ACh and others
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
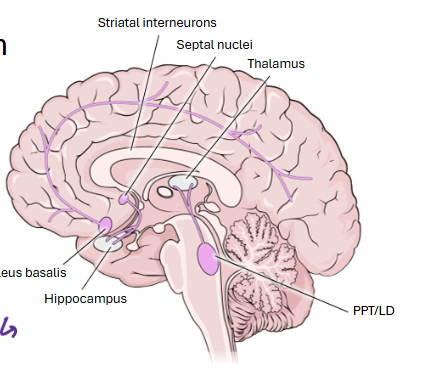
which groups of neurons do cholinergic pathways arise from?
Pedunculopontine (PPT)/ laterodorsaltegmentum (LD)
Magnocellular forebrain (Nucleusbasalis and septohippocampal))
Striatal interneurons
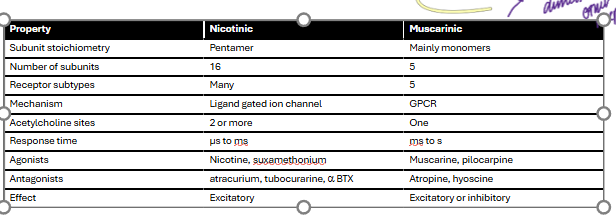
Name 4 differences between nicotinic (nAChR) and muscarinic (mAChR) acetylcholine receptors (2 marks).
differences
muscarinic can be either excitatory or inhibitory whereas nicotinic is excitatory only
nicotinic receptors are pentamers whereas muscarinic receptors are mainly monomers
nicotinic receptors are ligand gated ion channels whereas muscarinic receptor are GPCR
nicotinic receptors can bind 2 or more acetylcholine where muscarinic binds one only
nicotinic receptors have faster transmission than muscarinic
pednuculopontine and laterodorsaltegmentum nuclei
found in brainstem
send projections to thalamus
play important role in arousal and sleep wake regulation
Magnocellular neurons of the basal forebrain
project widely to the cerebral cortex, providing cholinergic input that supports attention, learning, and memory.
Septal nuclei
basal forebrain
send cholinergic projections to the hippocampus, contributing to memory formation and spatial navigation.
draw the distribution of the cholinergic neurons

roles of acetylcholine
Arousal, learning and memory
Pontine nuclei – arousal, sleep/wake
Magnocellular forebrain – arousal
Septohippocampal – learning (short term)
(striatal interneurons – motor control)
why are knockouts not useful for roles of ACh
the receptor subtypes can overlap so it is hard to isolate function/effect
inducible knockouts may provide more information
acetylcholine receptors structure and key information table

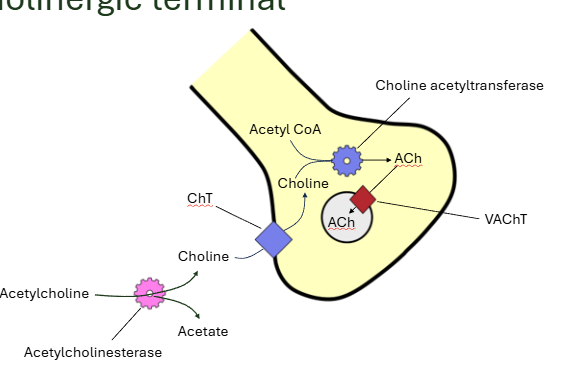
draw the cholinergic terminal

NMJ transmission of ACh (5 marks)
vesicle full of ACh fuses with the membrane and releases
ACh binds to nicotinic acetylcholine receptor on postsynaptic membrane (2 molecules each)
acetylcholine is broken down in the synapse by AChE into acetate and choline
choline is taken up by presynaptic terminal, joins with acetyl coa and forms acetylcholine
FAST SYNAPTIC TRANSMISSION
neuronal nicotinic transmission
dopamine is released from vesicles into synapse with calcium dependent manner
calcium voltage gated channels
nicotinic acetylcholine receptors which allow further calcium entry and enhance dopamine release - heteroreceptor
muscarinic cholinergic transmission
similar to periphery
acetylcholine binds to M2, M4, coupled to adenylate cyclase via Gi, inhibitory, often found on presynaptic nerve terminal
M1, M3 and M5 found on postsynaptic and foreign spaces, excitatory Gq coupled, inc calcium signalling
nicotinic receptor structure adult skeletal muscle
2 x alpha 1 subunits
beta 1
delta
epsilon
permeable to sodium, potassium and calcium
autonomic ganglia nicotinic receptor structure
alpha 3 ×2
beta 4 × 3
brain nicotine binding site
alpha 4 ×2
beta 2 ×3
brain bungarotoxin binding site
5 x alpha 7
nAChR gating
TM2 domains
in resting state leucine bends inwards, quite large, hydrophobic
activated state - rotation leads to leucine pointing away, serine pointing inwards, small, hydrophilic
where are M1 receptors located
glands
cerebral cortex
autonomic ganglia
M1 receptor agonist therapeutic targets
schizophrenia
alzheimers
M2 receptor locations
heart
cns
smooth muscle
M2 role and therapeutic antagonists
modulate heart rate
modulate smooth muscle
→ bradycardia, alzheimers, depression
M3 location
glands
smooth muscle - eye, bronchial tract, blood vessels
M3 role and therapeutic antagonists
smooth muscle contraction
exocrine secretion - saliva and tears
→ overactive bladder, asthma, IBS
M4 location
CNS
salivary glands
M currents
arises from PIP2 gated potassium channels
hyperpolarises, stabilises the RMP
which receptors activated shuts off M current
M1, M3 and M5 - Gq coupled
leads to depletion of PIP2
cells become more excitable
darifenacin
30 fold M3 selective
overactive bladder
where is histamine found
mast cells in the brain
magnocellular neurons in the posterior hypothalamus
histamine metabolism
histidine → histamine with histidine decarboxylase
N-methyltransferase
Monoamine oxidase
results in inactive metabolites
draw histaminergic synapse
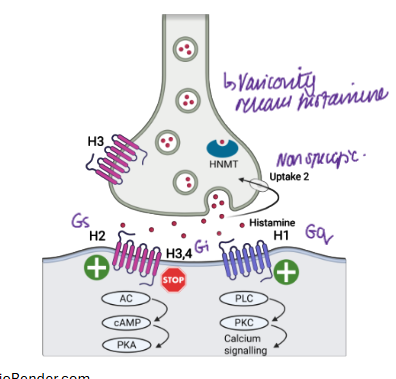

