Chapter 2: Chemical level
1/207
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
208 Terms
What are the two parts of a solution?
Solvent dissolves another substance called the solute
Hydrophilic
Solutes that have charges or polar covalent bonds, making it easier for water to tear them apart
Hydrophobic
Solutes that have nonpolar covalent bonds and therefore are not water soluble (eg lipids)
Hydrolysis
When water is a reactant in a decomposition reaction (breaks down a large molecule)
Dehydration synthesis reaction
When water is a product in a synthesis reaction
Heat capacity
Change in a substance's temperature when absorbing or releasing heat
Does water have high or low heat capacity?
High
Characterize water's heat of vaporization; give an example of how it's useful in the body
High (takes a lot of heat to make it become a gas); useful for cooling body by sweating
Water is an important component in this type of fluid, which helps reduce friction (eg mucus):
Lubricating fluid
Mixture
Combination of elements or particles blended physically but not chemically.
Colloid
When solute particles are large enough to scatter light. Look transparent or opaque (milk)
If material settles out at the bottom of the container, you have a:
Suspension
2 ways of measure concentration of a solution
Mass per volume percentage (ie isopropyl alcohol); Molarity (mol/L)
Define molarity:
Number of molecules in a given volume
Number of particles in a mole. Why?
6.02x10^23. This means that a mole of a substance has the same mass in g as the substance's atomic mass.
