Chem 104 Final Flashcards
1/102
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
103 Terms
Linear
2 electrons bonded with 0 lone pairs
Trigonal planar
3 elecrons bonded with 0 lone pairs
Bent
2 electrons bonded with 1 lone pair
Tetrahedral
4 electrons bonded with 0 lone pairs
Triangular pyramidal
3 electrons bonded with 1 lone pair
Bent
2 electrons bonded with 2 lone pairs
Tetrahedral bond angle
~109.5
Trigonal planar bond angle
~120
Linear bond angle
180
Hybridized bond
First bond connecting two molecules, formed by combining s and p orbitals
Unhybridized bond
Subsequent bonds after first bond connecting two molecules, formed exclusively of p orbitals
sp bond
One hybridized orbital, present in an atom bonded to 2 other atoms
sp2 bond
Two hybridized orbitals, present in an atom bonded to 3 other atoms
sp3 bond
Three hybridized orbitals, present in an atom bonded to 4 other atoms
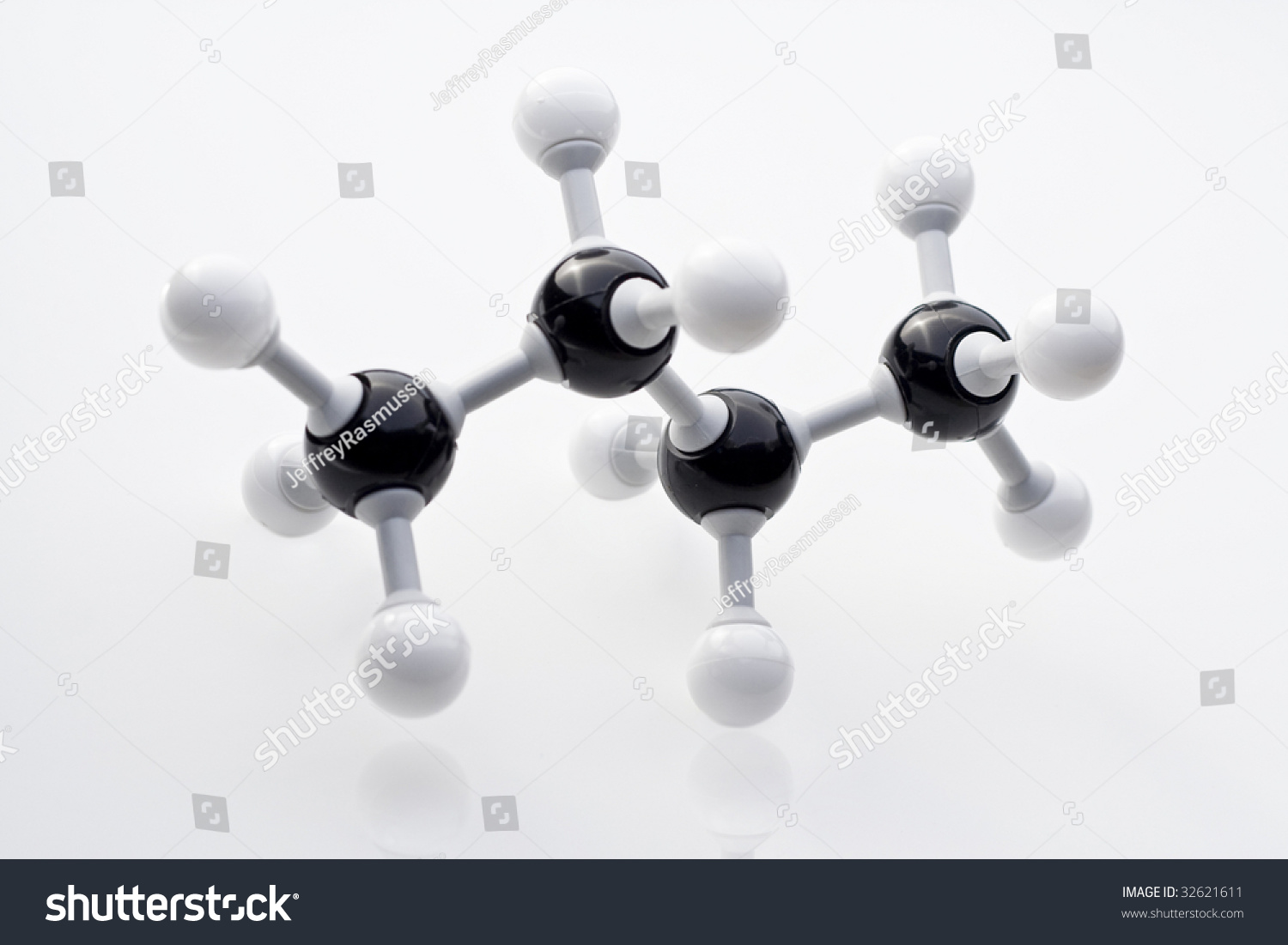
Ball-and-stick model
A 3d model of molecular structure with each atom represented physically as a “ball”
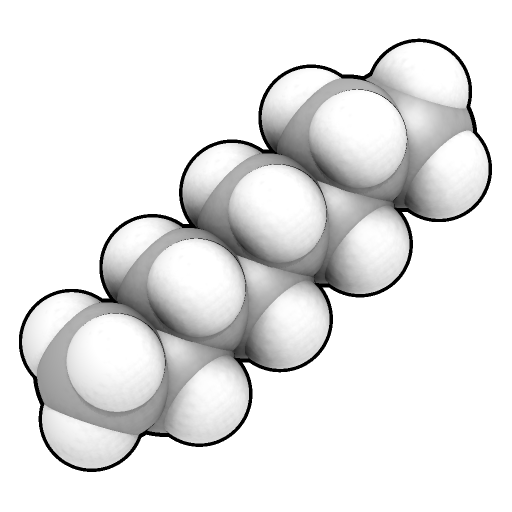
Space-filling model
A 3d model of molecular structure in which the distance between atoms is represented accurately to their actual radii
Molecular formula
