Chemistry Test 3
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Energetics
Thermodynamics and Kinetics
Thermodynamics
The study of energy and its transformation
Are reactants or products energetically favored?
Gibbs Free Energy (Change of G)
Need enthalpy (H) and entropy (S) to answer thermodynamic questions
Interested only in Einitial and Efinal (starting point and ending point)
Can predict whether a reaction is favorable or not
Kinetics
Study of the process by which reactants form products
How fast will the reaction happen, and by what mechanism?
Interested in rates and mechanism (how fast and by which route)
1st Law of Thermodynamics
Energy cannot be created or destroyed, but energy can be converted from one form to another
(Change of energy)universe = 0
Universe = system + surroundings
System: what is being studied
Surroundings: everything else
(Change of energy)universe = (Change of energy)system + (Change of energy)surroundings = 0
(Change of energy)system = -(Change of energy)surroundings
If the system gains heat, the surroundings release heat
(Change of energy)surroundings = -(Change of energy)system
If the surroundings gain heat, the system releases heat
Enthalpy (H)
The heat content of the reaction when the pressure is constant
Exothermic: (Change of H) < 0
Endothermic: (Change of H) > 0
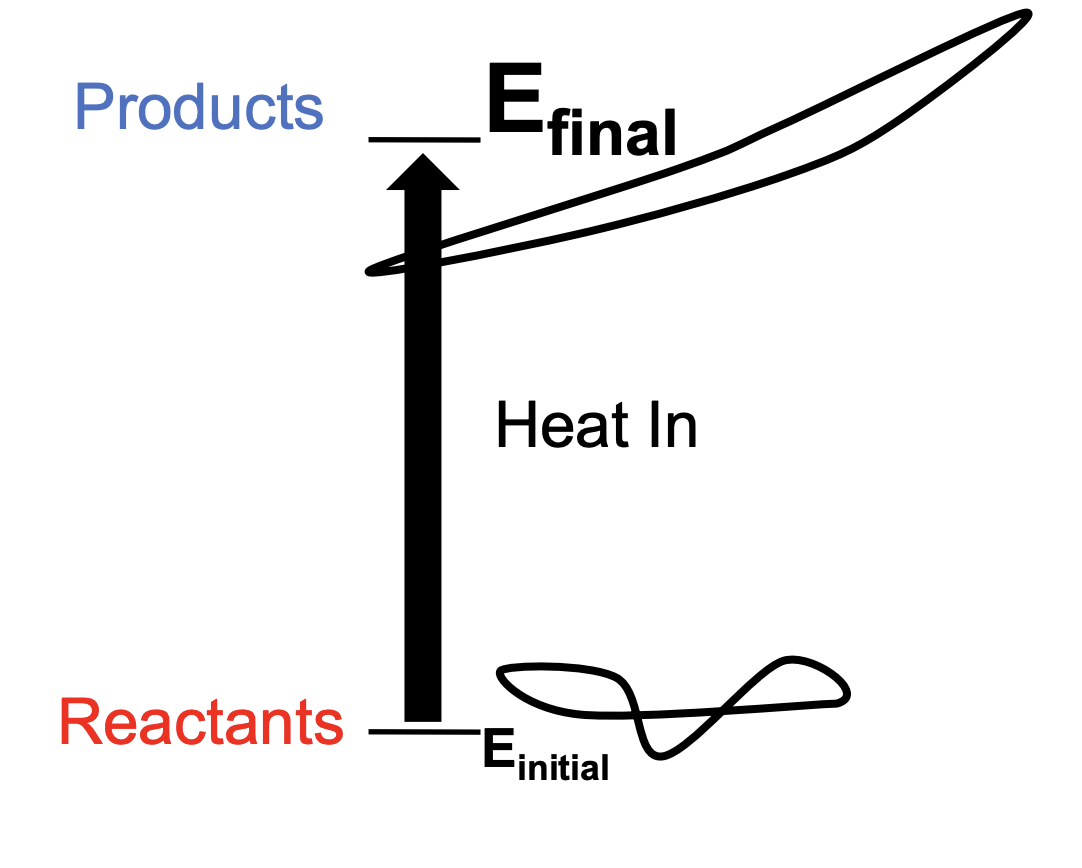
Endothermic reaction
Heat energy absorbed by the system, causing the surroundings to cool
(Change of H) > 0
Enthalpically unfavorable
low to high
bad for system
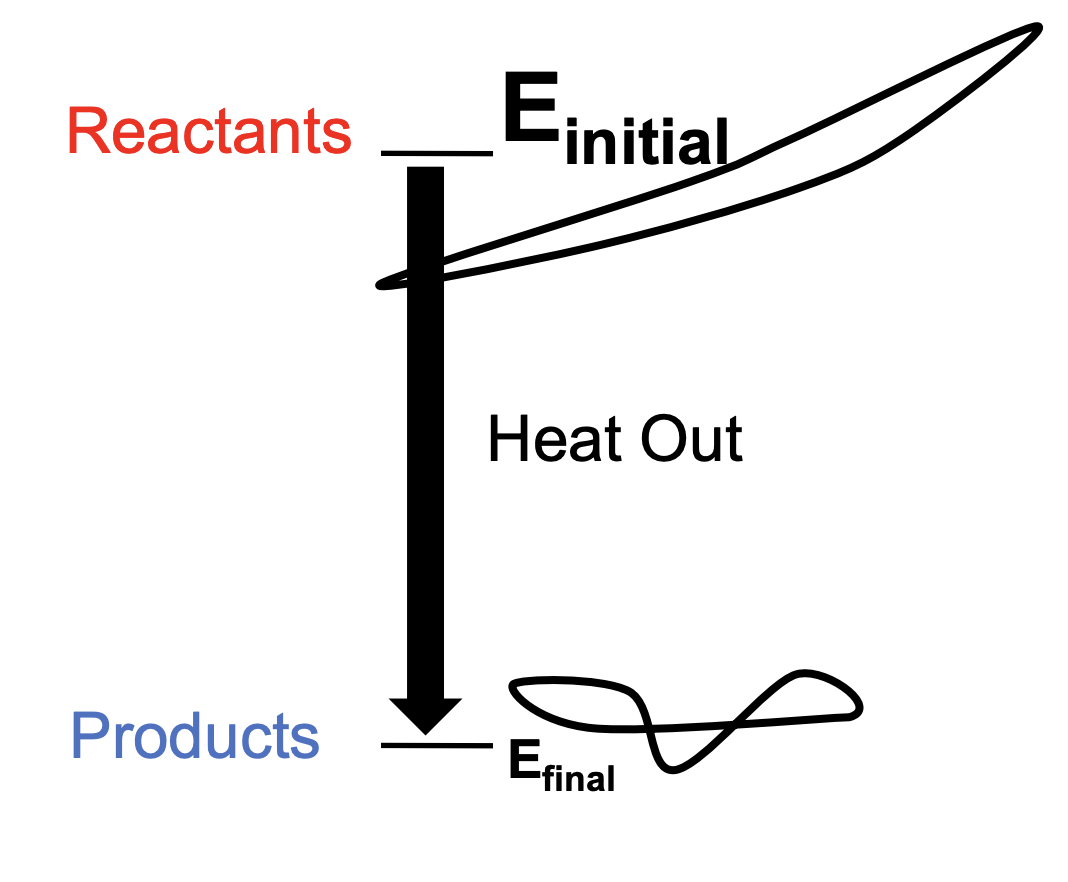
Exothermic reaction
Heat energy released by system (causing the system to warm)
(Change of H) < 0
Enthalpically favorable
High to low (Preferred by nature)
Good for system
When a species is higher in energy, that means its
more reactive
less stable
When a species is lower in energy, that means its
Less reactive
more stable
How to predict endothermic reactions