How do overly acidic substances damage the body?
H+ ions can change the structure of proteins
Molarity of H+ in substances with pH of 1, 7, and 14
1x10^-1 (0.1) MH+/L, 1x10^-7 MH+/L, 1x10^-14 MH+/L (just take pH number and make it negative, then put 10 to that power and add the molarity of H+ unit)
(remember higher on pH scale = less H+)
Dissociation
When inorganic acids, bases, or salts dissolve in water, separating into ions and getting surrounded by water.
Acid
Dissociates into one or more H+ ions and one or more anions (H+ Cl-)
Another name for acid. Why?
Proton donor. Creates an H+ ion that is just a single proton w/ no electrons.
Base
Removes H+ from a solution, aka proton acceptor.
Common ion that bases dissociate into
OH-
Salt
A substance that dissociates into cations and anions that aren't H+ or OH-, when dissolved in water.
When an acid and base react, the products are:
H2O and a salt (eg NaCl)
Role of salts in body
Source of electrolytes that help body carry electrical currents (think Gatorade/NaCl)
pH scale
Alkalinity or acidity of a solution measured from 0 (very acidic) to 14 (very basic). Scale of 10. pH 7 is neutral.
Buffer systems
Change strong acids and bases into weak ones
Carbonic acid-bicarbonate buffer system
Carbonic acid (H2CO3) donates H+ ions when needed and turns into bicarbonate (HCO3-), which can accept H+ ions if needed (weak acid/weak base)
Describe acid/base balance in ICF and ECF
Both have almost balanced quantities of acids and bases
Macromolecules
