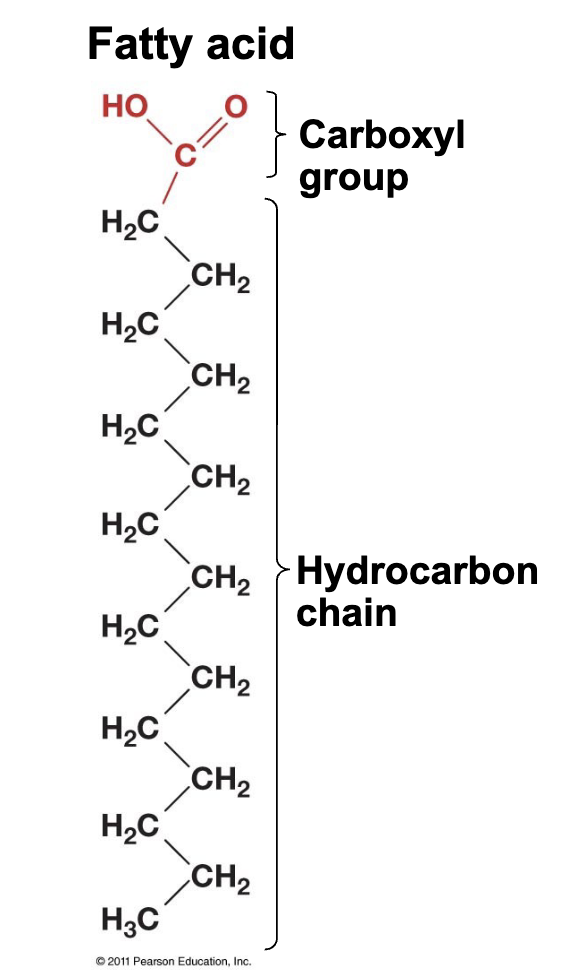
What are the most important lipids? and what are they made of?
Fats(solid) and oils (liquid); 3 fatty acids and 1 glycerol connected by covalent bonds.
Fatty acids have (functional group + smth else) and (are/aren’t) amphiphilic
1 carboxyl group + a long hydrocarbon chain; are amphiphilic
Phospholipids have (3 things) and (are/aren’t) amphiphilic
a hydrophilic head group and 2 hydrophobic fatty acid tails attached to glycerol; are amphiphilic
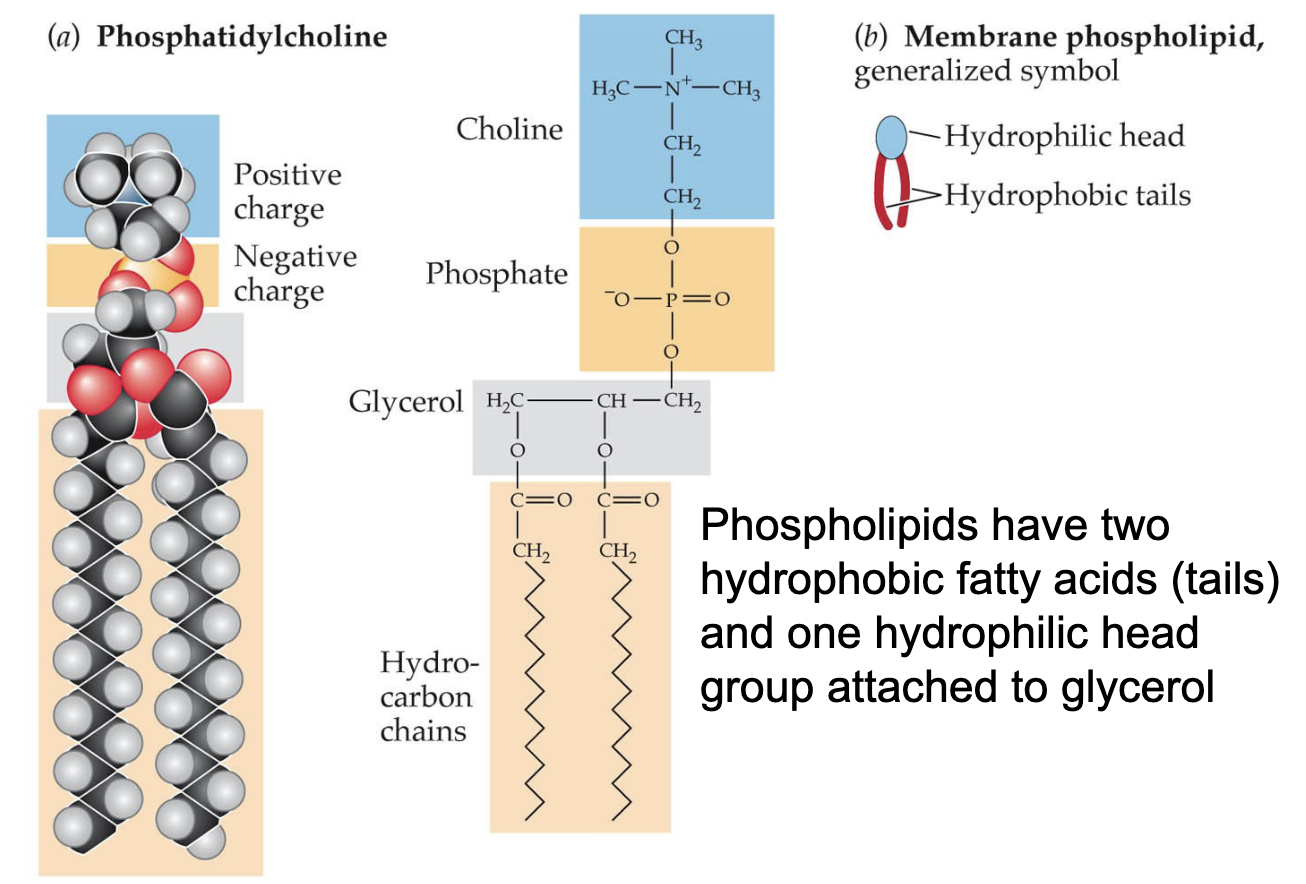
