alcohols and phenols
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
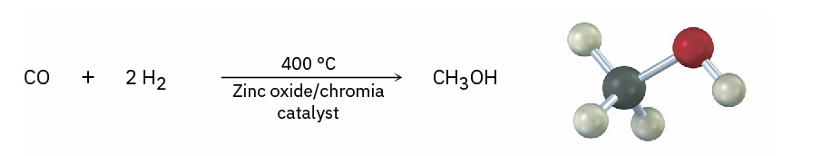
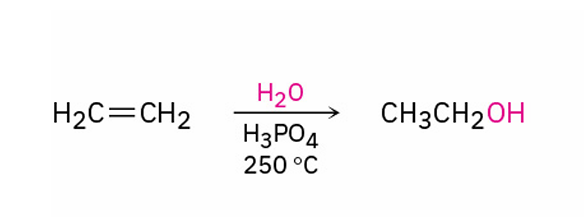
Methanol formation
Phenol
the word phenol is the name both of the specific compound (hydroxybenzene) and of the class of compounds.
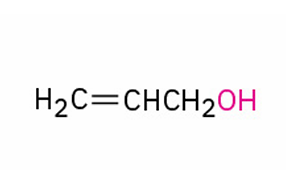
alternate IUPAC name?
Allyl alcohol
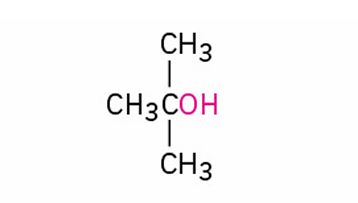
alternate IUPAC name?
tert-butyl alcohol
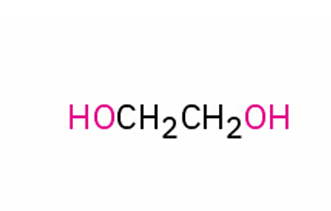
alternate IUPAC name?
ethylene glycol
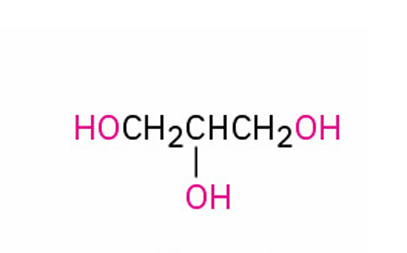
alternate IUPAC name?
glycerol
Ka (acidity constant formula)
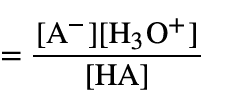
Ka and pKa relationship with acidity
bigger Ka and smaller pKa = more acidic and vice versa for bases
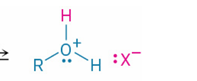
oxonium ion ( formed by weak base undergoing revers protonation ( accepting proton)
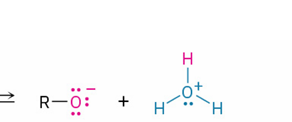
alkoxide ion (formed by weak acid donating a proton to H2O (also generates H3O+)
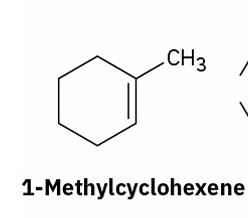
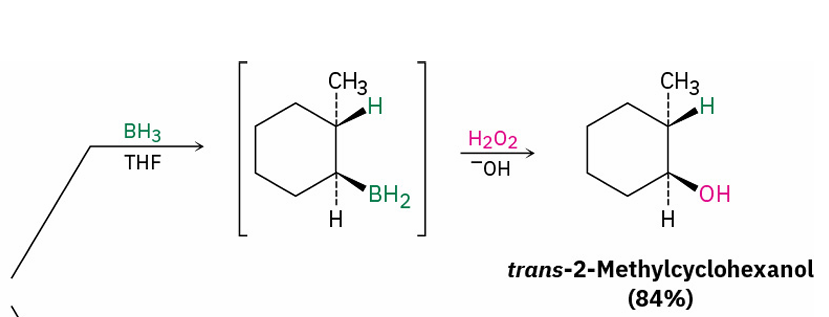
alkene hydration (Hydroboration–oxidation)
reagents
(syn, non-markovnikov)
BH3 and THF
H2O2 and OH
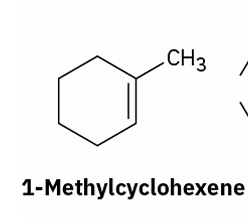
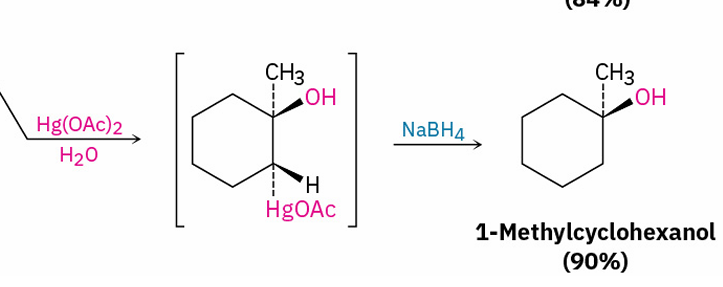
alkene hydration (oxymercuration– demercuration)
reagents ?