Very large molecules
Polymers
A large molecule created by covalent bonds between identical/similar building blocks (monomers) (remember ionic bonds don't scale as well)
Type of reaction that usually joins two monomers
Dehydration synthesis (forms H2O from H+ and hydroxl group)
Isomer
Molecule with the same molecular formula but a different shape/structure (such as glucose and fructose, C6H12O6)
Carbohydrates include what (4)
Sugars, glycogen, starches, and cellulose
Function of carbohydrates
Source of chemical energy for creating ATP → drives metabolic reactions
Explain the name of carbohydrates
Watered carbon -- they usually have about the same amount of C and H2O
Simple sugars can be which types of organic compounds? (2)
Monosaccharides or disaccharides
Monosaccharide
Monomer of carbohydrate; have 3-7 carbon atoms (for example glucose)
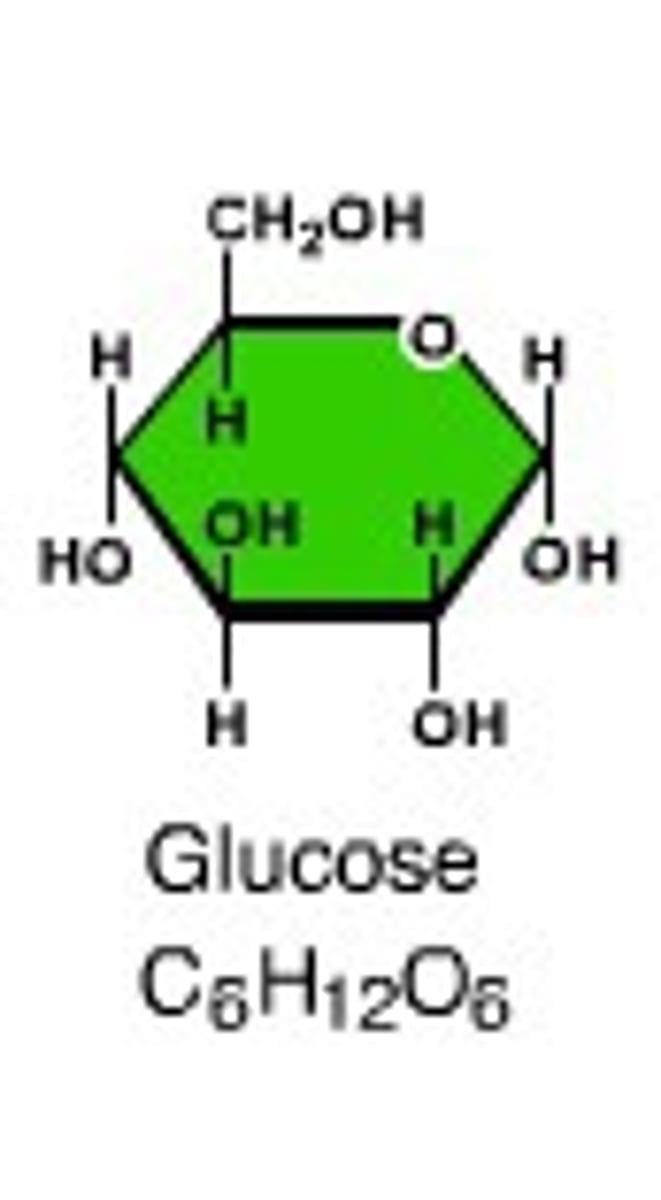
Disaccharide
Molecule created from the combination of two monosaccharides via dehydration synthesis eg glucose + fructose = sucrose
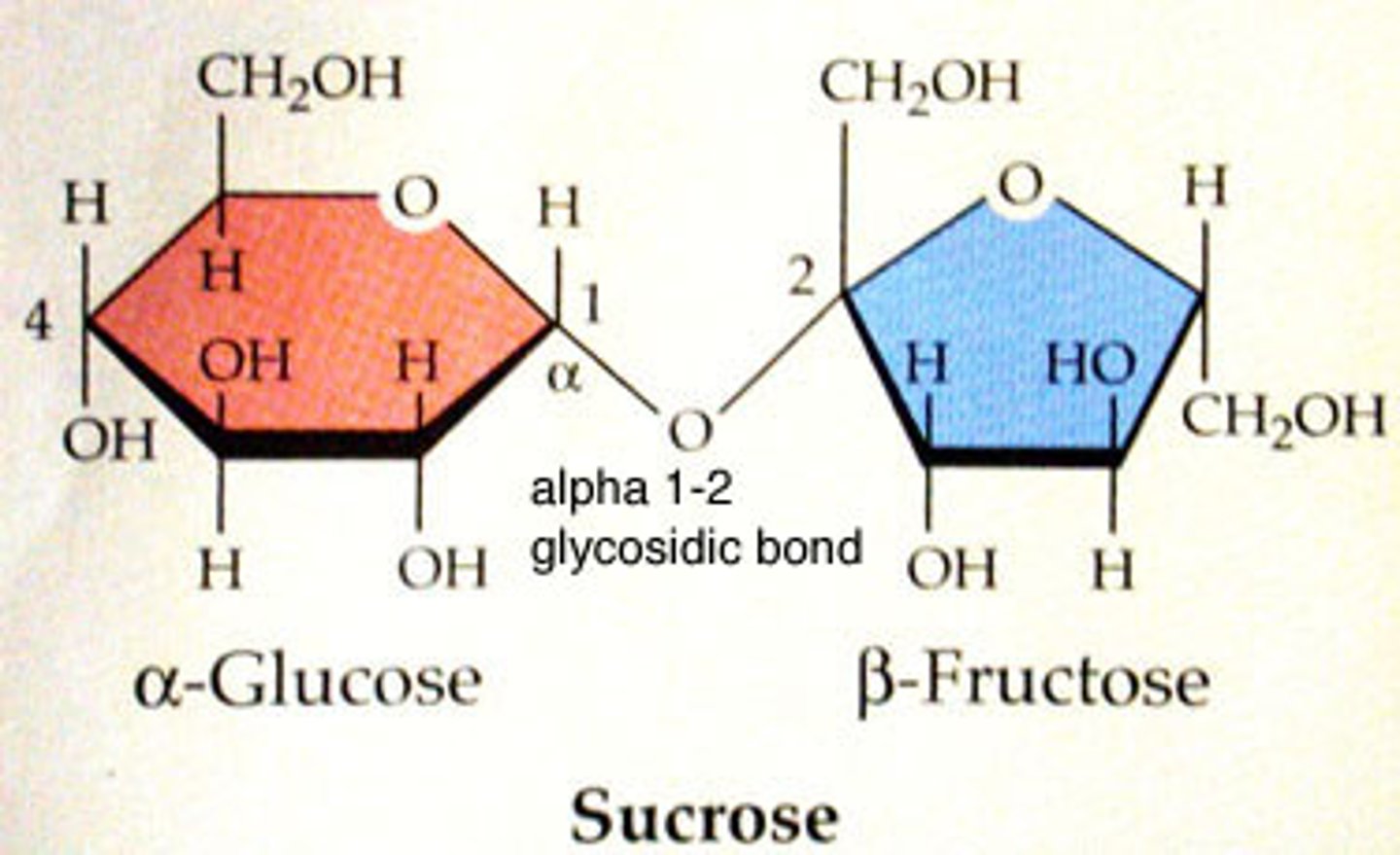
Polysaccharide (and three examples)
Tens or hundreds of monosaccharides joined by dehydration synthesis. AKA complex sugar; glycogen, starch, or cellulose (a type of dietary fibre)
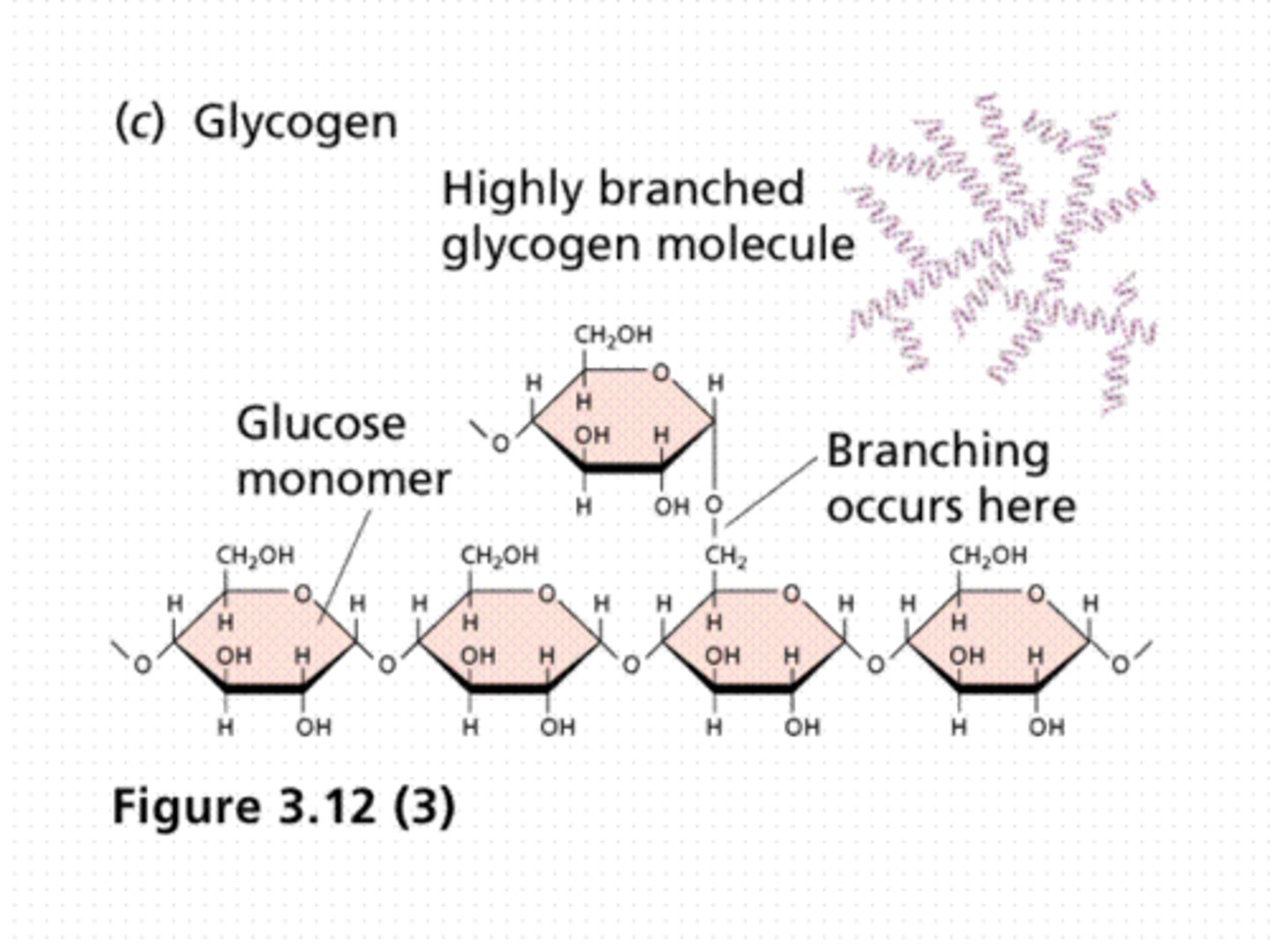
Main polysaccharide in body; it's makeup and role
Glycogen, made of branching glucose monomers; store glucose in liver/skeletal muscles
Body mass of carbs, lipids, and proteins
Carbs 2-3% Lipids 18-25% Protein 12-18%
Compare lipid composition to carbohydrate