Lipid bilayers have hydro(philic/phobic) heads and hydro(philic/phobic) tails
Hydrophilic heads that interact with water + hydrophobic tails that interact with each other
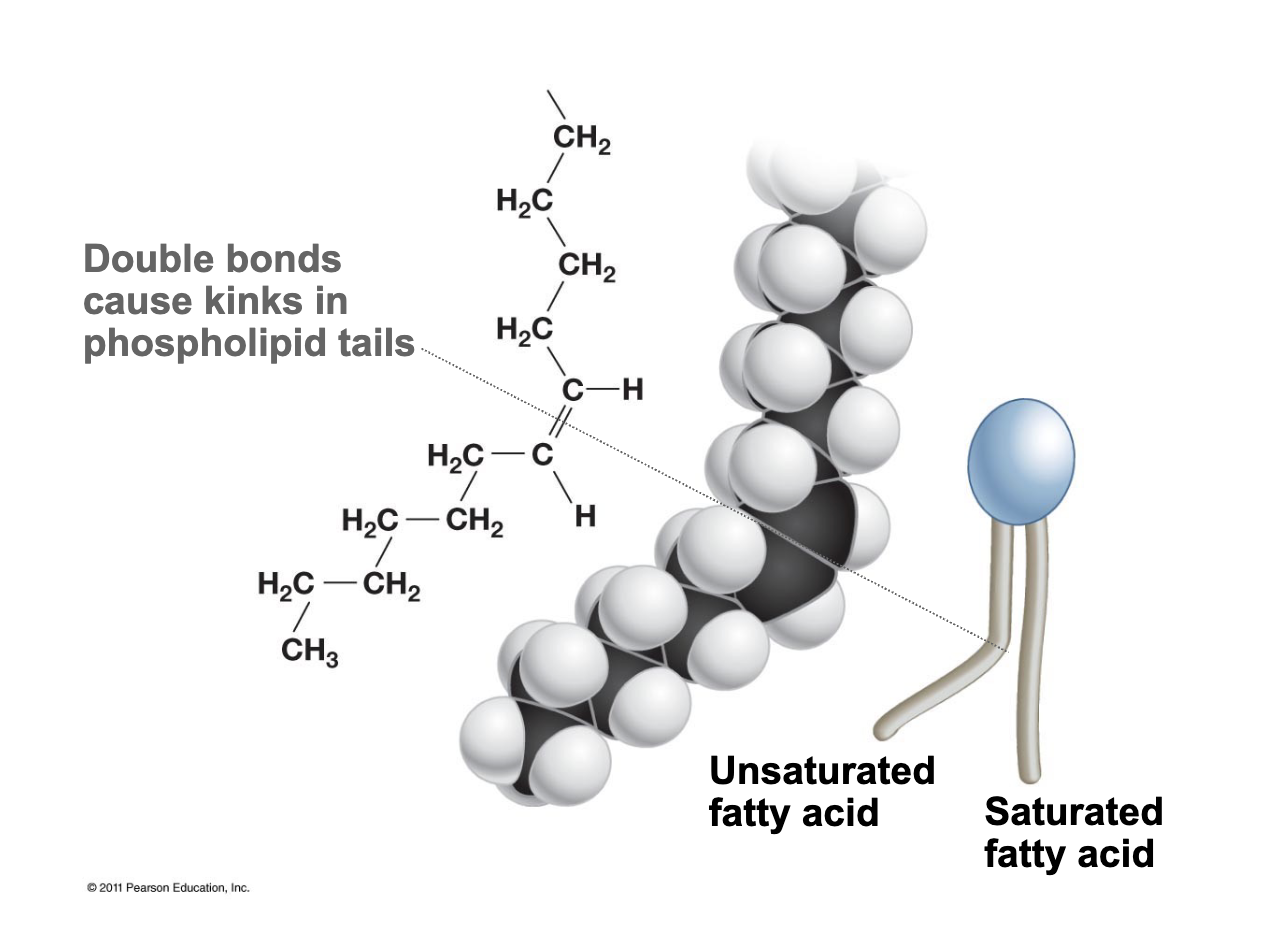
Unsaturated fatty acids are fatty acids that contain __, which cause __
fatty acids taht contain double bonds → double bonds cause kinks in the tails and can’t be packed as closely together compared with saturated fatty acids (reason why butter is solid at room temp and oils are fluid at room temp)