The formula of a molecule with the proper number of each type of atom
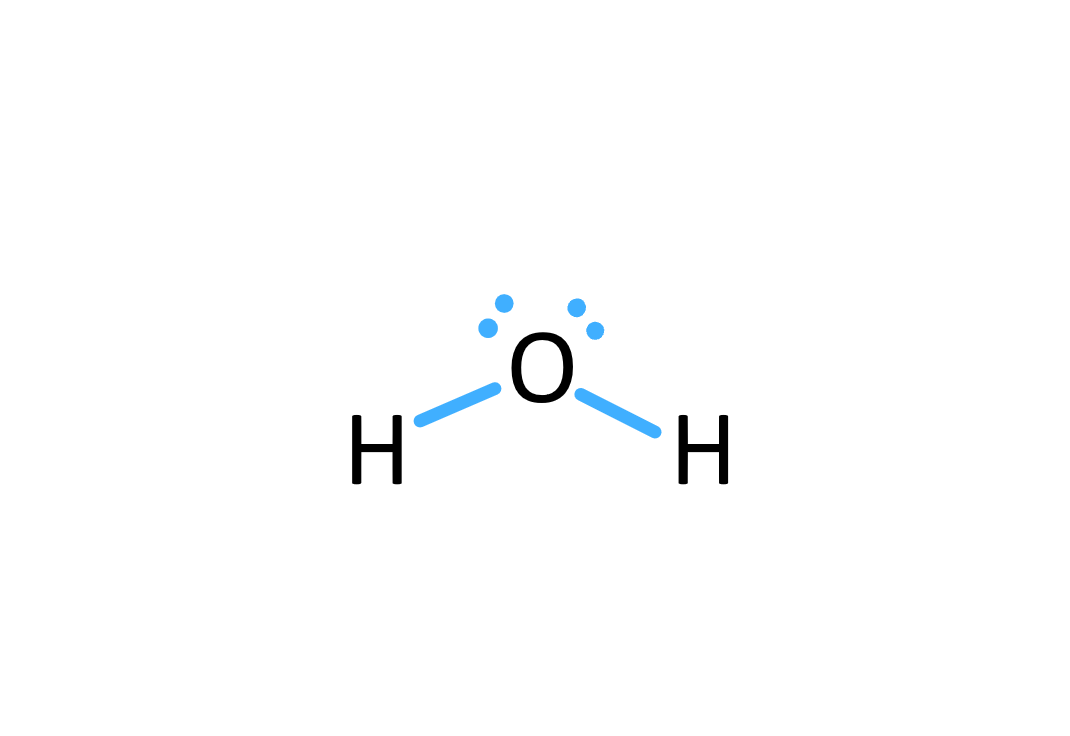
Lewis structure
A 2d representation of molecular structure in which bonds are represented by straight lines
Structural formula
A diagram of molecular structures similar to a lewis structure but without independent valence electrons
Condensed structural formula
A written out display of molecular structure where each hub is pronounced. Example: CH3CH2CH2CH3
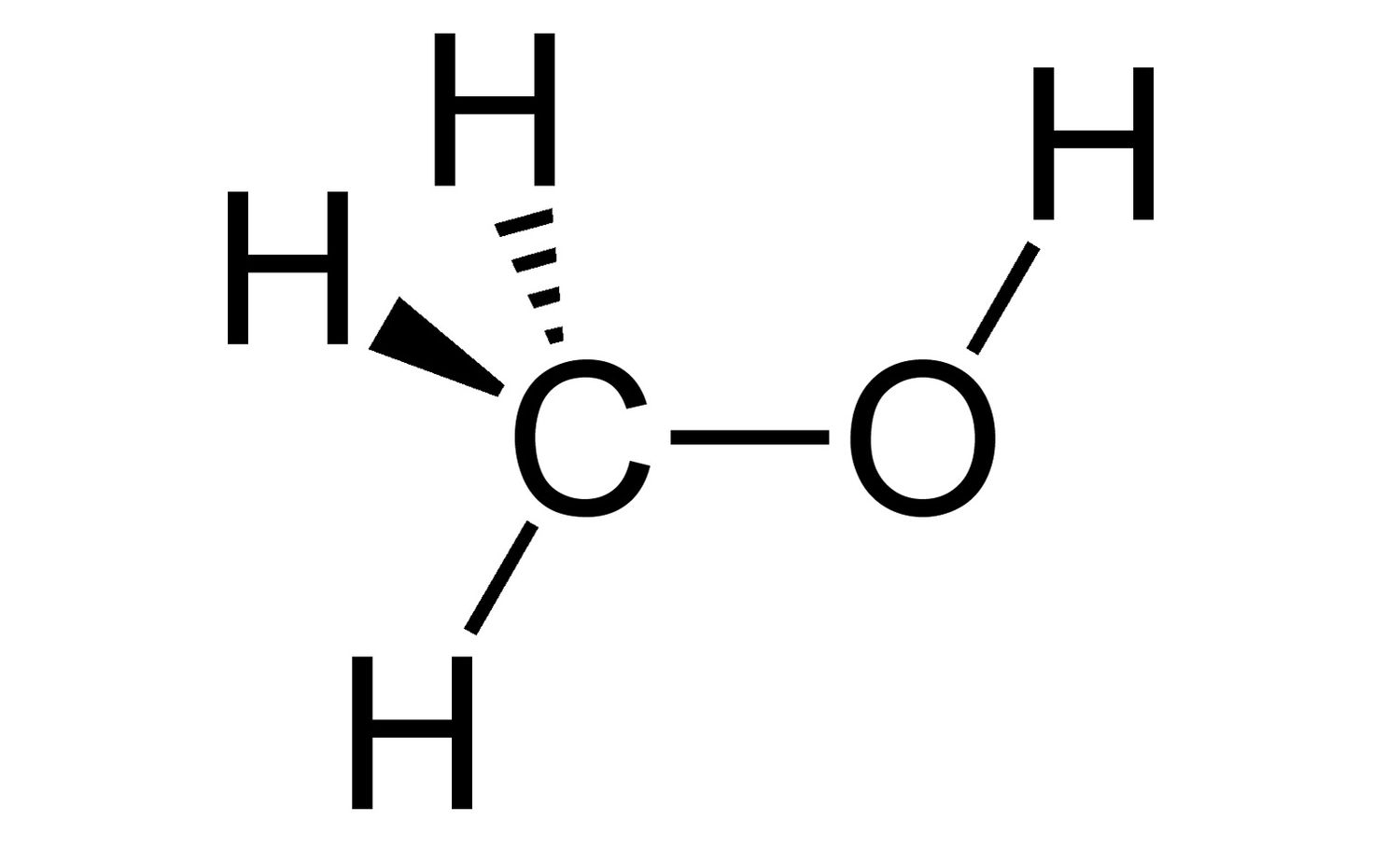
Dash-wedge model
3d model that uses “dashes” to represent bonds away from the plane and “wedges” for bonds moving forward from the plane.