Both contain carbon, hydrogen, and oxygen but carbs have a 2 H: 1 O ratio and lipids do not. Lipids have fewer electronegative O bonds; therefore hydrophobic)
Types of lipids in body
Triglycerides (fats and oils), phospholipids (cell membrane), lipoproteins (??), vitamins,
Triglycerides are also known as:
triacylglycerols
Lipoproteins -- what are they and describe their charge
Lipid molecules joined with hydrophilic protein molecules; overall hydrophilic (often occurs to allow lipids to dissolve in blood and be carried through bloodstream)
Examples of lipids (7)
Fatty acids (monomer), triglycerides (energy storage/insulation as fat or oil), phospholipids (bilayer membrane), steroids , eicosanoids (immune/regulatory), fat-soluble vitamins, lipoproteins
Makeup of fatty acid
Simple lipid; carboxyl group and hydrocarbon chain
Chemical role of fatty acid in body (what is it turned into?) (2)
Synthesizes triglycerides and phospholipids; or catabolized to generate adenosine triphosphate
Saturated fatty acid
Carbon atoms in hydrocarbon chain are joined together only with single bonds. This means they all have 2 hydrogen atoms each (and are therefore saturated)
Unsaturated fatty acid
Have a double covalent bond between two or more carbon atoms in hydrocarbon chain; this means the C atoms with double bonds aren't saturated by H (one each)
Describe the role of triglycerides in human body
Fat storage - they are the most highly concentrated form of chemical energy. Excess nutrients are stored as triglycerides in adipose tissue.