(markovnikov)
Hg(OAc)2, H2O
NaBH4
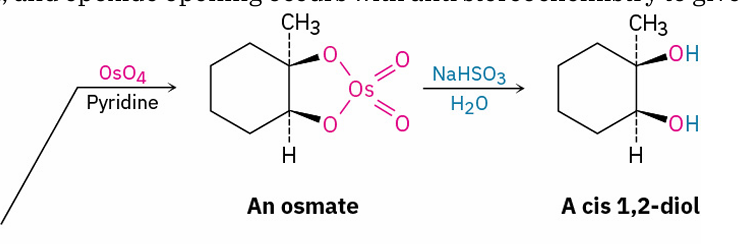
direct hydroxylation
reagents
OsO4 and pyridine
NaHSO3 and H2O
cis 1,2 diol
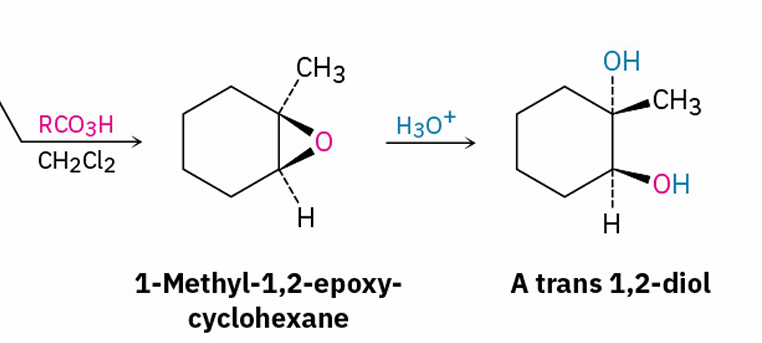
make a trans 1,2 diol
reagents?
RCO3H, CH2Cl2
H3O+
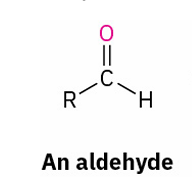
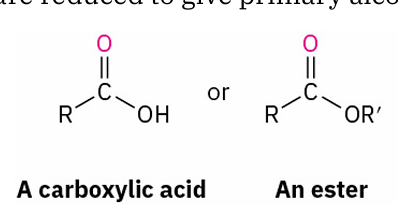
with reducing agent
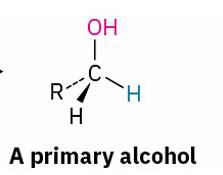
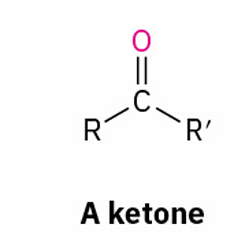
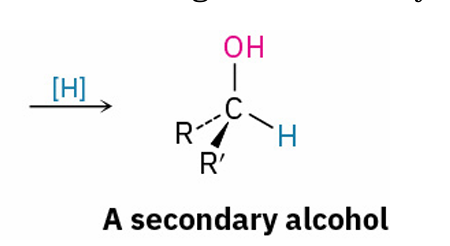
reduces to ?
