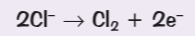C4 : Chemical Changes
1/100
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
What values are in the pH scale?
0 - 14
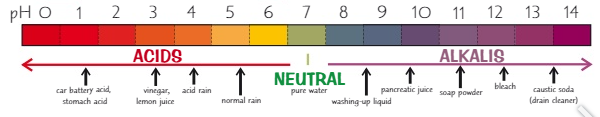
What is the pH scale a measure of?
How acidic or alkaline a solution is
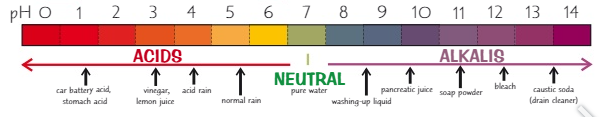
The lower the pH the more …… it is
acidic
What is the pH of an acid?
0 - 6
The higher the pH the more …… it is
alkaline
What is the pH of an alkali?
8 - 14
What is the pH of a neutral substance?
pH 7
What is an indicator + what is a wide scale indicator + give an example?
A dye that changes colour depending on whether its above or below a certain pH
A wide scale indicator is an indicator that is a mixture of dyes that means they gradually change colour
Universal indicator
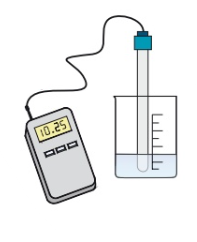
How can you measure pH electronically + what is the benefit of it?
A pH probe attached to a pH meter
More accurate
What is an acid?
A substance that forms an aqueous solution with a pH of less than 7
What are the 3 common acids + their formulas?
Hydrochloric acid → HCl
Sulfuric acid → H2SO4
Nitric acid → HNO3
What ion does an acid form in water?
H+ ion
What is a base?
A substance with a pH greater than 7
What is an alkali?
A substance that dissolves in water to form a solution with a pH of more than 7 (a type of base that are soluble in water)
What are the 2 common alkalis + their formulas?
Sodium Hydroxide → NaOH
Calcium Carbonate → CaCO3
What ion does alkalis form in water?
OH-
What is the reaction between acids and bases called + what is the equation for it?
Neutralisation

What is the chemical equation for neutralisation?

What is titration used for?
Find out to concentrations
Explain how to do titrations (8)
Using a pipette to transfer 25cm3 of sodium hydroxide solution (alkali) into a conical flask which reduces the risk of splashing
Add 5 drops of an indicator to the alkali in the conical flask
Place a conical flask on a white tile so we can clearly see a colour change
Fill a burette with sulfuric acid
Then add acid to the alkali, making sure to swirl the mixture and once a colour change is seen, add acid drop by drop until the acid turns neutral
Read the volume of acid added from the burette → make sure your eye is level with the surface of the liquid (meniscus)
Repeat this until you get 2 reading that are within 10cm3
Calculate a mean and remove anomalies
1cm3 = ….dm3+ Divide by what?
1cm3 = 0.001 dm3
Divide by 1000
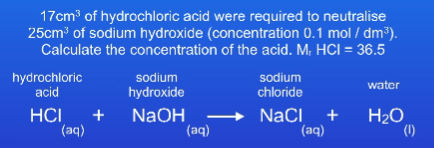
0.147 mol/dm3
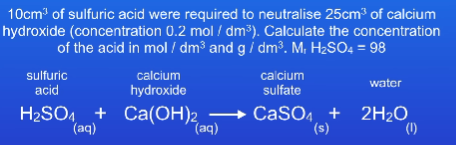
0.5 mol/dm3
49 g/dm3

0.25 mol/dm3
9.125 g/dm3

0.052 mol/cm3
5.096 g/cm3
What is a H+ ion?
A proton
What are the ions in HCl + HNO3
HCl → H+ + Cl-
HNO3 → H+ + NO3-
What happens in water for strong acids?
They ionise completely, all the acid particles dissociate to release H+ ion
What happens in water for weak acids?
They don’t ionise completely, only a small amount of the acid particles dissociate to release H+ ion
What type of reaction is the ionisation of weak acids + where does the equilibrium lay + why?
A reversible reaction
Equilibrium lies far to the left as there are more undissociated particles compared
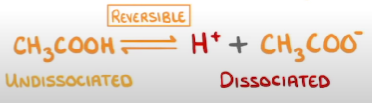
What is a reversible reaction?
When reactants are able to form products but then products can react to reform reactants
What happens if the concentration of H+ is higher in terms of reaction?
Rate of reaction is higher so stronger acids will be more reactive
What is pH?
Measure of the concentration of H+ ions
What is the difference between acid strength and acid concentration?
Strength → How much an acid dissociates
Concentration → How much acid there is in a certain volume
What is the relationship between the decrease of 1 on the pH scale and the concentration of H+ ion?
For every decrease of 1 on the pH scale, the concentration of H+ ion increases by a factor of 10
What pH would a stronger acid have compared to a weak acid at any given concentration?
A stronger acid would have a lower pH compared to a weak acid at any given concentration
What 3 bases used for neutralisation reactions?
Metal oxides
Metal hydroxides
Metal carbonates
What is the neutralisation reaction between an acid and metal oxide?

