UNIT 1 - Biological m
1/81
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
what is a monomer
Monomers are the smaller units from which larger molecules are made
what are polymers
Polymers are molecules made from a large number of similar monomers joined together
what are examples of monomers
Monosaccharides
amino acids
nucleotides
what is a condensation reaction?
A condensation reaction joins two molecules together with the formation of a chemical bond and involves the elimination of a molecule of water
what is a hydrolysis reaction
A hydrolysis reaction breaks a chemical bond between two molecules and involves the use of a water molecule.
what elements make a carbohydrate
carbon, hydrogen and oxygen.
The hydrogen and oxygen atoms are present in the ratio of 2 hydrogen : 1 oxygen.
what 3 groups can carbs be classified into?
monosaccharides
disaccharides
polysaccharides.
what are monosaccharides? what are they used for and what are some examples of them?
what kind of sugars are they?
Monosaccharides are the basic molecular units (monomers) of carbohydrates.
They are mainly used in respiration to provide energy and in growth during the formation of larger carbohydrates
glucose
fructose
galactose
they are reducing sugars - give a positive test in the benedict’s test
how are glucose, fructose and galactose isomers of one another?
all have the same formula C6H12O6 (they are hexoses) but different structures
name and suggest how you can differentiate between the two different isomers of glucose
two isomers: alpha glucose (α-glucose) and beta glucose (β-glucose).
ABBA
- alpha glucose has both hydroxide groups below while the beta glucose has on of the hydroxide groups above
therefore - alpha below, beta above
what are disaccharides
A disaccharide is formed when two monosaccharides are joined together by a glycosidic bond formed in a condensation reaction
what are examples of disaccharides and what are they formed from?
maltose
lactose
sucrose
glucose + glucose → maltose + water
glucose + fructose → sucrose + water
glucose + galactose → lactose + water
C6H12O6 + C6H12O6 → C12H22O11 + water
how can disaccharides be hydrolysed into their respective monosaccharides?
Disaccharides can be hydrolysed by heating with acid e.g. dilute HCl, or by an enzyme.
what is the test for reducing sugars and how do you carry it out?
Benedict’s test
A small amount of the sample is placed in a test tube with the same volume of Benedict’s solution.
This is heated to 95°C in a water bath.
A brick red or orange precipitate is a positive result.
If the Benedict’s solution remains blue, no reducing sugar is present.
what is a semi quantitative benedict’s test and why is it useful?
This is useful when comparing the concentration of reducing sugar in different solutions/samples based on the colour (appearence)
control variables must be standardised
resulting in different colours of precipitates being formed
the intensity of the colour produced depends on the concentration of the precipitate produced
problem with this is that the colour is subjective when judging the colour
what is a quantitative benedict’s test and why is it useful?
This is carried out to obtain numerical data to compare the concentration of reducing sugar in different samples. It is an objective test (not influenced by opinion).
Perform the Benedict’s test on reducing sugar solutions of known concentrations.
Control variables must be standardised: add the same volume of sugar solution and same volume of Benedict’s solution and heat in a water bath for the same length of time at the same temperature.
Use a colorimeter to measure the absorbance of each of the known solutions.
Plot a graph of known concentration (on x-axis) against absorbance value (on y-axis). This is called a calibration curve. Add a line of best fit
Repeat the Benedict’s test with the unknown sample (using the same volume of sample, same volume of Benedict’s and heat in a 95°C water bath for the same amount of time).
Use the absorbance value of the unknown to read off the calibration curve to find its concentration
how do you do a test for a non- reducing sugar
non reducing sugar is sucrose
Take a small sample and heat in a 95°C water bath with Benedict’s solution to confirm a negative result.
Hydrolyse another sample of the sucrose by heating in a 95°C water bath with dilute acid (e.g. HCl).
When cooled, neutralise the acid with an alkali (e.g. NaOH).
Add the same concentration of Benedict’s solution and heat in a 95°C water bath.
