Materials Chapter 3: Structure of Crystalline Solids
1/33
Earn XP
Description and Tags
Midterm 1
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
crystalline
periodic and repeating 3D array of atoms, ions, or molecules (metals, many ceramics, some polymers)
amorphous / noncrystalline
no periodic arrangement (complex structures)
ordered structures
tend to be nearer to the minimum in bonding energy, more stable
minimized
bonds tend to form between particles such that there is 0 net force exerted on them and overall energy is ________.
N = Ni + Nf/2 + Nc/8
number of atoms per cubic unit cell
1
Number of atoms per SC
4
Number of atoms per FCC
2
Number of atoms per BCC
6
Number of atoms per HCP
coordination number
number of nearest neighbor or touching atoms
6
coordination number for SC
12
coordination number for FCC
8
coordination number for BCC
12
coordination number for HCP
APF
the fraction of a unit cell occupied by hard-sphere atoms or ions, measure of the packing efficiency of a unit cell
APF = N*(V_atom)/V_cell
APF formula
6*R²*c*sqrt(3)
V_cell for HCP
hexagonal structure
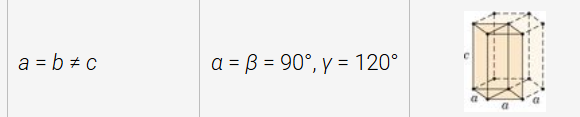
tetragonal structure
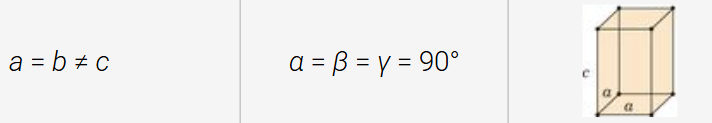
rhombohedral (trigonal) structure
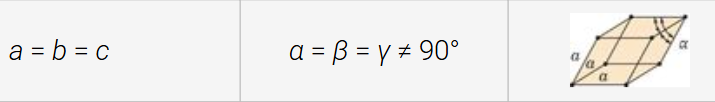
orthorhombic structure
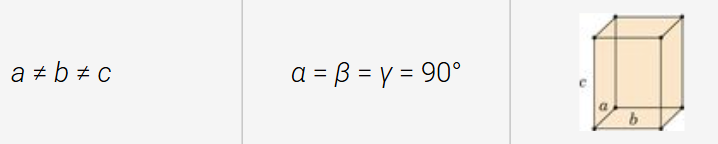
monoclinic structure
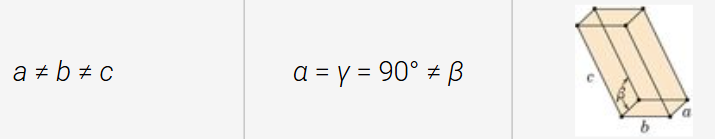
triclinic structure
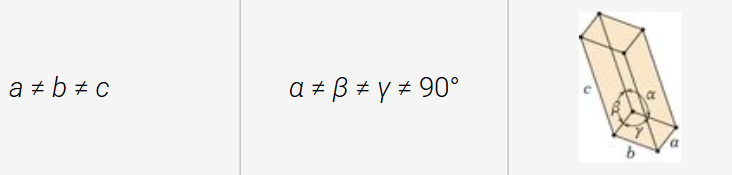
crystal system
A scheme by which crystal structures are classified according to unit cell geometry. This geometry is specified in terms of the relationships between edge lengths and interaxial angles
metals > ceramics > polymers
density comparison for 3 material types
single crystals
the periodic arrangement of atoms (crystal structure) extends without interruption throughout the entire specimen.
polycrystalline
consisting of many crystalline parts that are randomly oriented with respect to each other
Px Py Pz
notation for points
[n1 n2 n3]
notation for lattice vector (direction)
<h k l>
notation for set or family of directions
(h k l)
notation for crystal plane / Miller indices
{h k l}
notation for set or family of planes
(h k i l); i = - (h + k)
notation for hexagonal lattice
anisotropy
property value depends on crystallographic direction of measurement, observed in single crystals