Lesson 1 - Introduction to Chemistry
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is the atomic number for Hydrogen, Carbon, Nitrogen , Oxygen
1, 6, 7, 8
Atomic number = Number of electrons =
Number of protons
Atomic mass =
number of protons and neutrons
Isotopes is an element with the same atomic number but a …
different mass, differnt number of neutrons
This serves as a passage of electrons around the nucleus
Electron Shell
The inner shell can accomodate at most __ electrons
2
The outer shells can accomodate _____ electrons per shell
8
Molecules are formed by combination of
Atoms
In molecules, atoms are joind together by
Chemical bonds
If the outer most shell is full it ______ form chemical bonds
Cannot
If the outer most shell is NOT full it ______ form chemical bonds
Can
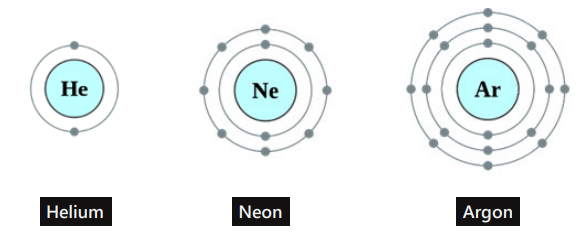
Inert gasses _____
Do not form chemical bonds
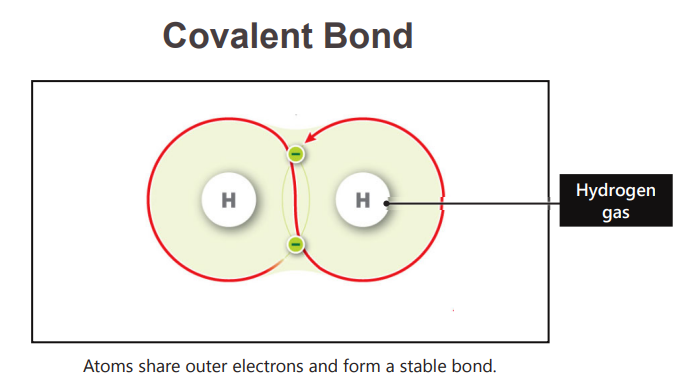
What is the strongest chemical bond?
Covalent bonds
What is the second strongest chemical bond?
Ionic Bonds
What is the weakest chemical bond?
Hydrogen bonds
In this bond, the atoms share the outer elctrons to form a stable bond
Covalent bond
In non polar covalent bonds, atoms are attraced by….
Equal Forces
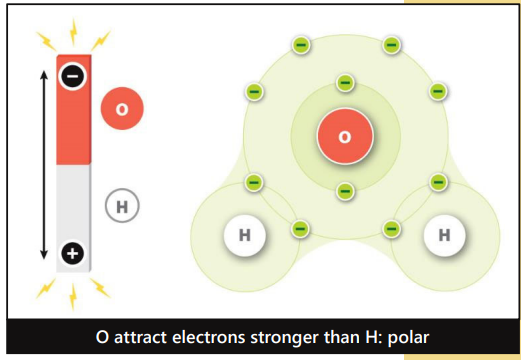
In polar covalent bonds, there is an ___________ between atoms, creating small charges
Unequal Pull
A water molecule is made via a
polar covalent bond
An electron moving from one shell (becomes positive) of an atom into the outer shell of another atom (becomes negative) is
An example of an ionic bond (there is an attraction due to electric charges from an ionic bond)
A slightly positive hydrogen of polar molecule is attracted to slightly negative atom ____
An example of hydrogen bond
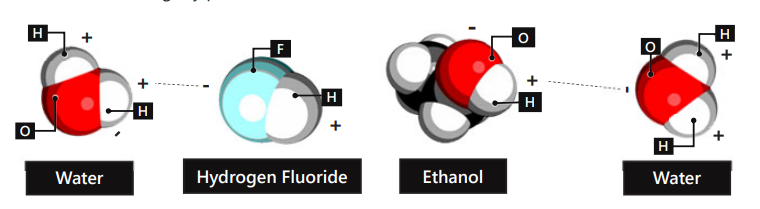
Polar-covalent bond is found ____ a water molecule
Within
Hydrogen bonds are found _____ water molecules
Hydrogen bonds
Hydrogen bonds make water molecules “stick together”. This is an example of
Cohesion
Cohesion gives water ____
Surface tension, net-like properties
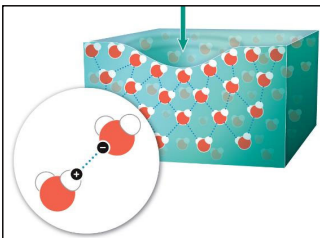
Heat from the sun ____ rather than increasing water temperature
rearranges hydrogen bonds
Water, when solid, has ____
Low density
This is a liquid that dissolves other substances
Solvent
This substance gets dissolved in a solvent
Solute
This is a mixture of solute and solvent
Solution
In hydrophilic, molecules with _____ dissolves in water
Charges (e.g. salt, sugar), ionic and polar bonds
Non-polar molecules do not dissolve in water. This is …..
Hydrophobic

Ok got it water
In ionization fo water, pure water contains an _______ of OH- and H+ ions
Equal amount
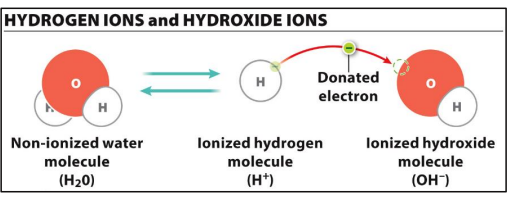
pH is an indicator of ______ in solution
H+ ions
The more H+ than OH-, the more
Acidic (reactive, sour)
The more OH- than H+ ions, the more __
Basic (caustic/burning to skin, slippery & bitter)