A positive brick red colour (precipitate) indicates a non-reducing sugar (sucrose) was originally present
what are examples of polysaccharides and what are they all made of?
cellulose - beta glucose
starch - alpha glucose
glycogen -alpha glucose
how do polysaccharides differ from the glucose molecules that they contain?
do not taste sweet and are relatively insoluble in water
are non-reducing – giving a negative result in the Benedict’s test
function as storage (starch and glycogen) or structural molecules (cellulose).
what is starch and where is it found?
give properties of starch and why it is useful as a storage compound
This is the storage carbohydrate found in plants, stored in starch grains in the cytoplasm of plant cells.
it consists of long, branched chains of α-glucose molecules linked together by glycosidic bonds as a result of condensation reactions
it is large so it cannot cross the cell-surface membrane and leave the cell
it is insoluble and therefore osmotically inactive (i.e. it does not draw water into a cell by osmosis which could lead to cell damage)
the molecule has a helical shape forming a compact store
it is branched so glucose is easily released for respiration
what is the test for starch and how do you carry it out?
iodine test:
Add 2-3 drops of iodine/potassium iodide solution.
If starch is present a blue / black colour is produced.
If no starch is present the iodine solution remains orange / yellow.
what is glycogen and where is it stored?
This is the storage carbohydrate found in the cytoplasm of animal cells. Glycogen is formed by the condensation of α-glucose. In mammals it is stored in large amounts in liver and muscles tissues.
Glycogen is ideally suited to its function as a storage compound as:
it is insoluble and therefore osmotically inactive i.e. it does not draw water into a cell by osmosis which could lead to cell damage
it has a similar structure to starch but with more branches (free ends) which means it can be hydrolysed more rapidly to release glucose for respiration. (more so than glycogen)
what is cellulose and where is it found?
cell wall of plants, which provides rigidity and shape to the cell preventing plant cell from bursting
polymer of β-glucose molecules joined together by glycosidic bonds, these bonds are formed in condensation reactions to produce long, straight chains of β-glucose molecules.
what is the structure of cellulose?
long, straight chains of β-glucose molecules.
glycosidic bonds
every other β-glucose monomer is inverted
hydroxyl (OH) groups project from either side of the β-glucose chain and form hydrogen bonds with the hydroxyl (OH) groups of other adjacent chains
producing a three-dimensional structure known as a microfibril
giving strength to the structure
this is because even though the hydrogen bonds are weak, overall their large number gives strength to the structure
what elements do lipids contain?
carbon, hydrogen and oxygen
what are the two types of lipids
triglycerides
phopholipids
what is the structure of a triglyceride?
have 3 fatty acids joined to one glycerol molecule.
They are joined in three condensation reactions with the loss of three water molecules.
A condensation reaction between glycerol and a fatty acid (R-COOH) forms an ester bond. One triglyceride molecule has 3 ester bonds.
what is the general formula of a fatty acid?
R-COOH
what does a saturated and an unsaturated fatty acid mean?
saturated - No double bonds between the carbon atoms
unsaturated: One or more double bonds between the carbon atoms
how can lipids be hydrolysed into fatty acids and glycerol.
heating with acid or alkali
using the enzyme lipase at its optimum temperature and pH.
why are lipids useful?
long term energy storage
organ protection
insulation
flexibility to the cell membrane
water - proofing (plants)
what are advantages of triglycerides?
possess a high proportion of carbon-hydrogen bonds so that they release twice as much energy as carbohydrates during aerobic respiration
high ratio of hydrogen to oxygen atoms, releasing water when respired - important for organisms in dry deserts
non - polar - insoluble in water and therefore osmotically inactive
helps reduce water loss - present in waxy cuticles of plants and insetcs
what is the structure of phospholipids
lipids containing a phosphate group
1 glycerol, 2 fatty acid and 1 phosphate group
The condensation reactions form ester bonds
The phospholipid molecule has a polar hydrophilic head (attracts water) containing the phosphate group, and a non-polar hydrophobic tail (repels water) consisting of the long fatty acids chains.
how are phospholipids relevant in cell membranes?
They form a bilayer (two layers) of phospholipids due to the hydrophobic tails being repelled by water which face towards each other.
what is the test for lipids and how do you carry it out?
Emulsion test -
a small amount of the sample is placed into a test tube with ethanol (alcohol)
shake the mixture so that the fat dissolves
add this to water in another test tube and mix the contents
a white emulsion of fat droplets indicates lipid is present.
what are similarities between phospholipids and triglycerides
are insoluble in water
contain glycerol
contain ester bonds
contain C, H and O but phospholipids also contain P
what elements are proteins made up of
carbon, hydrogen, oxygen, nitrogen and sometimes sulfur
what is the generalised structure of an amino acid?