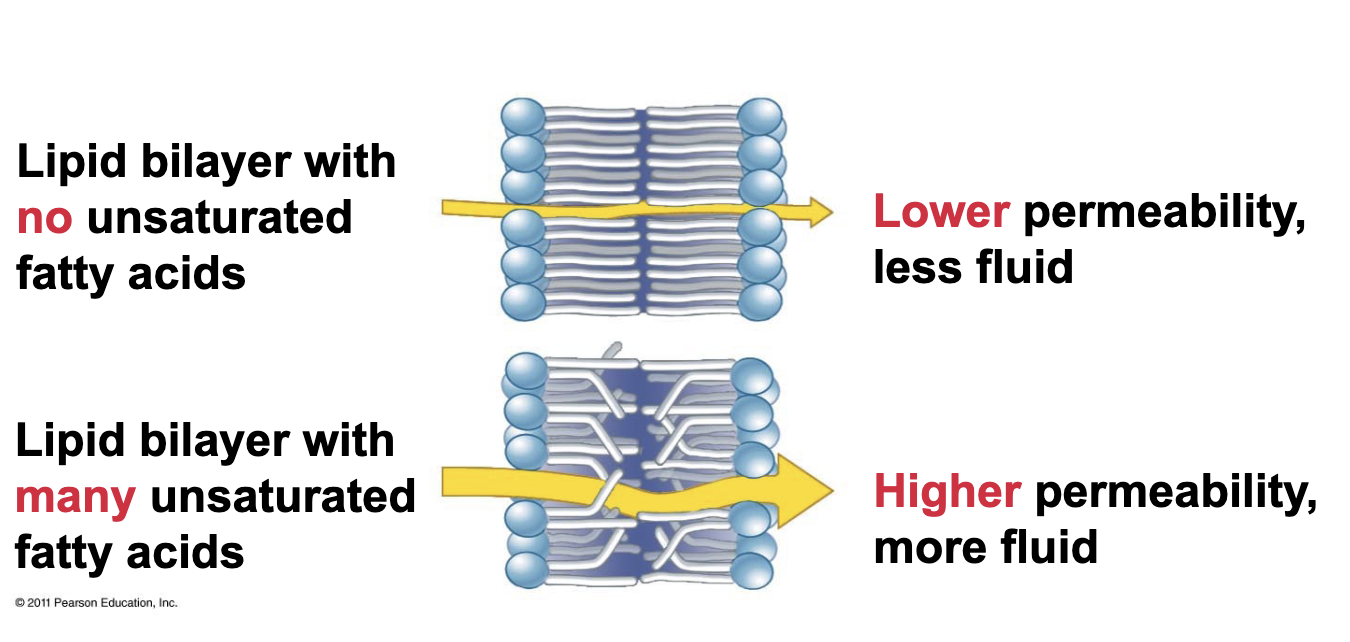
How do unsaturated fatty acids affect the lipid bilayer
They increase fluidity and permeability of the membrane
Integral proteins vs transmembrane proteins
Integral proteins are proteins permanently attached to the lipid bilayer, transmembrane are a subsection that cross the bilayer (instead of just being a part)
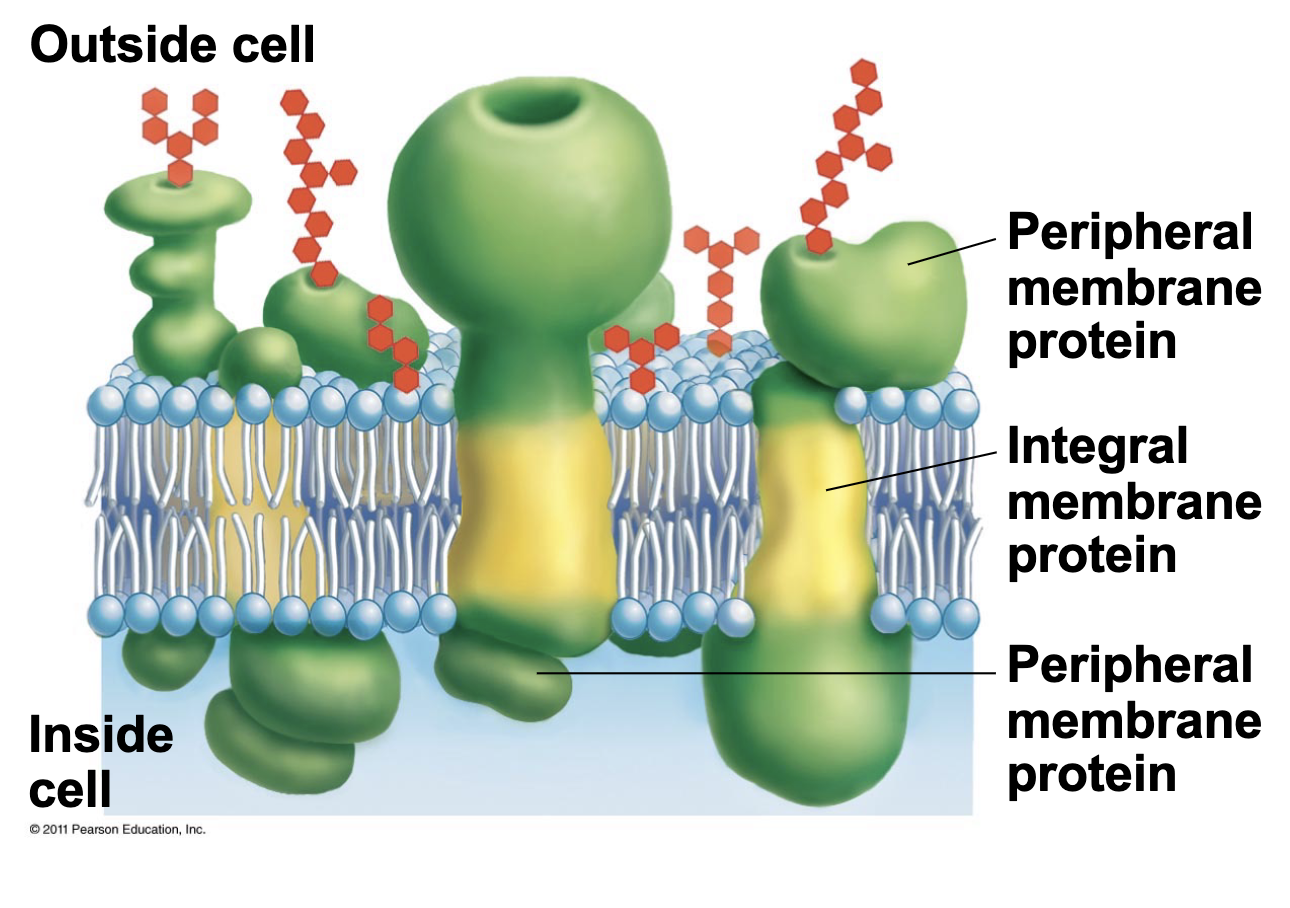
Membrane proteins are usually integrated into the membrane by what?
One or more alpha-helices
Main function of membranes is to…?
