the nuclear atom
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
nucleus
positively charged, highly dense structure that contains virtually all the mass of the atom
protons + neutrons
relative mass (number of neutrons)
the difference between the atomic number and mass number is the number of neutrons in the element
nuclear symbol notation
A = mass number
Z = atomic number
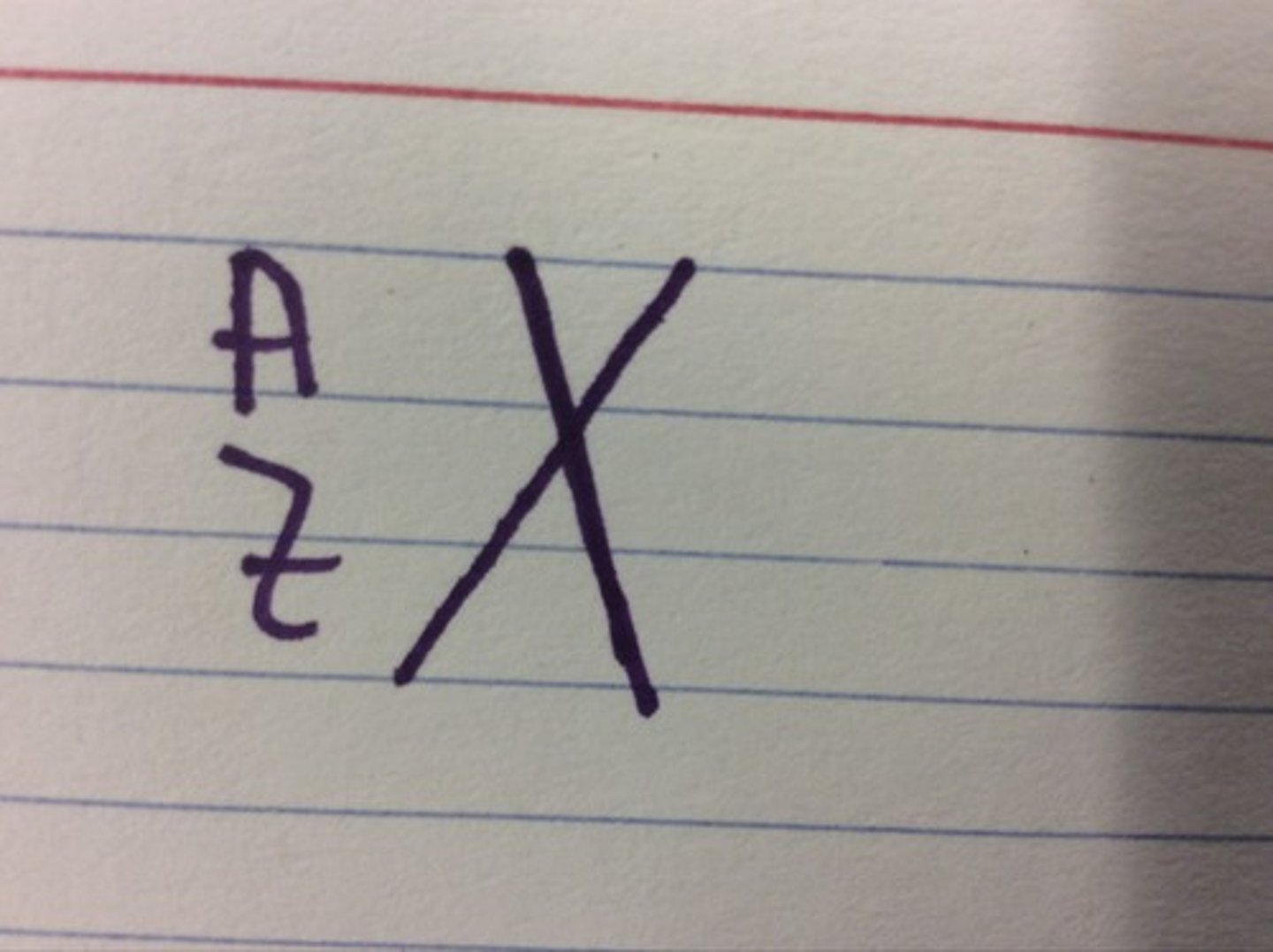
ionic bonds
opposite charges on two ions result in a force of attraction between the two
isotopes
different atoms of the same element with a different number of neutrons
- different mass numbers (A)
- same atomic number (Z)
properties of isotopes
similar chemical properties because they are the same element
different physical properties such as density
relative atomic mass (Ar)
average masses, taking into account the proportion of each isotope in a naturally occurring sample → weighed average mass
natural abundance (Na)
the percentage of its atoms among all atoms of the given element found on our planet
know how to calculate relative atomic mass and natural abundance
...
mass spectrometer
an instrument used to detect the relative abundance of isotopes in a sample
- results in cations (+)