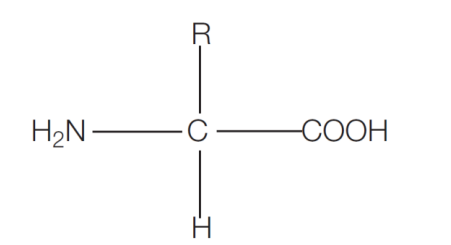
what are some properties of amino acids?
All amino acids have an amine group (NH2) and a carboxylic acid group (COOH) but differ in their carbon-containing side group R (as shown in the diagram above).
Amino acids are joined together by peptide bonds (-CONH-) formed by condensation reactions.
Two amino acids join together in a condensation reaction to form a dipeptide.
Many amino acids join together in a chain form a polypeptide.
A protein may consist of one or more polypeptides
how can protein be hydrolysed?
Proteins can be hydrolysed by heating with acid or by using enzymes (proteases).
what is the structure of a protein?
Primary structure refers to the sequence of amino acids in the polypeptide chain. This sequence determines the specific shape of the protein. - amino acids are joined together by peptide bonds
The secondary structure represents the folding or coiling of the polypeptide chain as a result of hydrogen bonding between amino acids. Secondary structures include the alpha helix and the beta-pleated sheet.
The tertiary structure is formed by further folding and coiling of the secondary structure due to hydrogen bonds, ionic bonds and disulfide bonds. - bonds are dependant on the sequence of amino acids in primary structure
Quaternary structure is when proteins consist of more than one polypeptide chain. The polypeptide chains are held together by ionic bonds, hydrogen bonds and sometimes disulfide bonds
why do proteins have a variety of roles
Tertiary structures are specific and unique,
what determines the shape of an enzymes active site?
The tertiary structure of an enzyme determines the shape of its active site and its precise function.
what is a globular protein and state some examples
Globular proteins are soluble and consist of a highly folded and coiled polypeptide chain to produce a compact, complex tertiary structure. They include enzymes and antibodies
what is a denaturation of a protein and what are some reasons that could cause it?
Denaturation is an alteration in the tertiary structure of a protein. This loss of the three-dimensional shape of the protein is often irreversible and the protein is no longer functional.
is usually caused by the breaking of hydrogen and ionic bonds;
can be caused by high temperatures above the optimum, extreme changes in pH and by heavy metals.
Disulfide bonds are not broken at the temperatures that break hydrogen and ionic bonds. In some proteins disulfide bonds can remain unbroken at 70 0C.
what is the test for protein and how can you perform it?
biuret test
add Biuret reagent
a purple or lilac colour indicates protein is present
if the solution remains blue, no protein is present.
describe the structure of water
A water molecule consists of two hydrogen atoms covalently bonded to an oxygen molecule.
Weak hydrogen bonds form between water molecules and these are responsible for many of the properties of water.
what are the properties of water and their respective functions
Metabolic reactions - Water is a metabolite in many metabolic reactions:
including condensation and hydrolysis reactions
required for photosynthesis
a metabolic product of respiration which is important for organisms living in dry habitats.
Solvent -
to transport nutrients e.g. glucose in blood, and sucrose in phloem of plants
to remove excretory products e.g. urea, ammonia
as a medium in which metabolic reactions occur.
Temperature regulation -
water has a high heat capacity - absorbs large amounts of heat before rising in temperature
buffers temp changes
it minimises the increase in temperature within cells as a result of biochemical reactions
it helps to prevent dangerous fluctuations in temperature in aquatic habitats.
Water has a relatively large latent heat of vaporisation, as energy is required to break the hydrogen bonds. This provides a cooling effect with little loss of water through evaporation
helps some animals to maintain a constant body temperature, as a high amount of heat energy is removed from the body to evaporate sweat
has a cooling effect in plants via transpiration.