What is the neutralisation reaction between an acid and metal hydroxide?

What is produced in the reaction of hydrochloric acid + copper oxide?
Copper chloride + water
What is produced in the reaction of sulfuric acid + potassium hydroxide?
Potassium sulfate + water
What is the neutralisation reaction between an acid and metal carbonate?

What is produced in the reaction of hydrochloric acid + sodium carbonate?
Sodium chloride + water + carbon dioxide
Explain how to make insoluble salts using an insoluble base (5)
Gently warm dilute acid using a bunsen burner then turn off the bunsen burner
Add the insoluble base into the acid a bit at a time until no more reacts (excess solids will start to clump at the bottom)
Filter out the excess solid to get the salt solution
To get pure solid crystals, heat the solution using a water bath and then stop heating it leaving it to cool
Crystals should start to form which can be filtered out of the solution and left to dry
State the reactivity series from most reactive to least reactive
Potassium – Please
Sodium – Stop
Lithium – Licking
Calcium – Calum
Magnesium – My
Carbon – Car (feels)
Zinc – Zesty
Iron – I
Hydrogen – Hate
Copper – Coffee
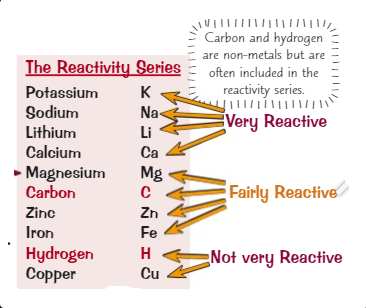
What is the reaction between an acid and a metal?

What is the speed of a reaction indicated by?
The rate at which hydrogen bubbles are given off
What is the relationship between the reactivity of the metal and the speed of the reaction?
The more reactive the metal, the faster the reaction
What do metals more reactive than hydrogen do?
Displace hydrogen
What does the salt end with which is produced by hydrochloric acid?
-chloride
What does the salt end with which is produced by sulfuric acid?
-sulfate
What is the reaction between hydrochloric acid + magnesium and what is visibly seen during this reaction?
Hydrochloric acid + magnesium → magnesium chloride + hydrogen
Magnesium reacts vigorously and produces lots of bubbles
What is the reaction between hydrochloric acid + zinc and what is visibly seen during this reaction?
Hydrochloric acid + zinc → zinc chloride + hydrogen
Zinc reacts slowly but more strongly if heated up
What is the reaction between hydrochloric acid + iron and what is visibly seen during this reaction?
Hydrochloric acid + iron → iron chloride + hydrogen
Iron reacts slowly but more strongly if heated up
What is the reaction between metal and water?

Which metals react with water?
Potassium – Please
Sodium – Stop
Lithium – Licking
Calcium – Calum
What is the reaction between potassium + water?
Potassium + water → potassium hydroxide + hydrogen
Which metals won’t react with water?
Zinc
Iron
Copper
What is oxidation?
When metals react with oxygen forming oxides (gain of oxygen)
What is reduction?
A reaction that separates a metal from its oxide (loss of oxygen)
What is the reaction for the oxidation of magnesium?
magnesium + oxygen → magnesium oxide
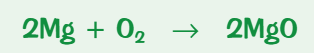
What is the reaction for the reduction of copper?
Copper oxide + carbon → copper + carbon dioxide

What are the 2 ways metals can be extracted?
Electrolysis
Reduction using carbon
Which 3 metals are extracted by carbon + why?
Zinc
Iron
Copper
Because they are below carbon in the reactivity series and carbon can only take the oxygen of metals which are less reactive than carbon itself