Building blocks of triglycerides (triacylglycerols)
Single glycerol molecule and 3 fatty acid molecules (one attaches to each C of glycerol)
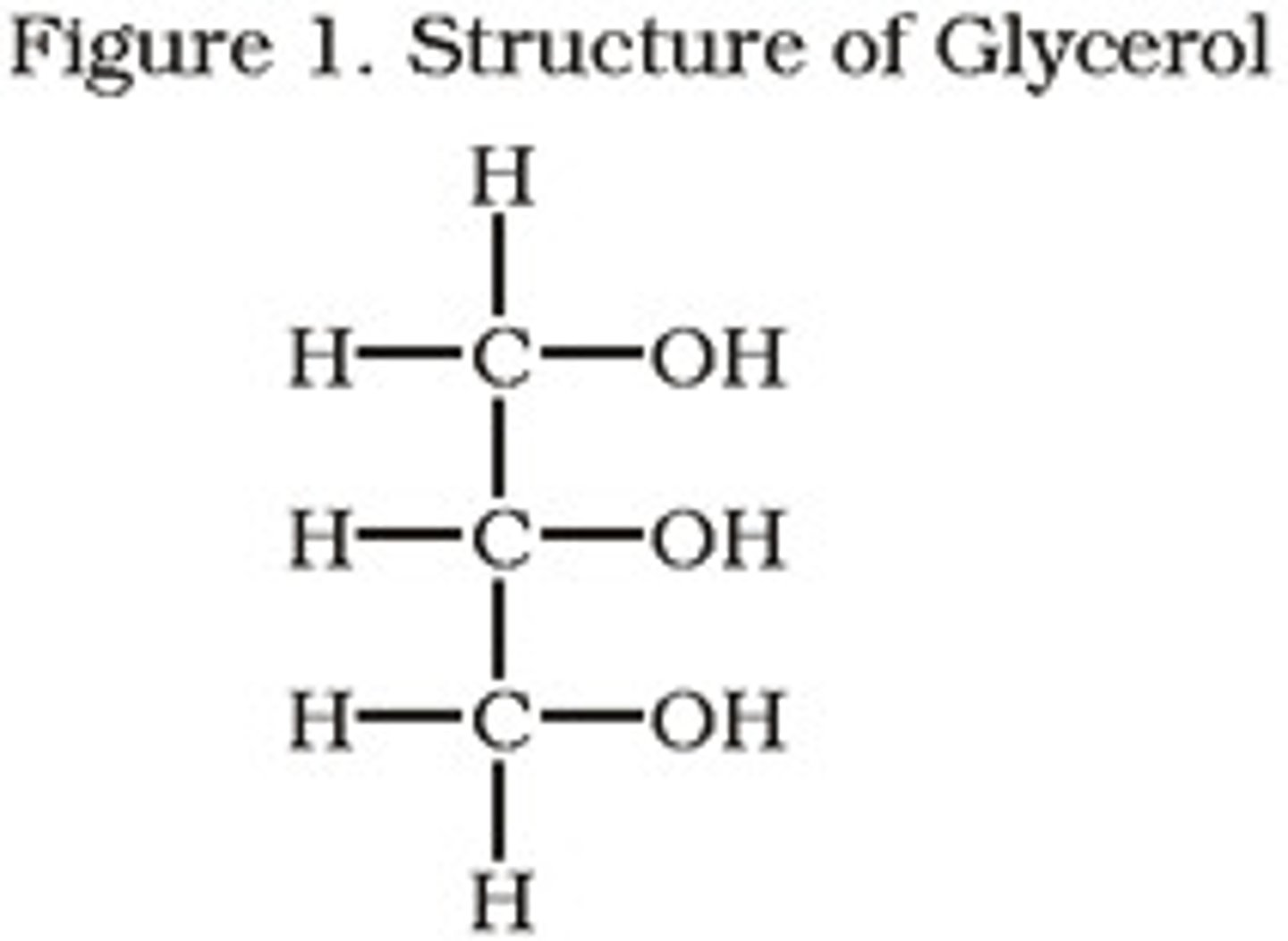
Glycerol
C3H8O3. Does a bunch of really cool shit, from being a sweetener to use in antifreeze. Mixes well with water. See also that song glycerine by Bush
Triglyceride that's solid at room temp
Fat :)
Saturated fat
Most fatty acids are saturated, letting the hydrocarbon chain pack together closely and solidify
Triglyceride that's liquid at room temp
Oil
Why is oil liquid at room temperature?