Support -
Internal support
Cohesive forces between water molecules due to hydrogen bonding support water columns in the tube-like transport cells (e.g. xylem) of plants, allowing transport from roots to leaves
Water is not easily compressed and therefore provides support in non-woody plants via turgor pressure and in some animals, e.g. hydrostatic skeleton in earthworms
External support:
Strong cohesive forces between water molecules due to hydrogen bonding produce surface tension where water meets air, which enable insects to walk on the surface of water (e.g. pond skaters) or be suspended at the surface (e.g. mosquito larvae)
Water provides buoyancy for aquatic organisms e.g. whales.
what are some inorganic ions and what are their roles?
Sodium - Co-transport of glucose and amino acids across cell membranes
Iron - Component of haemoglobin which transports oxygen
Hydrogen - Important in determining pH and thus affecting protein structure and enzyme activity
Phosphate - Important as a structural component of DNA, RNA and ATP
what are enzymes?
Enzymes are biological catalysts regulating biological processes in living organisms.
what is an important feature of enzyme?
lowering the activation energy
The activation energy is the energy required for chemical reactions to take place
By lowering the activation energy the rate of reaction is increased
Enzymes lower the activation energy through the formation of enzyme-substrate complexes
what does it mean if an enzyme is specific?
Enzyme specificity is a feature of the unique tertiary structure of an enzyme.
This tertiary structure determines the shape of the active site.
Only molecules complementary in shape to the active site will bind to it to form an enzyme-substrate complex.
Initially the shape of the active site was considered to be fixed or rigid. This was known as the Lock and Key model.
what is the induced fit model?
suggests that the active site of an enzyme is flexible and can slightly change its shape. The presence of the correct substrate induces the conformational change in the shape of the active site so that it moulds itself around the substrate
The active site and the substrate are now complementary in shape and form an enzyme- substrate complex
explains how an enzyme catalyses more than one substance
what is an active site?
small section of an enzyme where a substrate binds to the enzyme to undergo a reaction
what are factors that effect the rate of enzyme reactions?
substrate concentration
enzyme concentration
temperature
pH
elaborate how substrate concentration effects an enzyme reaction
For a given concentration of enzyme, increasing the substrate concentration will increase the rate of reaction to a particular point at which the rate reaches a constant maximum rate; Vmax.
The rate of reaction initially increases as collisions between substrate and enzyme molecules are more likely.
The rate of reaction then levels out as the active sites of all the enzyme molecules are taken up by substrate molecules.
The active sites are all occupied (saturated).
At this point the rate of reaction is limited by the concentration of enzyme.
The only method of increasing the rate of reaction at these higher substrate concentrations is by the addition of more enzyme
what is a saturation point?
when all of the enzyme active sites are occupied - limiting factors are present
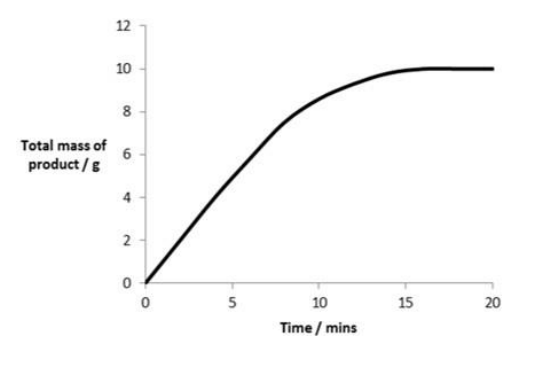
describe and explain this graph
This graph shows how the concentration of product formed in an enzyme-controlled reaction changes over time.
Initially the product is formed at a fast rate as there is a high concentration of substrate.
As the reaction progresses, the concentration of substrate decreases and so the rate at which the product is formed also decreases.
This is because the collisions between the substrate and active site are less frequent, so less enzyme-substrate complexes are formed.
The concentration of product then levels off as all of the substrate has been converted to product
elaborate how enzyme conc effects an enzyme reaction
When the concentration of substrate is in excess (i.e. it is abundant and not a limiting factor) then an increase in enzyme concentration increases the rate of reaction.
This is because when there are more enzyme molecules there are more active sites available.
This increases the number of collisions between active sites and substrates to form more enzyme-substrate complexes
elaborate on how temperature effects the enzyme reaction, what happens if you increase the temp beyond the optimum value?