Aldehyde and ketone reduction reagent
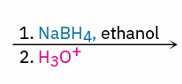
much more reactive and dangerous alternative reagent for aldehyde and ketone reduction
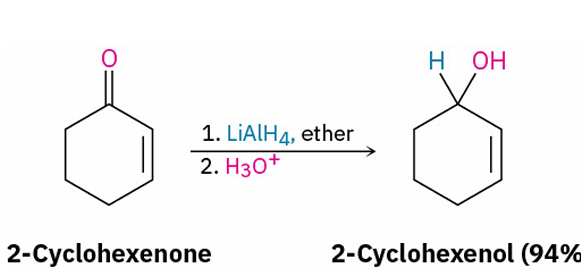
reduced to
what reduces all carbonyl groups
LiAlH4
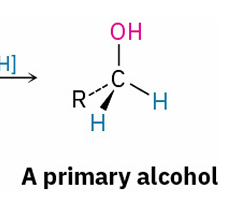
primary alcohol prepared by
reduction of an aldehyde, an ester , or a carboxylic acid
secondary alcohol prepared by
reduction of a ketone
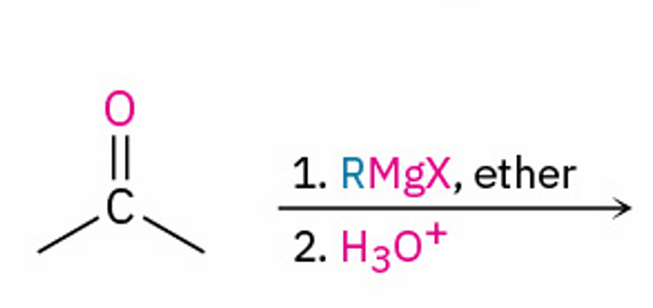
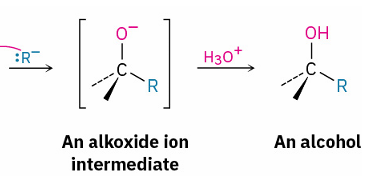
adds OH and R to carbonyl carbon
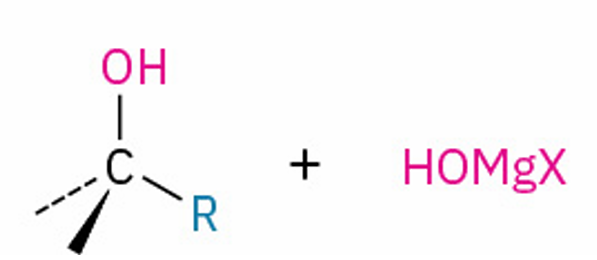
Grignard reagent
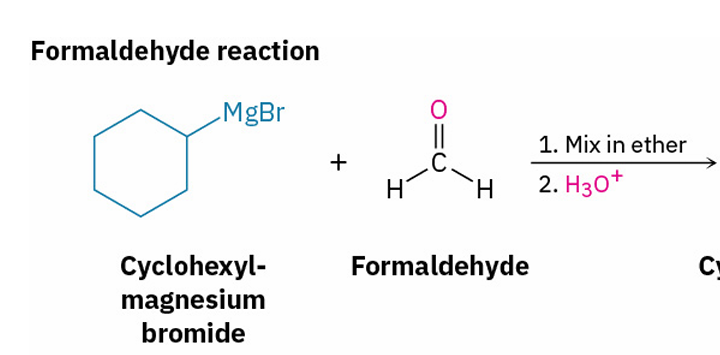
primary alcohol
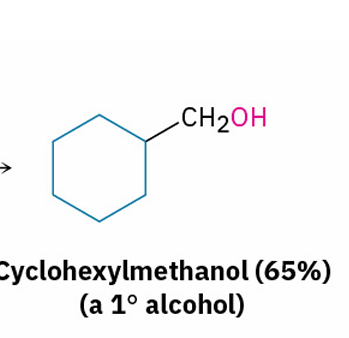
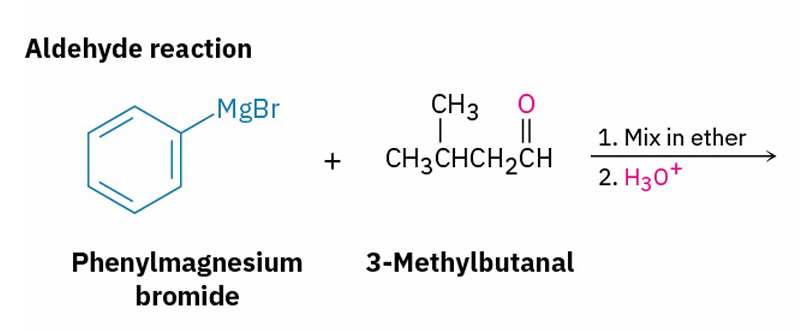