Unsaturated fatty acids create kinks at double bonds, preventing them from packing together closely
Monounsaturated fats
Triglycerides that have mostly one double covalent bond between C atoms in hydrocarbon chain
Polyunsaturated fats
Triglycerides that mostly have polyunsaturated fatty acids (multiple double covalent bonds between C atoms in hydrocarbon chain)
Phospholipid
Lipid with glycerol backbone and two fatty acid chains, but PO43- phosphate group is on the last carbon of glycerol (like a triglyceride with one fatty acid replaced by phosphate)
Describe chemical makeup of typical cell membrane
Phospholipid bilayer - Fatty acid part of the molecule is nonpolar/hydrophobic but head of molecule is charged and bond with H in water molecules (hydrophilic). They line up tail to tail (fatty acids inwards) to make up a cell membrane, with some cholesterol, glycolipids, and proteins thrown in.
Describe structure of a steroid
Four rings of carbon atoms
Examples of common steroids (7)
Cholesterol, testosterone, progesterone, estrogen, cortisol, bile salts, vitamin D
Role of cholesterol in body (2)
Precursor for other steroids; required for cell membrane structure
Structure of steroids
4 rings of carbon
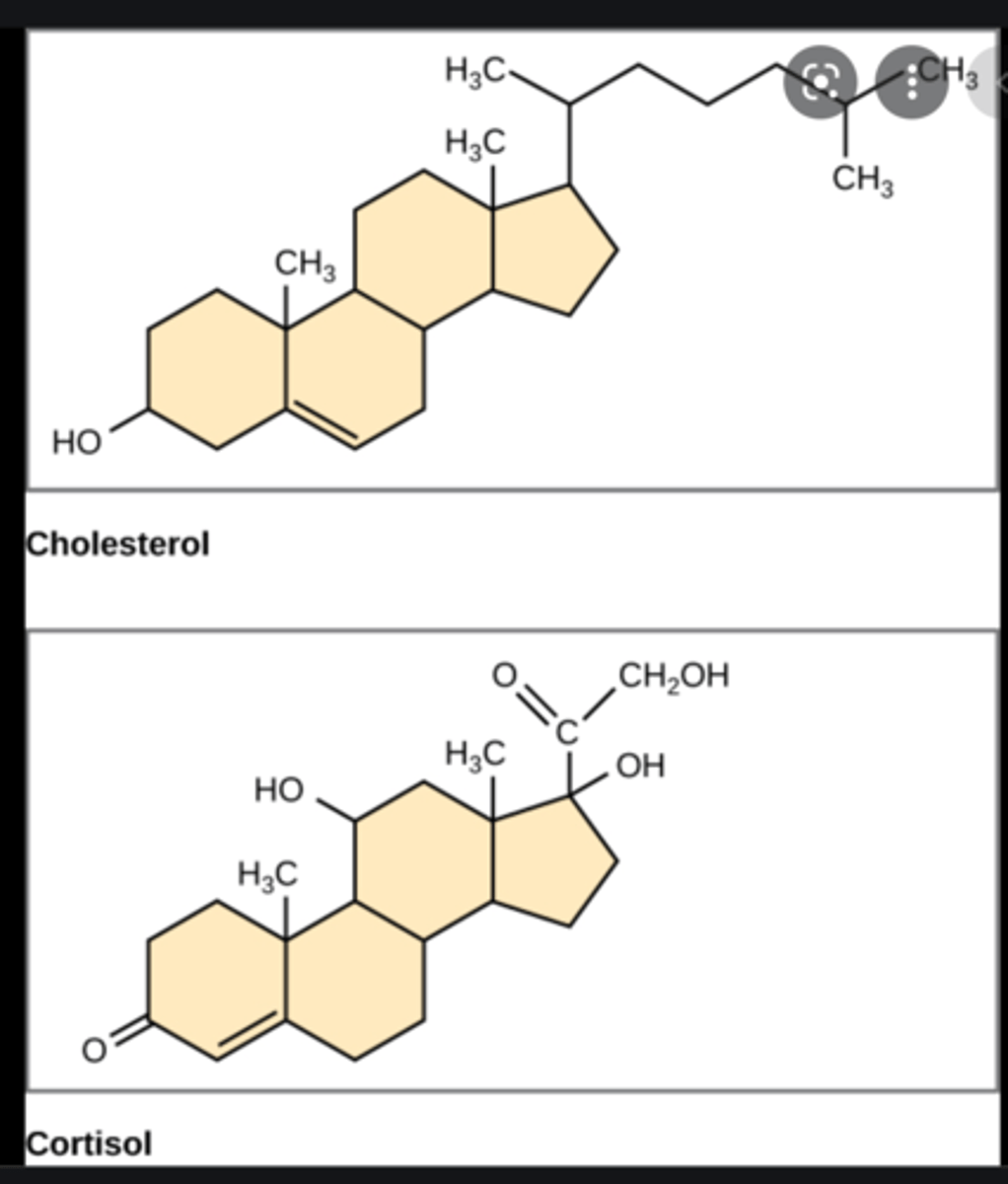
Steroids in body are synthesized from:
Cholesterol (which is a steroid itself)
Lipids derived from a 20-carbon fatty acid (arachidonic acid)
Eicosanoids
Prostaglandins
An eicosanoid that has a variety of regulating functions in the body (hormones, inflammation, airway dilation, ulcer prevention, etc)
Leukotrienes
A type of eicosanoid that helps with allergic and inflammatory responses.