Serve as a barrier and selectively transport molecules
Order in terms of high to low permeability: small uncharged polar molecules, ions, large uncharged polar molecules, small non-polar molecules
small non-polar molecules
small uncharged polar molecules
large uncharged polar molecules
Ions
Whats a means of passive transport (and explain it)
Diffusion = passive mixing of substances resulting in net transport along a concentration gradient
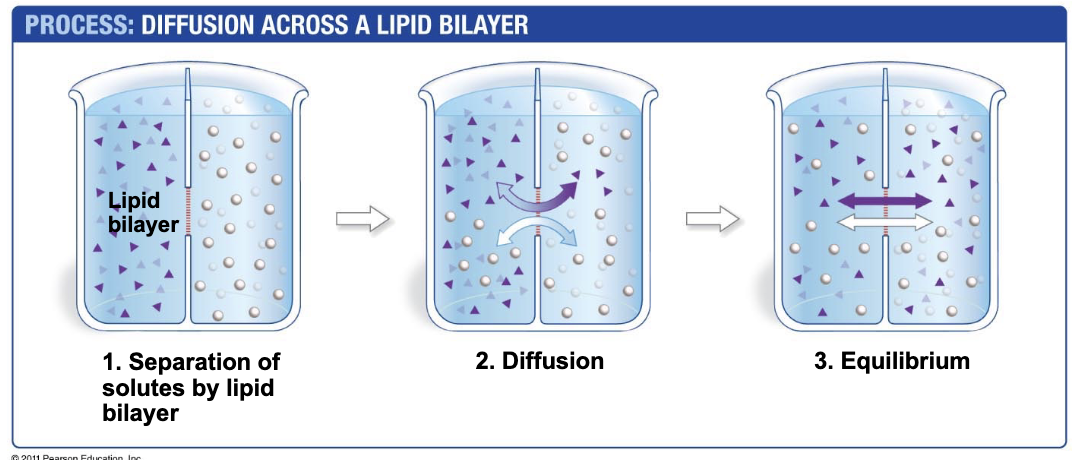
Why does diffusion happen?
Brownian motion (random walk) of molecules due to thermal motions and collisions
What are diffusion rates determined by? (3 things)
Temperature, size of molecule, and steepness of concentration gradient
What is osmosis?
Diffusion of water across a selectively permeable membrane
Water moves from regions of (low/high) solute concentration to regions of (low/high) solute concentration
low; high
Characteristics of a hypertonic solution
higher solute concentration (inside/outside) membrane
net flow of water is (in/out)
cell (grows/shrinks)
Higher solute concentration outside membrane, net flow of water is outwards, cell shrinks
Characteristics of a hypotonic solution
higher solute concentration (inside/outside) of cell
net flow of water is (in/out)
Cell (expands/shrinks)
Higher solute concentration inside membrane, net flow of water is inwards, cell expands
Isotonic solution
higher solute concentration (inside/outside) of cell
net flow of water is (in/out)
cell (grows/shrinks)
Solute concentrations are equal on both sides of membrane, no net flow of water, cell doesn’t change size
Facilitated diffusion is (passive/active) and depends on what 2 types of membrane proteins?
Passive - only flow along the concentration gradient
Gated channel proteins - ions flow along their electrochemical gradient when the channel is open
Carrier proteins - binds the transported substance
Passive transport vs active transport
Passive - no energy needed, flow with concentration gradient
Active - energy needed, flow against concentration gradient
Primary vs secondary active transport
Primary - directly relies on ATP hydrolysis to move against concentration gradient
Secondary - uses energy from concentration gradient set up by primary active transport
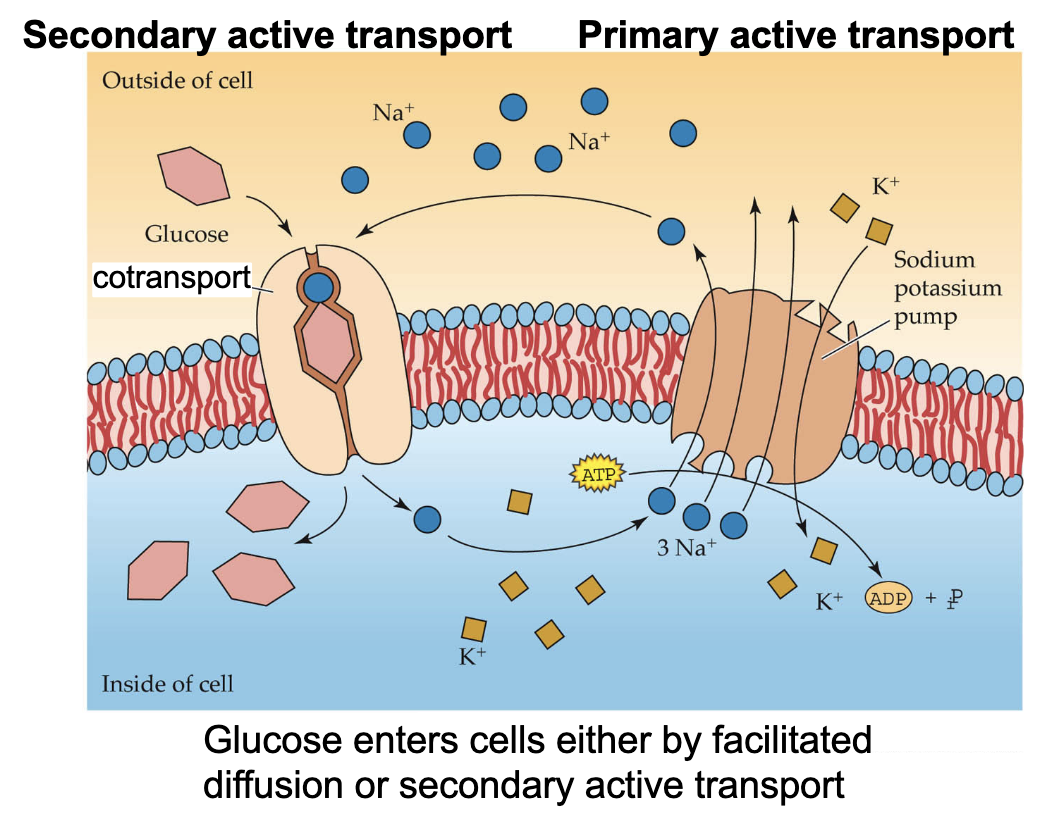
(most important) example of active transport
Sodium potassium pump
counteracts hypotonic drinking water
only in animals (not plants bc they have stable cell walls)
controls osmolarity, generates the resting potential, and sets up ion gradients
How is a sugar sodium co-transporter an example of a secondary active transport system
It uses energy from sodium inflow to transport sugar into cells w/ high interior concentration of sugar
Nucleotides:
Are building blocks of?