H1 antagonists
used widely therapeutically for allergies
some cross bbb to produce marked sedation
eg chlorophenamine and tripolidine
H2 receptors therapeutics
used for ulcer treatment
not much BBB penetration
neuropeptides
Must be synthesised in soma
produced as “pro” transmitter to be “finished” at the terminal
Stored in vesicles, Ca2+ dependent release
Postsynaptic action on GPCR
theory for neuropeptides
peptides eg VIP co exist with ACh in parasympathetic innervation of salivary glands
low firing rates - only ACh
high firing rates - both ACh and VIP
extension of dynamic range perhaps
examples of peptides
Cholecyctokinin (CCK8)
Corticotrophin releasing factor (CRF)
Galanin
Neurotensin
Enkephalins, endorphins, dynorphins(opioid peptides)
Substance P
Calcitonin gene-related peptide
Vasopressin
Oxytocin
Neuropeptide Y
Vasoactive intestinal polypeptide (VIP)
purines - adenosine
adenosine can act as a neurotransmitter
not vesicular - ATP dependent release
acts on GPCR A1, A2A, A2B and A3
form heteromers
protective
stabilisation
what do adenosine receptors form heteromers with?
family A and C GPCR
dopamine receptors
mGluR
caffeine
adenosine receptor antagonist
A2A highest affinity
melatonin
derived from tryptophan
made in the pineal gland from 5HT
driven by circadian light from retinal input
2 receptors MT1 and MT2 found in brain and retina
anti jet lag drugs
NO
produced by NOS, controlled by [Ca2+]i
inhibitory and excitatory
not synaptic or vesicular
sometimes found as co transmitter
role in LTP and LTD and neurotoxicity
lipids derived from arachidonic acid
prostaglandins
leukotrienes
endocannabinoids
where are cabbinoid receptors expressed
CB1 and CB2 throughout CNS and PNS
endocannabinoids - give 2 examples, mode of synthesis and termination
Two main ECs: anandamide, 2-arachidonoyl glycerol (2-AG)
Synthesised as needed from membrane lipids
Termination: Endocannabinoid Membrane Transporter (EMT)
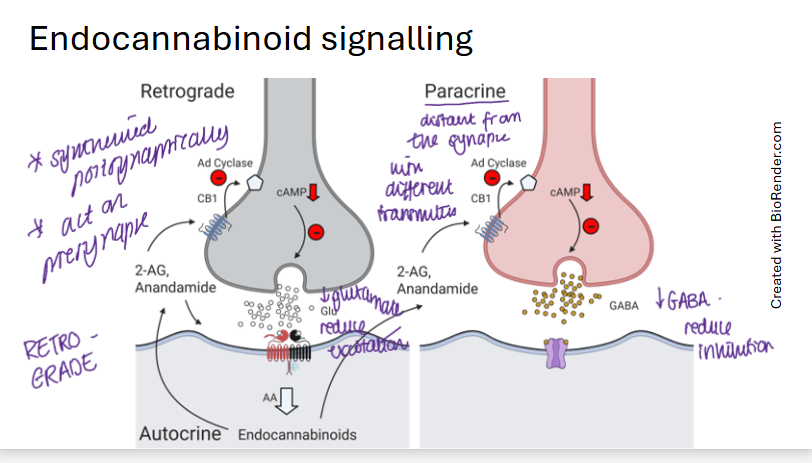
endocannabinoid signalling retrograde vs paracrine
retrograde is synthesised postsynaptically
paracrine is synthesised from another terminal
retrograde acts on presynapse whereas paracrine acts on different synapse with different transmitter
paracrine releases GABA, autocrine releases glutamate
draw a diagram of both retrograde and paracrine endocannabinoid signalling