Proteins contain which elements? (4.5)
C, H, O, N (sometimes S)
General functions of proteins in body (6)
Regulatory (hormones), structural (keratin and collagen), contractile (myosin/actin), immunological (antibodies), transport (hemoglobin), catalytic (enzymes)
Amino acids
Monomer of protein (one building block)
Chemical makeup of amino acid
Central carbon atom with three groups attached: amino (NH2), acidic carboxyl (COOH), and side chain R
Peptide bond (type of bond and which molecules it joins)
Covalent bond joining a pair of amino acids
Between which two molecules (and which atoms on these molecules) does a peptide bond form?
Between Carbon of carboxyl group (-COOH) and nitrogen of amino group (-NH2) of two amino acids
Dipeptide
Two amino acids combined
Adding one amino acid to a dipeptide results in a:
Tripeptide
How many amino acids in a peptide?
4-9 (note: more than a di or tripeptide!)
How many amino acids in a polypeptide
10-2000+ (proteins are complicated)
Why does denaturation occur?
Protein molecule loses its characteristic shape because homeostasis isn't maintained (different temperature or chemical composition of body fluids)
Nucleic acids - why are they nucleic
First discovered in nuclei of cells
What are two varieties of nucleic acids?
Deoxyribonucleic acid (DNA) and Ribonucleic acid (RNA)
DNA
Forms inherited genetic material in each cell
Gene
A segment of a DNA molecule
Role of ribonucleic acid (RNA)
Relays instructions from genes, telling cell how to synthesize proteins from amino acids
Monomer of nucleic acid
Nucleotide
Parts of a nucleotide