Functions?
RNA + DNA
Signaling + energy
Nucleotides are made of (3 things)
a nitrogen base, a 5-carbon sugar, & a phosphate group (A, G, C, or T/U)
Which of the phosphates are pyrimidines?
Cytosine, Uracil (RNA), Thymine (DNA)
Which of the phosphates are purines?
Guanine, Adenine
Phosphate group pairs + why 1 of each type?
G & C /// A + T/U
pyrimidine + pyrimidine = too much space
purine + purine = not enough space
pyrimidine + purine = just right
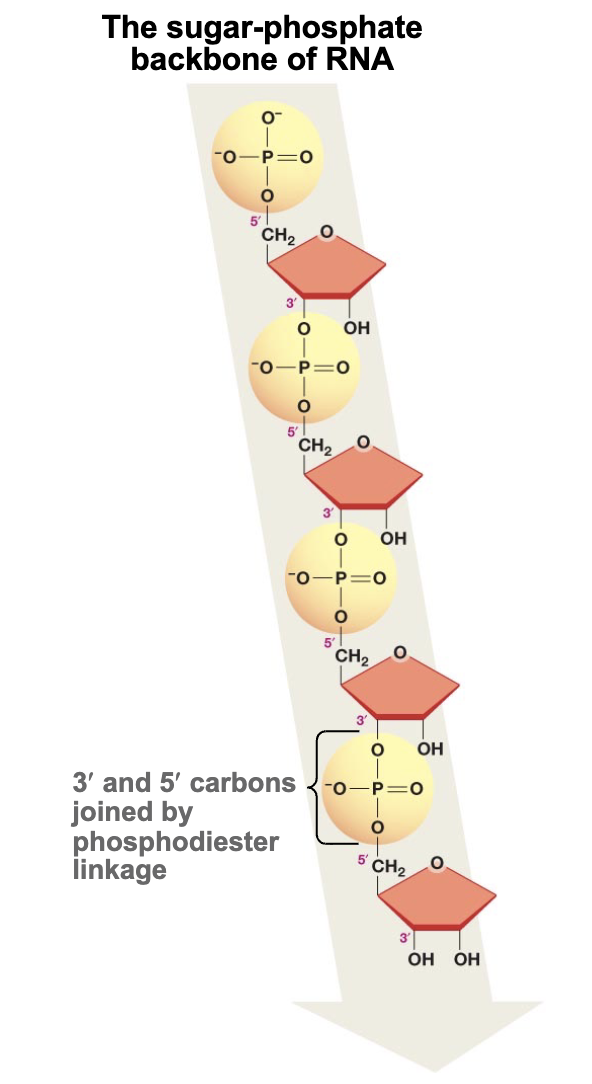
Nucleotides polymerization is what kind of reaction?
Condensation reaction → needs energy → anabolic
3’ hydroxyl group forms bond w/ 5’ phosphate group of next nucleotide
During the polymerization of nucleotides, the [#] [part of nucleotide] of the monomer joins with the [#] [part of nucleotide] of the current polymer
The 5’ phosphate group of the monomer joins with the 3’ of the end sugar (the -OH)
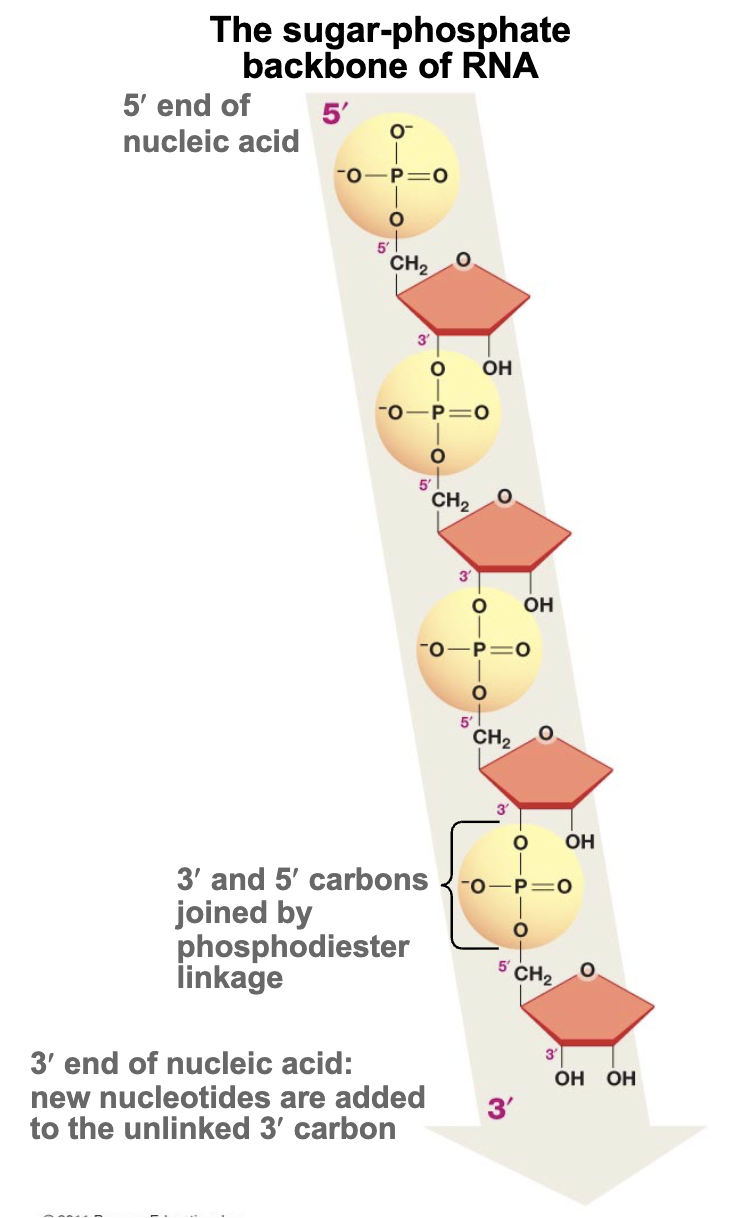
Long (nucleic acid) polymers start with the [#] of the [part of nucleotide] and end with the [#] of the [part of nucleotide]
Start with 5’ of the phosphate group & end with 3’ of the sugar (hydroxyl group part)
Metabolism is subdivided into what 2 things
Anabolism & Catabolism
Anabolic vs Catabolic reactions
Anabolic → need/ take energy
Catabolic → release/give energy
Energy wants to __ over time
evenly disperse
Energy change equation
ΔG = ΔH - TΔS
(Free energy; heat; absolute temp; disorder within cell)
ΔG
Change in free energy