What is oxidation in terms of electrons + what is reduction in terms of electrons? OILRIG
Oxidation
Is
Loss (of electrons)
Reduction
Is
Gain (of electrons)
What is a redox reaction?
Reduction or oxidation → transfer of electrons
What will a more reactive metal do to a less reactive metal?
A more reactive metal will displace a less reactive compound from its compound
What is the reaction between hydrochloric acid + magnesium + what are the 2 oxidation and reduction half equations?
The reaction
Hydrochloric acid + magnesium → magnesium chloride + hydrogen
Half equations
Oxidation
Mg → Mg2+ + 2e-
Reduction
2H+ + 2e- → H2
What is the reaction between iron + copper sulfate + what are the 2 oxidation and reduction half equations?
The reaction
Iron + copper sulfate → iron sulfate + copper
Half equations
Oxidation
Fe → Fe2+ + 2e-
Reduction
Cu2+ + 2e- → Cu
In displacement reactions what always gains the electrons + is reduced?
Metal ion
In displacement reactions what always loses the electrons + is oxidised?
Metal atom
Why can solid ionic compounds not conduct electricity?
Because the ions are locked in place and are not free to move
What happens when an ionic compound is melted or dissolved in water + what does this lead to + what are the liquids called?
The forces of attraction are broken between particles and the ions are free to move
This leads to the liquid being able to carry charge
This is called an electrolyte
What is another word for melted?
Molten
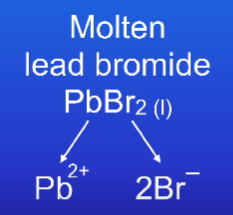
What are the 2 ions in molten lead bromide PbBr2?
What are electrodes made of + give an example?
Conducting materials such as graphite or a metal
What is the negative electrode called + what terminal of the powerpack is is connected to?
Cathode
Connected to the negative terminal of the power pack
What is the positive electrode called + what terminal of the powerpack is is connected to?
Anode
Connected to the negative terminal of the power pack
What type of ions are connected to the cathode + why?
Positive ions because opposites attract
What is the lead ion + what is the bromide ion?
Lead ion → Pb2+
Bromide ion → Br-
What happens at the cathode in the electrolysis of lead bromide + what is this type of reaction called?
The Pb2+ ions gain 2 electrons and form Pb atoms
A reduction reaction
Write the half equation for the reaction at the cathode for the electrolysis of lead bromide

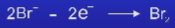
What happens at the anode in the electrolysis of lead bromide + what is this type of reaction called?
The Br- ions loses an electron and joins another Br atom to form Br2
An oxidation reaction
Write the half equation for the reaction at the anode for the electrolysis of lead bromide
What metals does electrolysis extract?
Metals that are more reactive than carbon
What are the aluminium ion + what is the oxide ion?
Aluminium ion → Al3+
Oxide ion → O2-
Explain the electrolysis of aluminium oxide (4)
First, aluminium oxide is mixed with cryolite which lowers the melting point saving money and reducing energy needed
Then set up the apparatus
The aluminium ions are attracted to the cathode, they then gain 3 electrons in a reduction reaction and form aluminium atoms
The oxygen atoms are attracted to the anode, they lose 2 electrons in an oxidation reaction and form oxygen atoms which then combine to form oxygen molecules
Write the half equation for the reaction at the cathode for the electrolysis of aluminium oxide

Write the half equation for the full reaction at the anode for the electrolysis of aluminium oxide

Why are the anodes replaced regularly?
The anode is made out of graphite (carbon) and this reacts with oxygen to produce carbon dioxide
Why is electrolysis expensive? (2)
Melting compounds requires a lot of energy
A lot of energy is required to produce a current
When water molecules ionise (split) what is produced?
Hydrogen ions (H+)
Hydroxide ions (OH-)
In electrolysis aqueous solutions what is produced at the cathode if the metal is more reactive than hydrogen + if the metal is less reactive than hydrogen?
More reactive → Hydrogen gas
Less reactive → A solid layer of pure metal
In electrolysis aqueous solutions what is produced at the anode if halide ions are present + if halide ions aren’t present?
Halide ions are present → Molecule of halide will be formed
Halide ions aren’t present → OH- are discharged and oxygen is formed
What ions are present in the aqueous solution copper sulfate?

Write the half equation for the reaction at the cathode for the electrolysis of the aqueous solution copper sulfate

Write the half equation for the reaction at the anode for the electrolysis of the aqueous solution copper sulfate
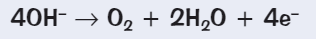
What ions are present in the aqueous solution sodium chloride?

Write the half equation for the reaction at the cathode for the electrolysis of the aqueous solution sodium chloride

Write the half equation for the reaction at the anode for the electrolysis of the aqueous solution sodium chloride