An increase in temperature provides the molecules with more kinetic energy resulting in more collisions between the active site of enzymes and substrate molecules and therefore more enzyme-substrate complexes are formed.
enzymes less efficient at low temp due to less kinetic energy of reactant molecules
rate of reaction increases till the optimum temperature (rate is maximum)
if you increase the temp beyond optimum temp - causes the tertiary structure of the enzyme to denature as hydrogen and ionic bonds are broken.
rate of reaction decreases as the substrate has difficulty binding to the altered active site. The active site has changed shape and is no longer complementary, so less enzyme-substrate complexes form
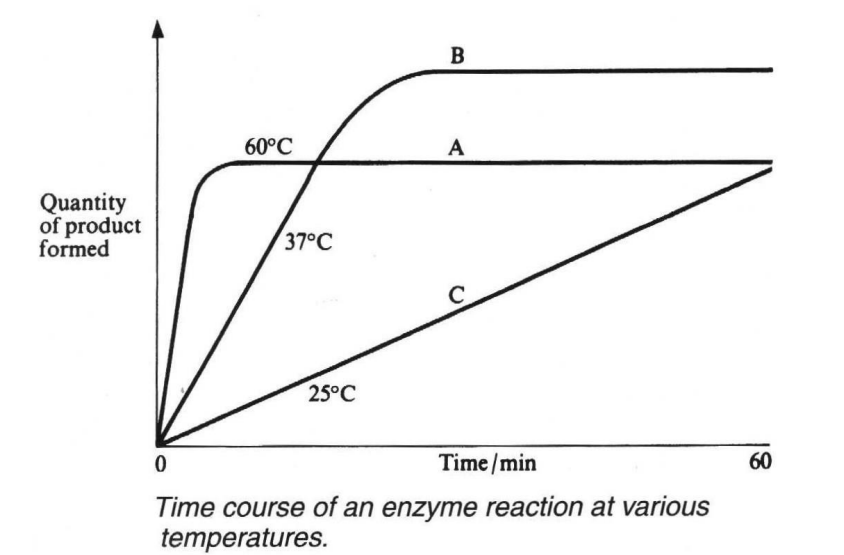
describe and explain the different temperatures drawn on this graph
At 25C (line C) there is low kinetic energy, so the reaction proceeds at a slow rate due to fewer collisions between enzyme and substrate molecules. Eventually all the substrate will be converted into product and line C will level out at the same point as line B. They level out at the same point because the initial substrate concentrations were the same, so the quantity of product will be the same.
At 37C (line B) there is more kinetic energy, therefore the reaction proceeds at a faster rate due more frequent collisions between enzyme and substrate molecules. All substrate is converted into product.
At 60C (line A) there is a very high level of kinetic energy so the reaction proceeds at a very fast rate initially due to many collisions between enzyme and substrate molecules. However, due to the very high kinetic energy hydrogen bonds begin to break, the enzyme molecules begin to denature, so that eventually substrate molecules cannot bind to the altered active site. The reaction stops before all the substrate is converted into product.
elaborate on the effect of pH in an enzyme reaction
Enzymes possess an optimum pH at which the rate of reaction is at a maximum.
Most enzymes are active over a narrow pH range
Very different pHs from the optimum can cause denaturation.
The change in pH alters the ionic charges of acidic and basic groups
Hydrogen and ionic bonds are broken altering the tertiary structure and shape of the active site so that the substrate cannot bind.
re the two types of inhibitors
competitive
non- competitive
how does a competitive inhibitor work?
and how can it effect be reduced?
The inhibitor has a similar structure to the normal substrate molecule and competes with it for attachment to the active site.
The rate of reaction is reduced as the substrate cannot bind when the inhibitor molecule is occupying the active site
Competitive inhibition can be reduced by the addition of more substrate as at high substrate concentrations the substrate out competes the inhibitor molecules for attachment to the active site.
how does a non- competitive inhibitor work and how can its effect be reduced?
A non-competitive inhibitor is not similar in structure to the substrate.
It combines at a position other than the active site to form an enzyme/inhibitor complex, altering the tertiary structure and shape of the active site of the enzyme.
The substrate cannot attach to the altered active site as it is no longer complementary.
The degree of inhibition is completely dependent on the amount of inhibitor present.