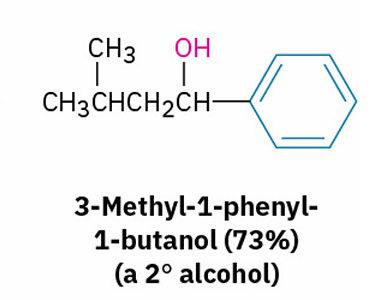
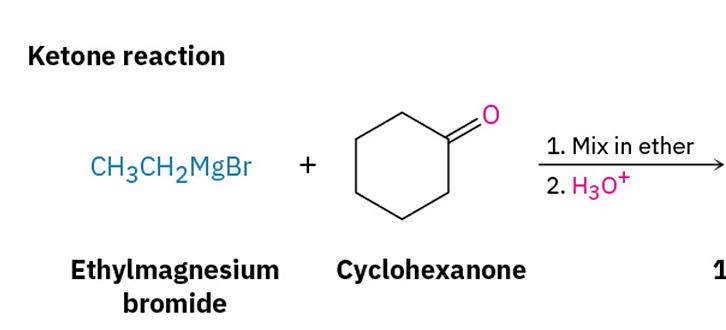
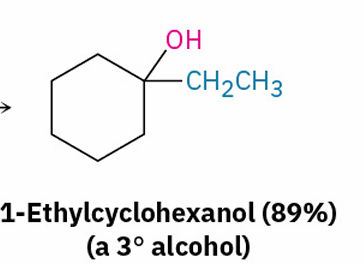
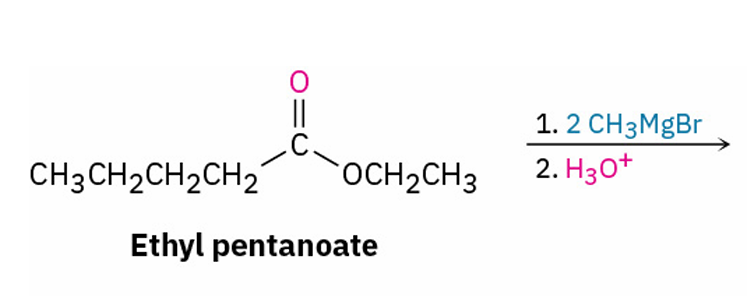
Grignard reagents with esters and their similarities to esters with LiAlH4
adds 2 R groups , just as LiAlH4 reduction of an ester adds two hydrogens
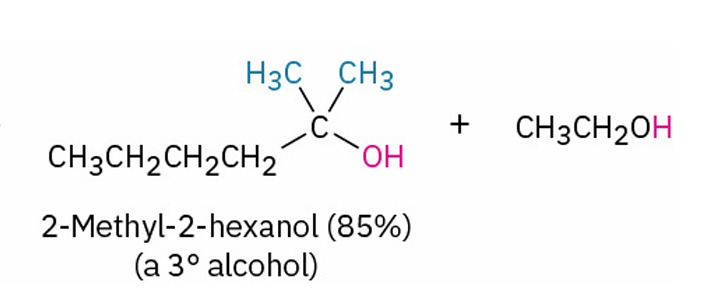
ester - MgX
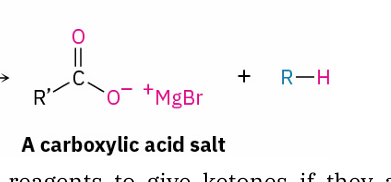
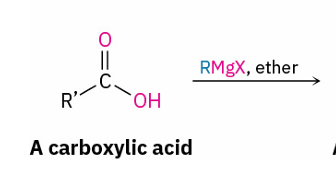
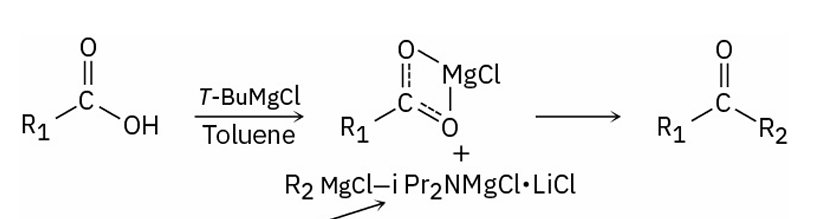
When do carboxylic acids react with Grignard reagents to give ketones
if first treated with i-Pr2NMgCl-LiCl (turbo-Hauser base)
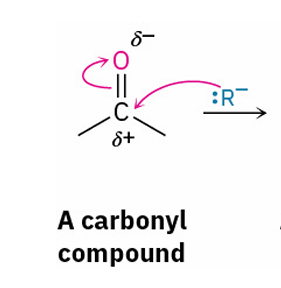
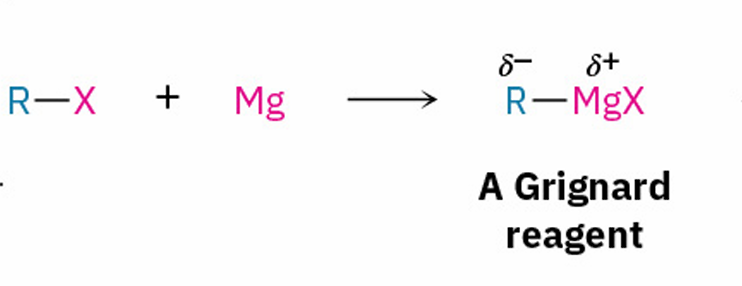
general Grignard reagent reaction
Grignard reagent + x = 1 alcohol