ΔH
Change in heat
ΔS
Change in entropy AKA change in disorder within a cell
If ΔG is negative, energy is (released/required)
released → catabolic
If ΔG is positive, energy is (released/required)
required → anabolic
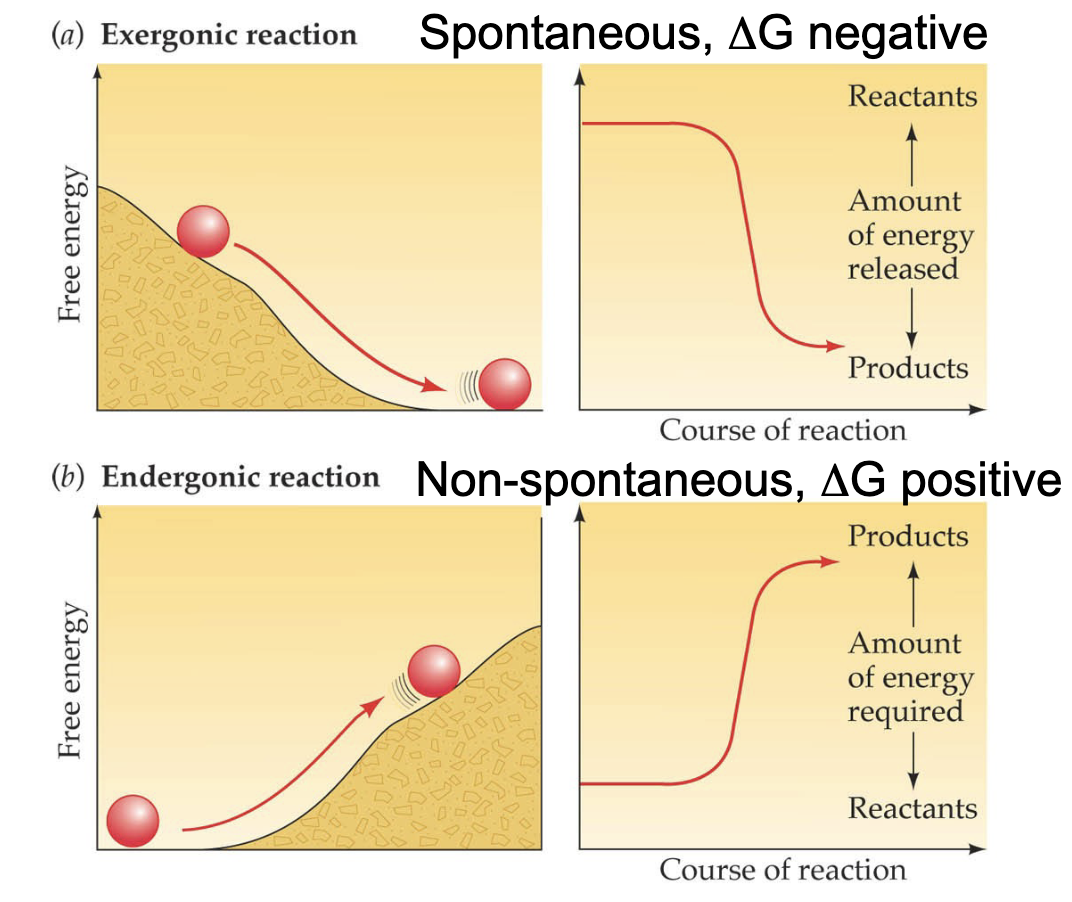
Reaction types: Exergonic vs Endergonic
catabolic/anabolic
pos/neg ΔG
spontaneous?
Exergonic = catabolic (energy released) → ΔG is negative; always spontaneous
Endergonic = anabolic (energy required) → ΔG is positive; never spontaneous
Reaction types: Heat released & disorder decreases;
Write equation
spontaneous (above/below) specific temp?
Example/explanation of when this happens
Below a specific temp; -ΔG = -ΔH - TΔS;
In protein folding, hydrogen bonds are formed which releases heat (-ΔH); order is increasing, and therefore disorder is decreasing (-ΔS)
Reaction types: Heat required & disorder increases;
Write equation
spontaneous (above/below) specific temp?
Example/explanation of when this happens
-ΔG = ΔH + TΔS; Above a specific temp;
when dissolving NaCl into water, a lot of heat is needed to break bonds in NaCl (+ΔH); breaking of bonds = increase in disorder (+ΔS)
Since endergonic (anabolic) reactions are never spontaneous, how do they occur?
Endergonic reactions are coupled with exergonic reactions → some of the free energy released by exergonic reacions is captured in ATP, which can then drive endergonic reactions
What do all cells use for capture, transfer, and storage of energy?