A high concentration of substrate will not reduce non-competitive inhibition.
what is ATP? and what is it made up of?
Adenosine Triphosphate used by cells as the immediate energy source for biological processes.
formed from a molecule of ribose, a molecule of adenine and three phosphate groups
what is the enzyme used to hydrolyse ATP and what are the products of this hydrolysis?
ATP hydrolase- forms (ADP +Pi)
adenosine diphosphate and an inorganic phosphate
what can the inorganic phosphate further partake in?
used to phosphorylate other compounds, often making them more reactive
what is the enzyme used to reform ATP and what is it formed from
ADP+ Pi → ATP
with use of ATP synthase
AND the energy released from respiration / photosynthesis also used for resynthesis
compare the two graphs showing reaction of enzyme - when a competitive inhibitor is present and when there is a non- competitive inhibitor is present
non - comp:
v- max not reached as the enzyme’s tertiary structure is altered so the highest rate cannot be reached as there are less functioning enzymes
comp:
v-max is eventually reached as the inhibition is temporary and doesn’t necessarily result in non - functioning enzyme just the time at which it reaches v-max is longer (the graph is steeper)
what does DNA do? what does RNA do?
DNA holds genetic information and RNA transfers genetic information from DNA to the ribosomes.
what is the structure of a nucleotide?
a Pentose (five carbon sugar)
a nitrogen-containing organic base
and a phosphate group (phosphoric acid).
what is the structure of DNA?
It consists of two polynucleotide strands joined together by hydrogen bonding to form a double helix.
Each DNA nucleotide has:
the Pentose sugar Deoxyribose
a phosphate group
one of four nitrogen containing bases : Cytosine, Thymine, Adenine or Guanine
The nucleotides within each strand are joined together by phosphodiester bonds formed by condensation reactions.
The sugar and phosphate form the backbone of the polynucleotide strands.
The organic bases are orientated towards the centre of the helix, protecting them from reacting with other chemicals.
The bases on one of the strands have a specific complementary base pairing with the other strand i.e. Adenine always pairs with Thymine Guanine always pairs with Cytosine
The bases are joined by hydrogen bonds which, although individually weak, due to their large number collectively maintain a stable structure.
The DNA helix is further coiled to produce a super helix, providing a compact store of genetic information
what are some basic functions of DNA
Genes are sections of DNA which code for polypeptides which determine nature and development of organism
DNA has the ability to self-replicate (due to complementary base pairing) which is essential for cell division (mitosis and meiosis).
alterations in DNA base sequences can occur which cause genetic diversity, via natural selection - basis for evolution
what are components of RNA nucleotide
ribose
phosphate
one of the organic bases: adenine, cytosine, guanine, uracil (replaces thymine)
short polynucleotide chain
how did the structure of DNA lead scientists to believe that it did not contain the genetic codee?
the relative simplicity of DNA led them to not believe
what are ribosomes formed from?
formed from RNA and proteins
how is the structure of DNA adapted to its function?
Sugar-phosphate backbone -
Gives strength
Helix -
Gives compact shape
Protects sequence of bases
Double stranded -
Each strand serves as a template in replication
Protects sequence of bases
Makes molecule more stable
Large molecule -
Large amount of information can be stored
Many hydrogen bonds -
Gives stability
Individual hydrogen bonds are weak allowing helix to unzip easily for replication
Sequence of bases -
Codes for specific sequence of amino acids in a polypeptide
Complementary base-pairing -
Enables information to be replicated accurately
describe the process of semi- conservative DNA replication
the enzyme DNA helicase unwinds the double helix and breaks the hydrogen bonds between the complementary bases of the two polynucleotide strands.
Each of the strands acts as a template for the formation of two new complementary strands.
Individual DNA nucleotides align and attach by hydrogen bonding to the exposed bases of each template strand according to specific complementary base pairing.
Adenine pairs with Thymine and Cytosine pairs with Guanine.
The DNA nucleotides in each new strand are joined together by phosphodiester bonds in condensation reactions by the enzyme DNA polymerase to form complementary strands to the original DNA strands.
The two DNA molecules are identical to each other and to the original DNA.
Each newly formed DNA molecule contains one of the original polynucleotide strands and a new strand, hence the term semi-conservative replication