x = formaldehyde
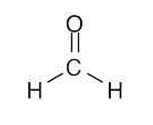
Grignard reagent + x = 2 alcohol
X= Aldehyde
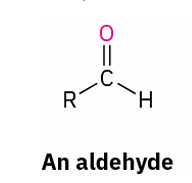
Grignard reagent + x = 3 alcohol
X= ketone
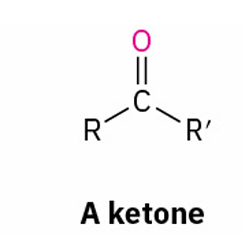
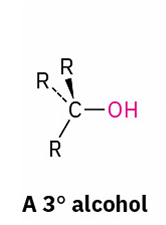
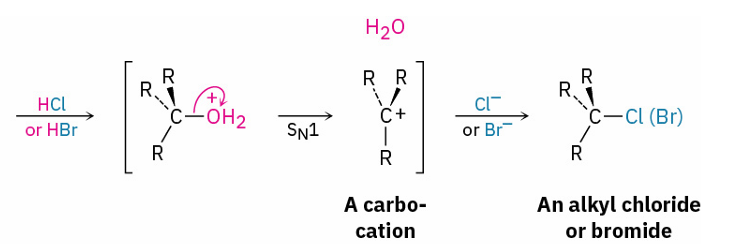
tertiary alcohols into Alkyl Halides
reagents
mechanism
HBr/ HCl
SN1
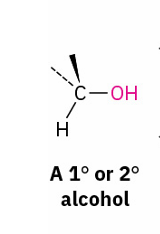
Primary and secondary alcohols to alkyl halide
mechanism
reagents
SN2
SOCl2 or PBr3
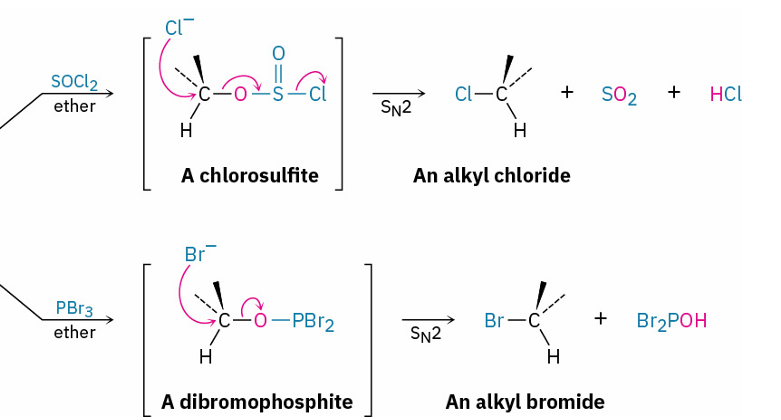
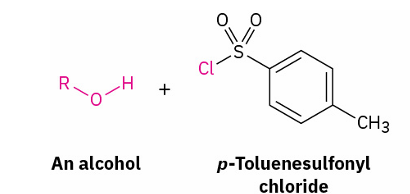
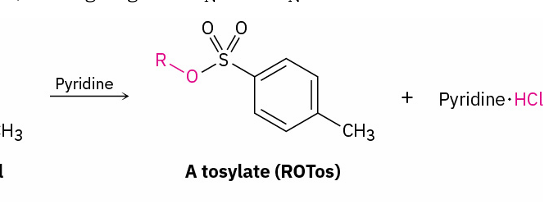
Alcohols into Tosylates
which bond is broken
reagent
O-H
p-TosCl
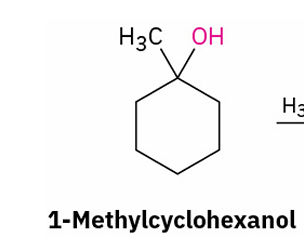