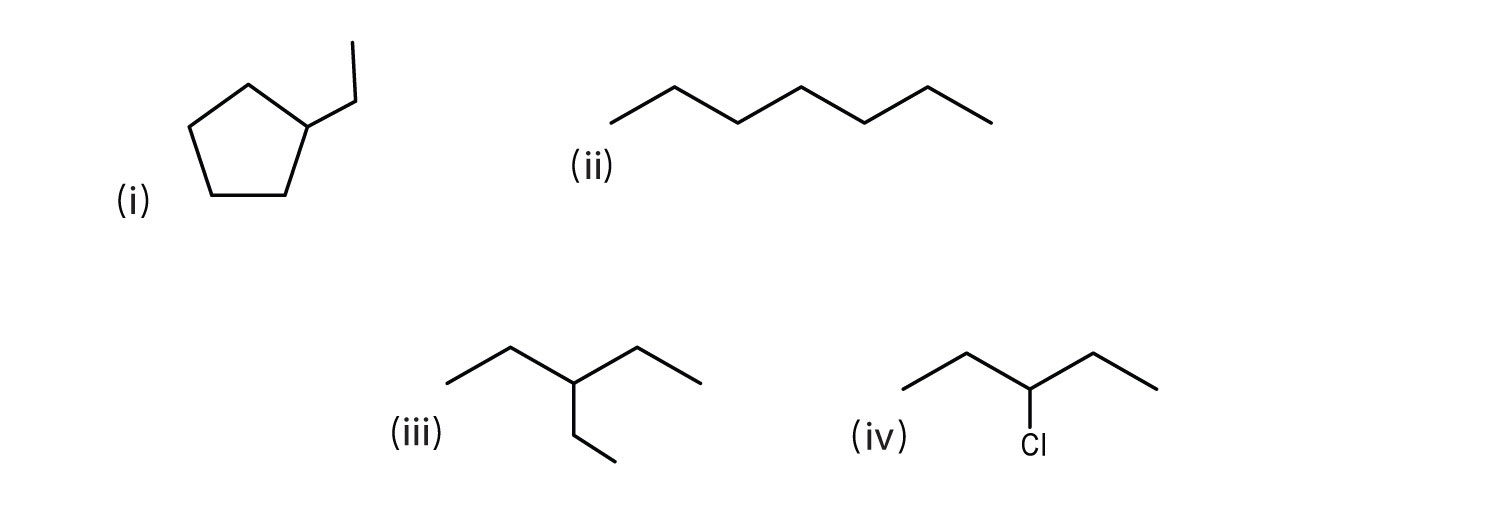
Line formula
A display of organic molecular structure in which a chain of carbons is drawn as diagonal lines, each apex representing a carbon, from which hydrogen bonds are not displayed but implied.
London dispersion forces
Type of IMF in which a temporary dipole is created as electrons are pulled to one side of a molecule.
Dipole-dipole forces
IMF from the attraction of two opposed permanent dipoles
Hydrogen bonds
IMF formed from the highly polar bonds between hydrogen and nitrogen, oxygen, and fluorine.
Chiral molecule
A molecule that is bonded to four entirely different groups that cannot form identical mirror images.
Chiral center
The central atom of a chiral molecule/molecular bond.
Alkanes
Singular bonds between two carbons or a carbon and a hydrogen
Cycloalkanes
Singular bonds among carbons and between carbons and hydrogens in a cyclical chain.
Alkenes
Double bonds between carbons
Alkynes
Triple bonds between carbons
Aromatics
A cyclo- organic molecule containing alternating double bonds between the carbons and an equal number of carbons and hydrogens. The “alternating” double bonds can alternatively be represented as electrons more thoroughly dispersed throughout the molecule.
Cis- isomer
A type of geometric isomer in which two atoms/groups of the same type are on the same side of a molecule. This type of molecule is more polar.