Adenosine triphosphate (ATP)
For a reaction to occur, ΔG has to be (neg/pos)
Negative
ATP hydrolysis is an (endergonic/exergonic) reaction that can be used to drive (endergonic/exergonic) reactions like making a polymer
Exergonic; endergonic
The (direction/rate) of a reaction if ΔG is known but not the (direction/rate)
Direction; rate
Exergonic reactions happen only after the addition of a small amount of added energy, called __ __?
Activation energy - needed to put molecules into a transition state
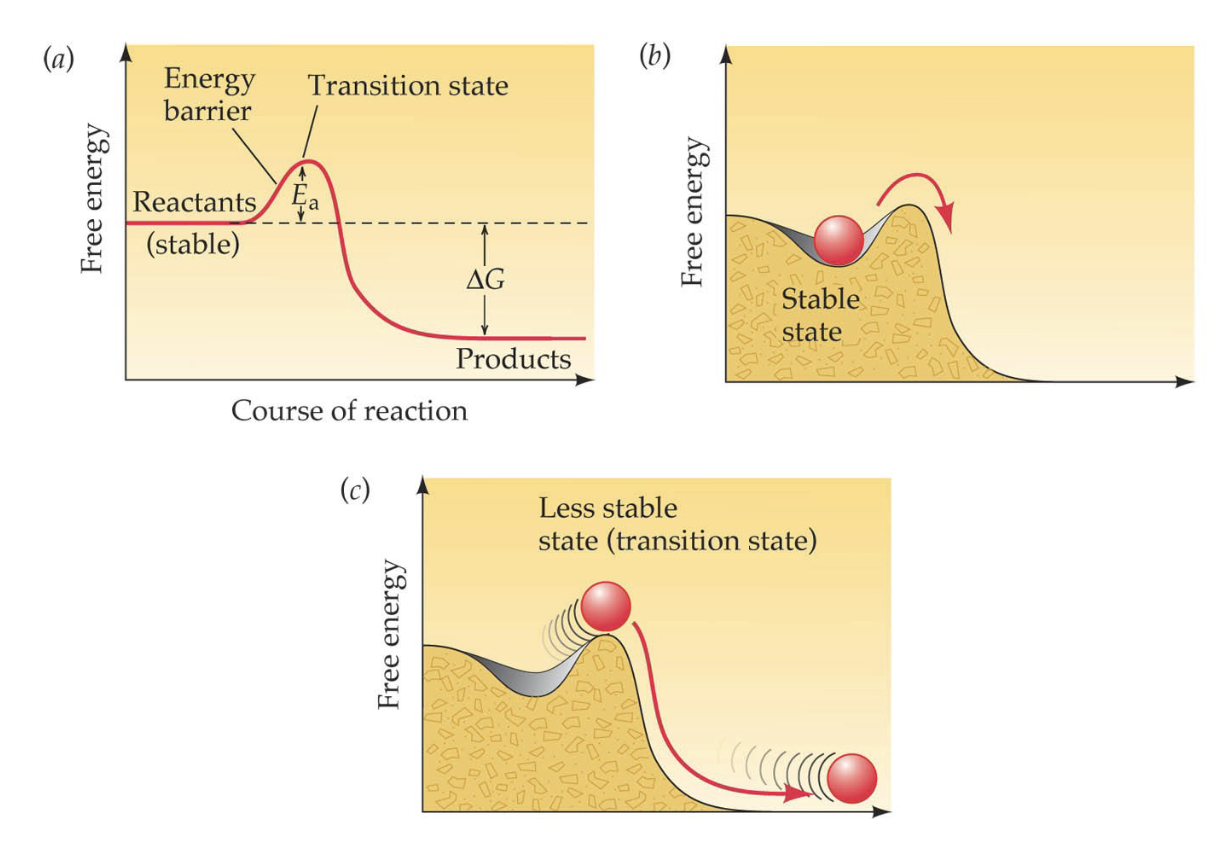
What is a Catalysis? What are they usually?
Any substance that speeds up a chemical reaction without itself being used up; usually an enzyme - they help overcome the need for activation energy
Do catalysis’s change ΔG values?
No!
ONLY reactions with overall (pos/neg) ΔG can be catalyzed
Negative ΔG
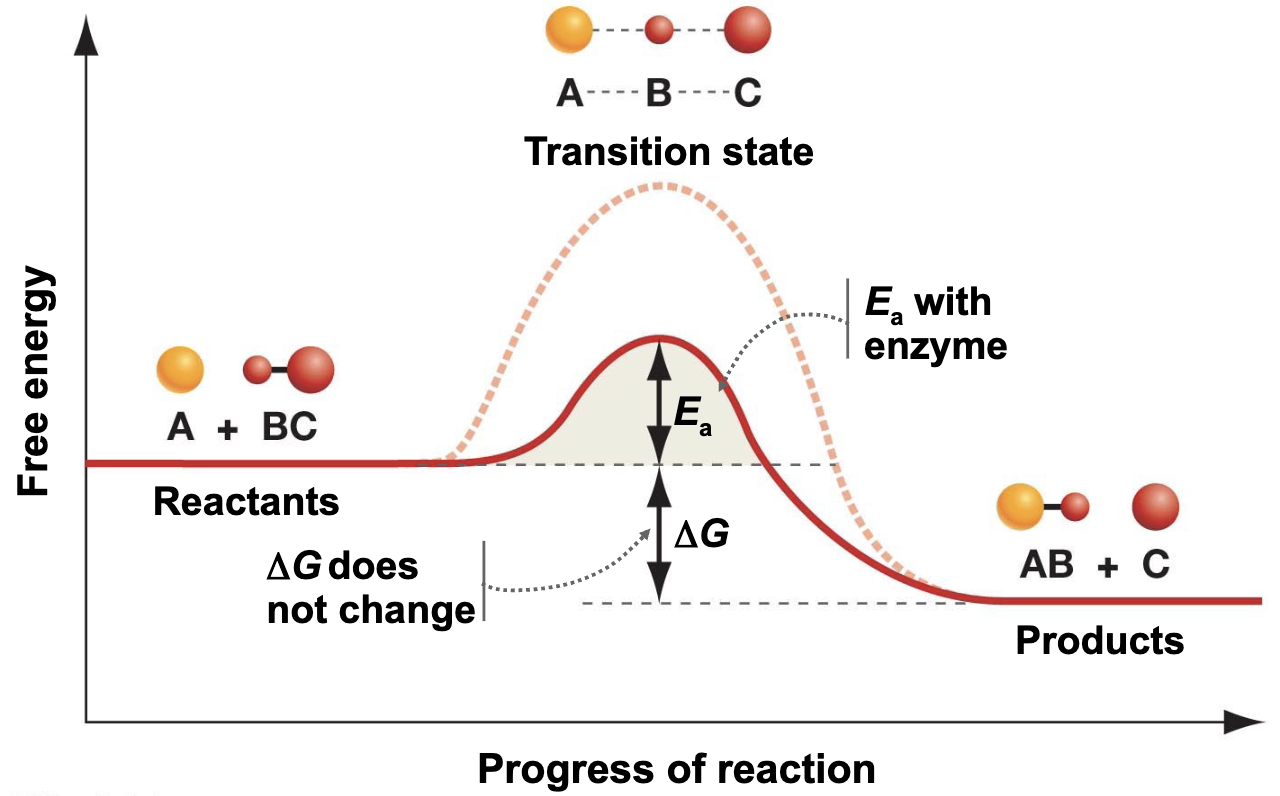
How do enzymes work?