Tertiary alcohol to alkene (acid catalyzed reaction)
reagents
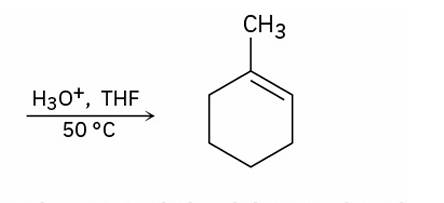
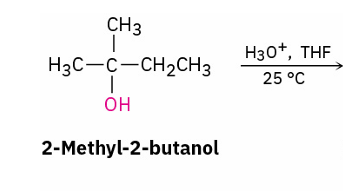
stereochem of acid catalyzed reactions
follow Zaitsev’s rule and yield the more stable alkene as the major product. (more substituted)
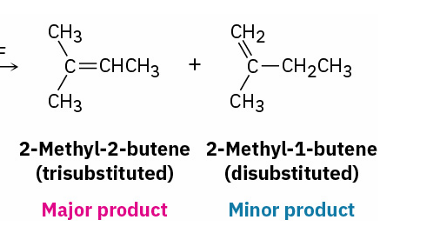
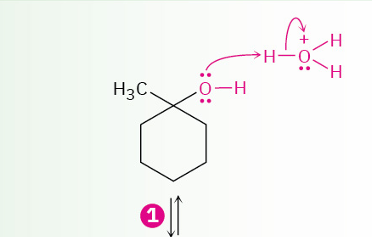
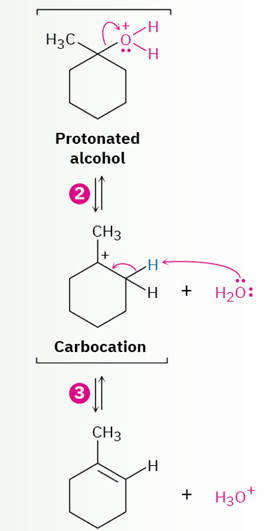
acid-catalyzed dehydration of tertiary alcohol to yield an alkene(mechanism)
intermediate( E1 mechanism)
milder way to dehydrate alcohol(turn to alkene)
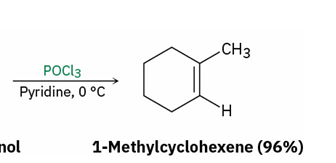
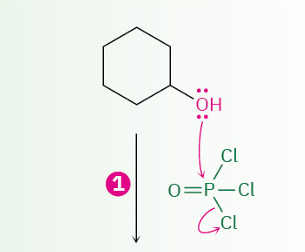
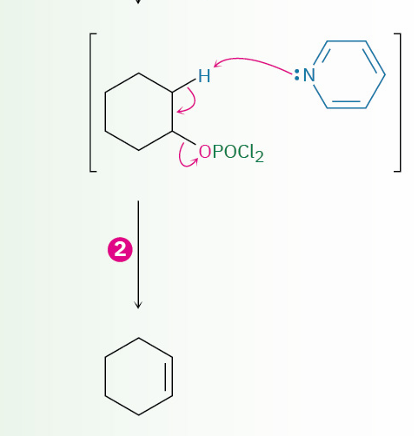
dehydration of secondary and tertiary alcohols by reaction with POCl3
OH converted to ?