Trans- isomer
A type of geometric isomer in which two atoms/groups of the same type are on the opposite side of a molecule. This type of molecule is less polar.
Alcohol
A carbon bonded to an -OH group.
Ether
An oxygen bonded to two seperate carbon chains
Aldehyde
A carbon at the end of a chain double bonded to an oxygen.
Ketone
A carbon in the middle of a chain double bonded to an oxygen.
Carboxylic acids
A carbon double bonded to an oxygen and separately single bonded to an -OH group.
Ester
A carbon double bonded to an oxygen and separately single bonded to an oxygen. The oxygen is attached to another carbon chain.
Amine
A carbon bonded to a nitrogen
Amide
A carbon double bonded to an oxygen and separately single bonded to a nitrogen
Primary alcohol/amine
An alcohol/amine located at the end of a carbon chain
Secondary alcohol/amine
An alcohol/amine connected to a carbon that is connected to two other carbons.
Tertiary alcohol/amine
An alcohol/amine connected to a carbon that is connected to three other carbons.
Condensation reaction to form ester
A reaction of a carboxylic acid + alcohol to form an ester and water
Condensation reaction to form amide
A reaction between a carboxylic acid + a primary amine, secondary amine, or ammonia to form an amide + water. The nitrogen in the reaction MUST be attached to a hydrogen—the reaction does not work with a tertiary amine.
Hydrolysis of amide
A reaction between an amide + water to form a carboxylic acid + amine/ammonia.
Hydrolysis of ester
A reaction between an ester + water to form a carboxylic acid + alcohol.
Factors that increase the rate of reactions
Rising temperature, presence of catalyst, increased concentration/pressure, and more surface area for a reaction.
Average rate of reaction
The rate of a reaction between two different time stamps
Initial rate of reaction
The rate of a reaction at the start of the reaction when the slope is almost straight.
Order of reaction
The number that defines the impact the concentration of a compound has on the rate of reaction.
Flooding
Filling a reaction with an excess of one reactant, so that the other substance reacts to completion and can be analyzed. In this scenario, the concentration of the excess reactant does not decrease when measured using proper sigfigs.
Rate constant (k)
The constant used to connect the concentrations of substances to the rate of the reaction.
Half-life of first-order reaction
A constant reduction in concentration, found mathematically through the equation (ln2/k).
Half-life of second-order reaction
A reduction in concentration that takes longer at each interval, found mathematically through the equation 1/(k*[initial]).
Half-life of zero-order reaction
A reduction in concentration that speeds up at each interval, found mathematically using the equation [initial]/2k
k obs
k (rate constant) multiplied by the concentration of the flooded compound to its specified power.
First-order reaction integrated rate law
ln[A]t=-kt+ln[A]0
Second-order reaction integrated rate law
(1/[A]t)=kt+1/[A]0
Zero-order reaction integrated rate law
[A]t=-kt+[A]0
Reason temperature increases rate of reaction
Rising temperature means more energy in each molecule, creating more collisions.
Reason concentration increases rate of reaction
More of a compound means more compound to react.
Reason surface area increases rate of reaction
More surface area means more space for collisions
Reason higher energy phase increases rate of reaction
More energy in a phase means more movement and more likely to collide.
Reason catalyst increases rate of reaction
Catalyst separates one difficult reaction into multiple easy reactions, making products occur sooner.
Unimolecular reaction
One molecule becoming a different species
Bimolecular reaction
Two molecules colliding in a reaction.
Termolecular reaction
Three molecules colliding in a reaction. This is much less common than two molecules colliding.
Transition state
The point at the apex of a reaction in which the reaction holds the most energy.
Elementary step
An individual step in a larger reaction
Equilibrium
The final status of a reaction in a system, with proper amounts of both reactants and products. At this point, the rate of the forward reaction and reverse reaction are equal.
Le Chatelier’s Principle
The idea that, when an equilibrium is disturbed, a reaction will correct to produce either the proper proportion of reactants or products to restore equilibrium.
Kc
An equilibrium constant determined using the concentration of reactants/products. This primarily applies to a reaction that forms a solution.
Kp
An equilibrium constant determined using the partial pressures of reactants/products. This primarily applies to a reaction in a gas.