They lower the activation energy → look at diagram
Initiation
Transition state facilitation
Termination
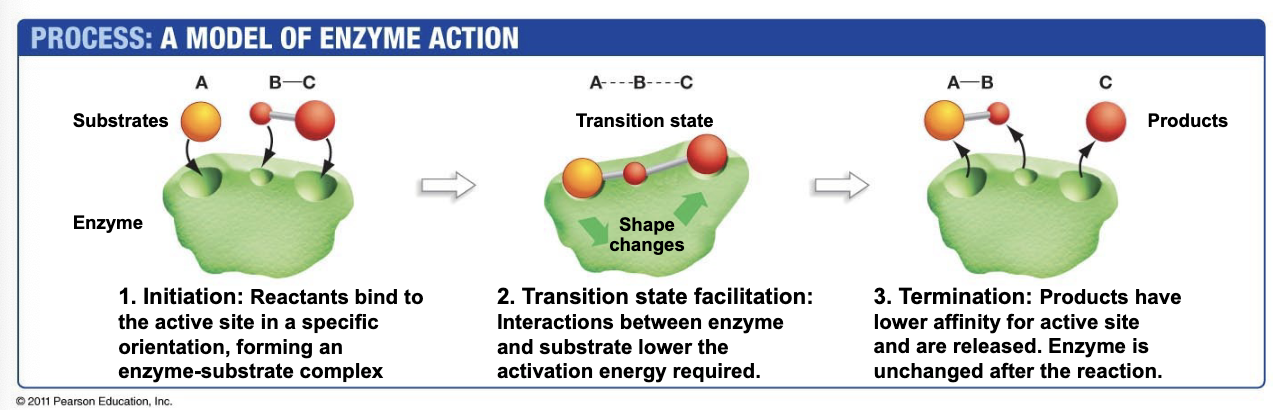
Some enzymes require ___ to function?
Cofactors (can be metal ions or small organic molecules that are either temp or perm bound to the enzyme)
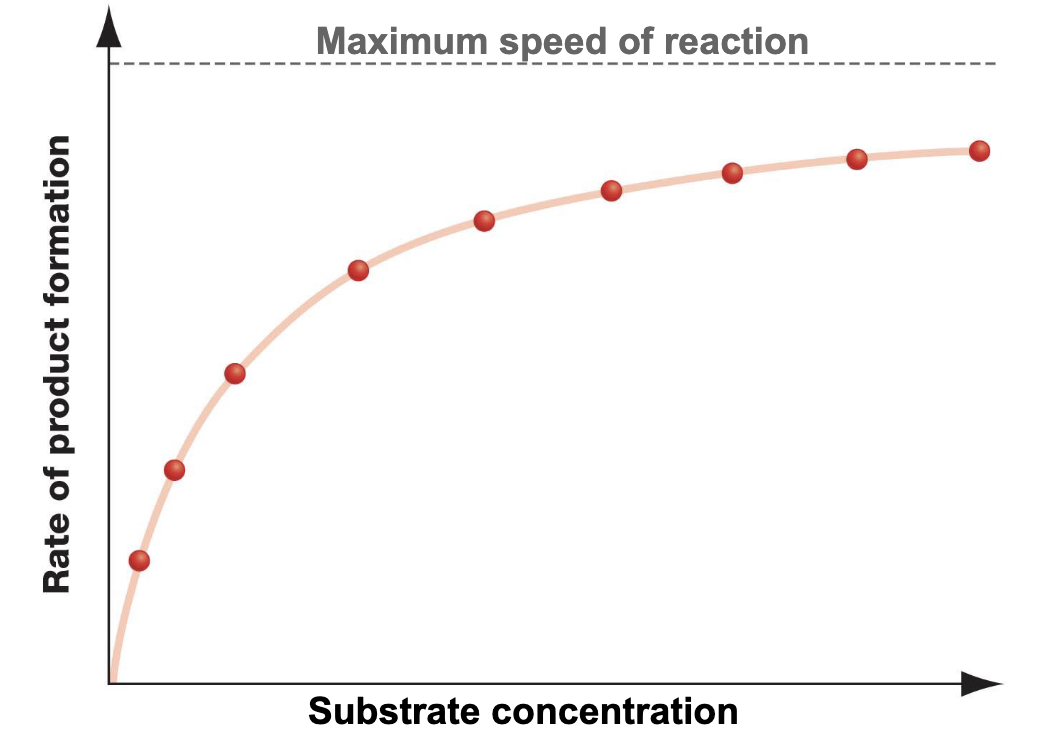
What does it mean for an enzyme to be saturated?
All binding sites are occupied