mechanism
–OPOCl2
E2 ( follows E2 rules {leaving group have to be trans to each other (antiperiplanar })
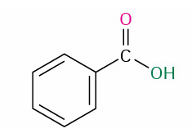
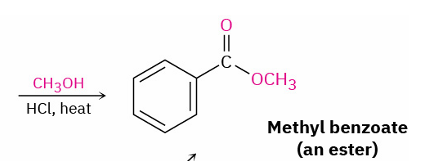
Direct carboxylic acid (alcohol) to ester
reagent ?
strong catalyst
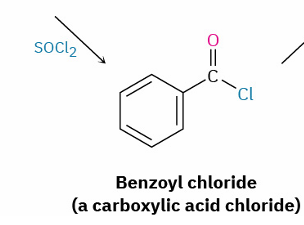
Indirect: carboxylic acid to ester by first converting carboxylic acid chloride
reagent ?
SOCl2
Ch3OH
oxidizes to
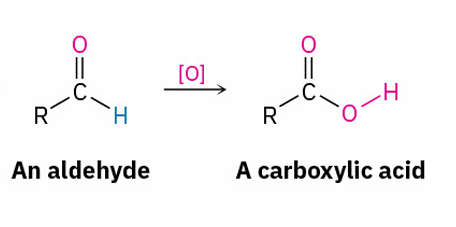

oxidizes to ?
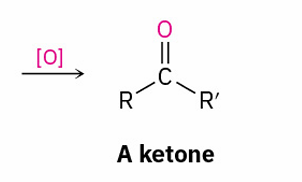

oxidizes to ?
no reaction
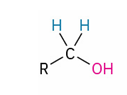
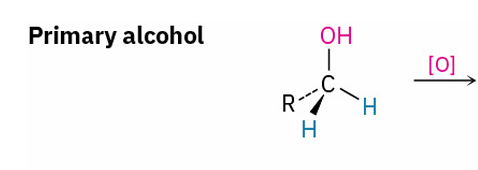
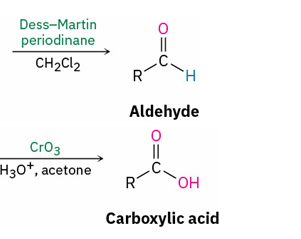
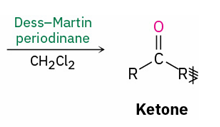
Reagents to oxidize primary alcohol to
aldehyde
carboxylic acid
Dess- Martin periodinane , CH2Cl2
CrO3 , H3O+ and acetone
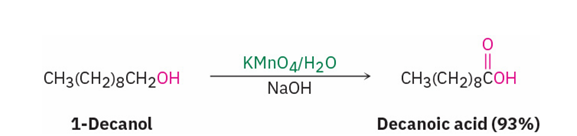
Primary alcohols are oxidized to carboxylic acids by …
KMnO4 in a basic aqueous solution
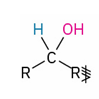
secondary alcohols oxidation to what?