K less than 1
The equilibrium constant of a solution that is reactant favored.
K greater than 1
The equilibrium constant of a solution that is product favored.
Reaction quotient (Q)
The number that defines the number of reactants/products in a solution, used to determine the distance of a reaction from equilibrium.
Q<K
A numerical value that indicates the reaction has too many reactants and will proceed in the forward direction.
Q>K
A numerical value that indicates the reaction has too many products and will proceed in the reverse direction.
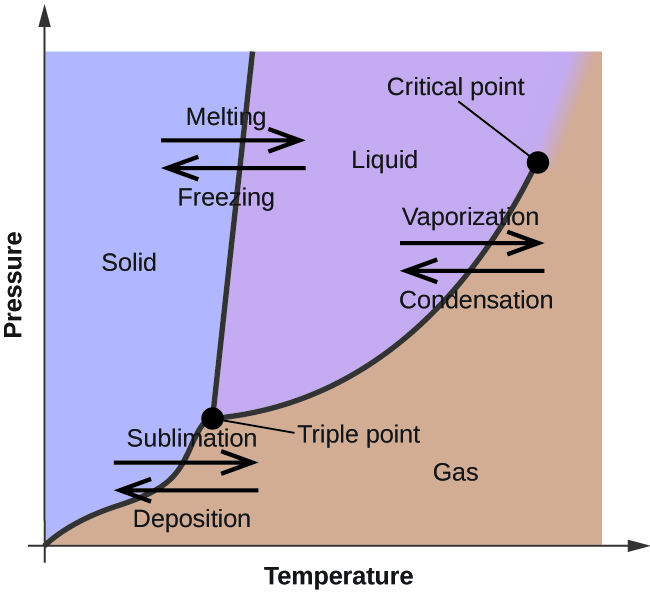
Phase diagram
A diagram that displays the relationship between temperature and pressure in a system, communicating the exact temperatures and pressures at which a compound will enter a certain phase.
Triple point
The point of pressure and temperature at which a substance will be found as a solid, a liquid, and a gas.
Bronsted-Lowry acids/bases
A definition of acids/bases in which an acid donates a proton, through an H+ ion, to a base.
Arrhenius acids/bases
A definition of acids/bases in which an acid dissolves to add H+ ions and a base dissolves to add OH- ions to a solution of water.
Lewis acids/bases
A definition of acids/bases in which an acid receives an electron pair and a base donates an electron pair.
Amphiprotic compound
A compound that can serve as both an acid and a base; it can both accept and donate a proton.
Strong acids
Acids that dissolve entirely in water, such as hydrochloric acid (HCl), hydrobromic acid (HBr), hydroiodic acid (HI), nitric acid (HNO3), perchloric acid (HClO4), and sulfuric acid (H2SO4).
Strong bases
Bases that dissolve entirely in water, such as lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), rubidium hydroxide (RbOH), cesium hydroxide (CsOH), calcium hydroxide (Ca(OH)2), strontium hydroxide (Sr(OH)2), and barium hydroxide (Ba(OH)2).
Weak acids
Acids that do not dissolve entirely in water. There are too many to list, but one example is acetic acid (CH3COOH).
Weak bases
Bases that do not dissolve entirely in water. There are too many to list, but one example is NH3.
Ka
The reaction constant of an acid. If this is greater than 1 the acid is strong, and if it is less than 1 the acid is weak.
Kb
The reaction constant of a base. If this is greater than 1 the base is strong, and if this is less than 1 the base is weak.
Like dissolves like
A compound that is polar will dissolve in polar liquids, and a compound that is nonpolar will dissolve in nonpolar liquids, but a compound that is polar will struggle to dissolve in nonpolar liquids and vice versa. This means more polar compounds are more soluble in water.
Kw
The autoionization constant of water, 10^-14.
Polyprotic acid/base
An acid/base that can react multiple times (i.e. the acid can receive multiple electron pairs).
Percent ionization
The numerical value of the amount of a compound ionized in solution. The equation to find this, for an acid, would be [H3O+]/[acid].
pH
The negative log of the concentration of hydronium ions in solution.
pOH
The negative log of the concentration of hydroxide ions in solution.
pH+pOH=14
The relationship between pH and pOH.