Nitrogenous base, pentose sugar, phosphate group
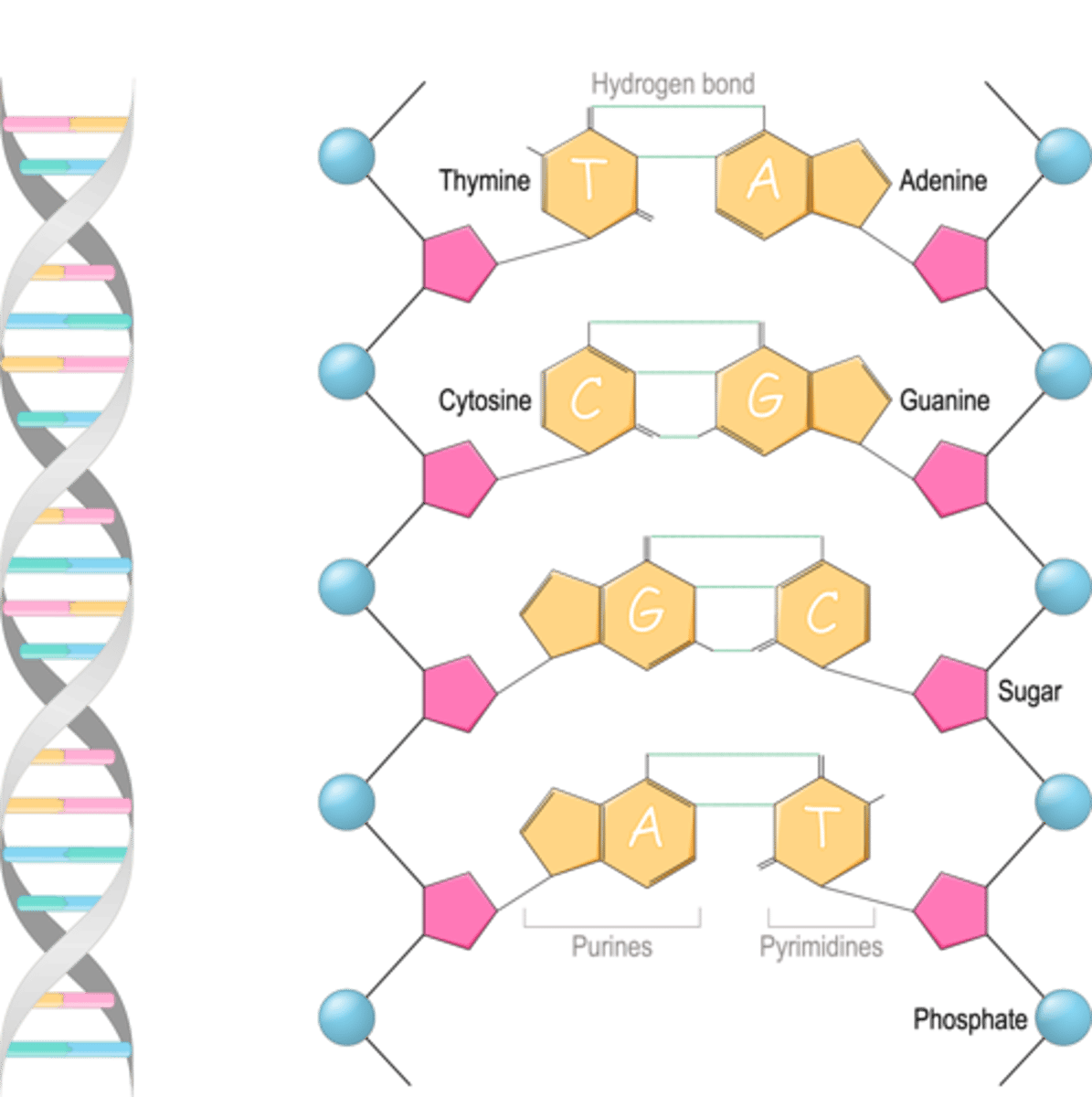
What are 4 nitrogenous bases of DNA?
Adenine (A), thymine (T), cytosine ©, and guanine (G)
What are purines? (2)
Adenine and guanine (larger, double-ring nitrogenous bases)
Pyrimidines (description and three examples)
Thymine, (uracil in RNA), cytosine (Smaller, single-ring nitrogenous bases)
What elements are in nitrogenous bases?
CHON (typical organic compound atoms)
Pentose sugar
A five-carbon sugar called deoxyribose (DNA) or ribose (RNA); attaches to a nitrogenous base and to a phosphate group to form a nucleotide, the monomer of a nucleic acid
Phosphate group
Alternates with pentose sugars to form backbone of DNA strand (bases project inwards from pentose sugars)
Describe double helix
A spiral ladder; shape of a DNA molecule. Alternating phosphate groups and deoxyribose sugars form uprights; pairs of bases form rungs.
How does RNA structure differ from DNA? (3)
Single strand with ribose sugar and uracil (U) instead of thymine base
Adenosine triphosphate
The "energy currency" of living systems. Transfers energy from exergonic catabolic reactions to power the cell's endergonic reactions.
Give 5 examples of ATP consuming activities
Muscle contractions, chromosome movement during cell division, moving structures within cells, transporting stuff across cell membranes (think carriers!), synthesis of large molecules (anabolic reactions)
Describe reaction that releases energy from ATP
Hydrolysis reaction; ATP + H2O -> ADP (one fewer phosphate) + PO4 + energy.
What catalyzes breakdown of ATP?
Enzyme called ATPase