reagents
Ketones
Dess-Martin periodinane
CH2Cl2
alcohol protection
stopping one functional group in a molecule form interfering with an intended reaction on another functional group elsewhere in the same molecule
alcohol protection method
chlorotrimethylsilane, and a base (CH3CH2)3N
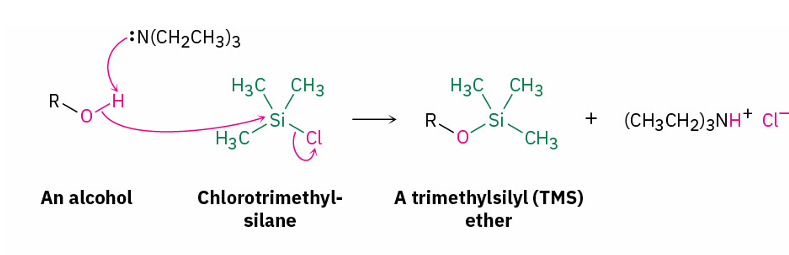
mechanism for alcohol protection with (CH3)3SiCl
SN2 (cl leaves and R-O binds at the same time )
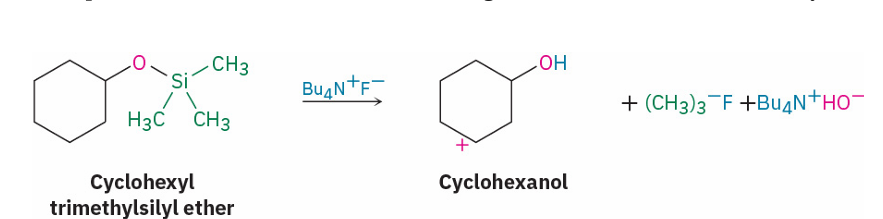
regenerate alcohol from trialkylsilylethers
reagent
Bu4NF
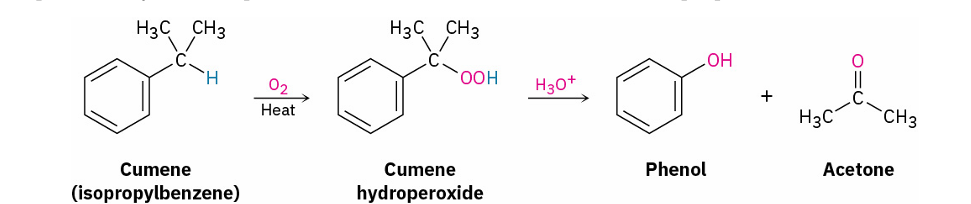
modern phenol formation from Isopropyl benzene (cumene)
O2/heat => peroxide
H3O+
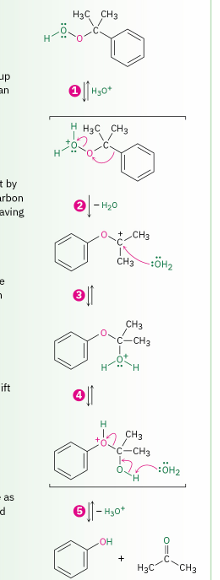
mechanism of phenol formation by acid catalyzed rearrangement of cumene hydroperoxide
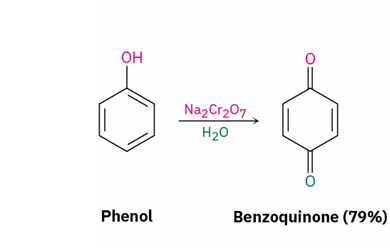
phenol oxidation
reagent
product
fremy’s salt (KSO3)2NO or Na2Cr2O7
quinone
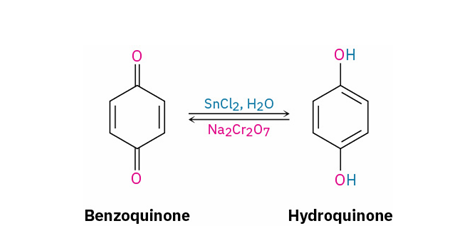
quinone to hydroquinone
reagent
SnCl2, H2O
NMR of phenols
the –OH signal of a tertiary alcohol (R3COH) is unsplit, the signal of a secondary alcohol (R2CHOH) is split into a doublet, and the signal of a primary alcohol (RCH2OH) is split into a triplet.
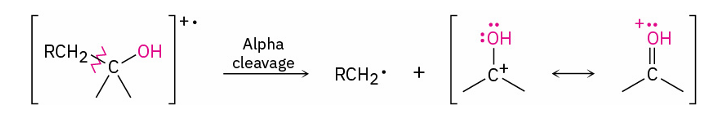
alpha cleavage
C–C bond nearest the hydroxyl group is broken