Reactants: Strong bonds — less reactive; more stable
Products: Weak bonds — more reactive; less stable
How to predict exothermic reactions
Reactants: Weak bonds — more reactive; less stable
Products: Strong bonds — less reactive; more stable
Bond Energy / Bond Dissociation Energy (BDE)
Energy input required to break 1 mole of a bond in the gas phase (fully separate the 2 bound atoms)
Used to find Change of H
Weak bonds – more reactive; require less energy to break
Strong bonds – more stable; require more energy to break

Entropy (S)
Related to the amount of disorder present in a system
Related to the number of ways energy can be distributed in a system
Low entropy system:
ordered system
energy is concentrated
High entropy system
Disordered system
energy can be spread out
(Change of S) > 0
increase disorder
entropically favorable
Solid → Liquid → Gas
Products have more gas molecules
(Change of S) < 0
Increase order
entropically unfavorable
Gas → Liquid → Solid
Products have fewer gas molecules
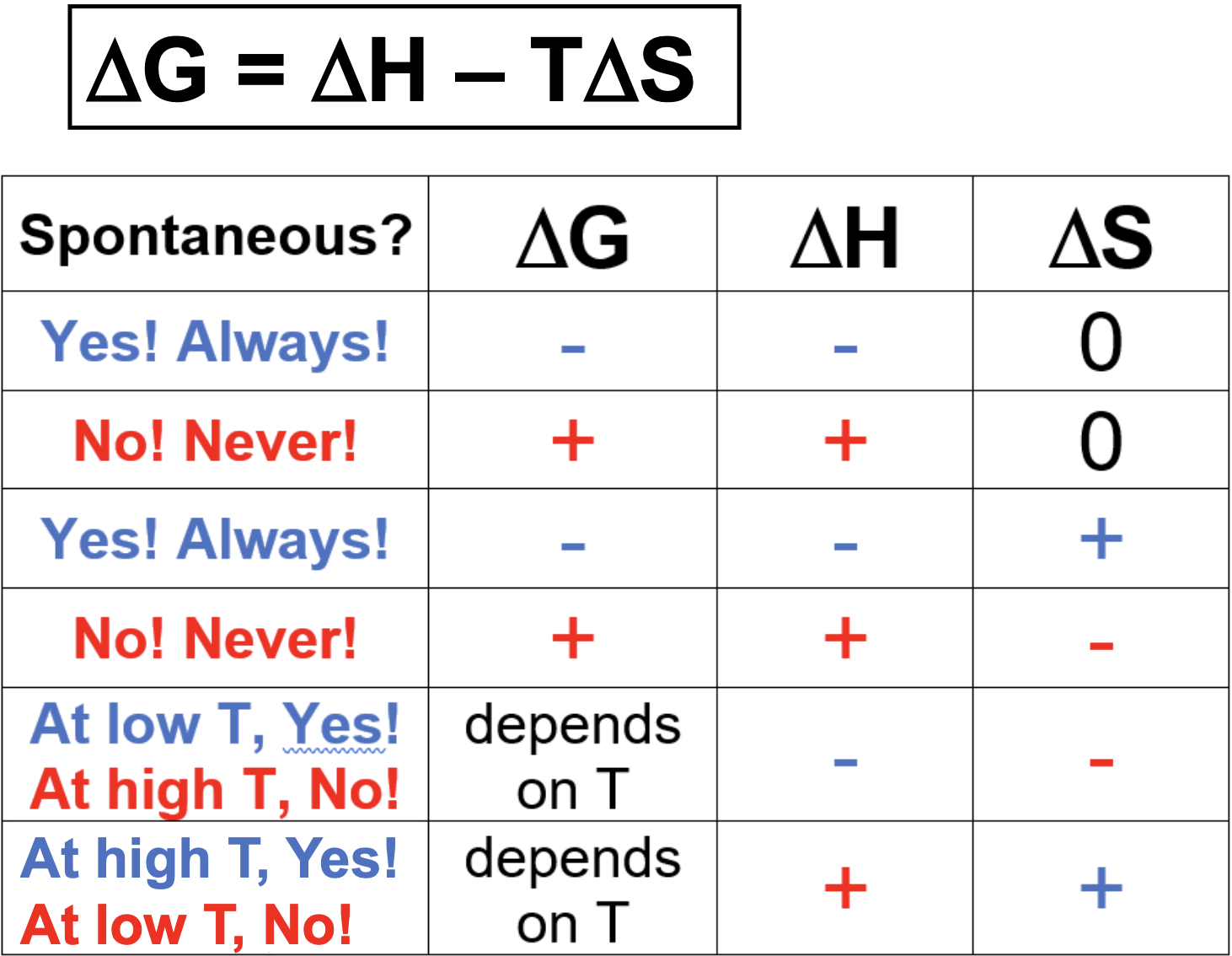
Gibbs Free Energy (G)
Equation:
T= Temp. (T in K)
(Change of G) < 0; reaction is spontaneous (products lower in energy than reactants; reaction proceeds)
(Change of G) > 0; reaction is non-spontaneous (Products higher in energy than reactants; reaction does not proceed)
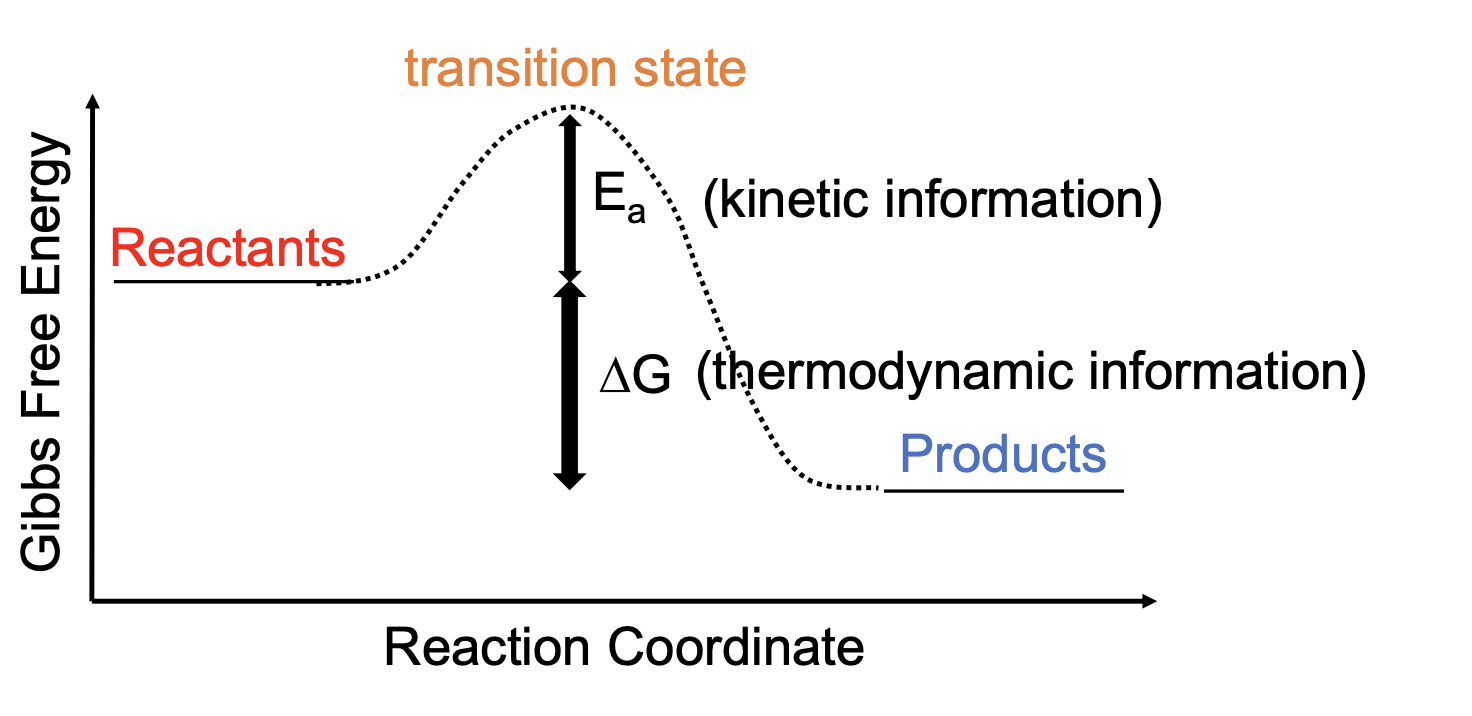
Gibbs Free Energy Reaction Coordinate
Ea reverse = Ea forward + Change of G
The larger the Ea, the slower the reaction
The smaller the Ea, the faster the reaction
How to determine transition state
All bonds that form
No bonds broken yet
Consider atom charges when forming bonds
How can we speed up a reaction?
1. Increase temperature
More frequent collisions
Gives collisions more energy to overcome Ea
2. Increase the concentration of reactants
More frequent collisions
3. Use a catalyst
Brings reactant molecules together to facilitate their collisions
Lowers the Ea
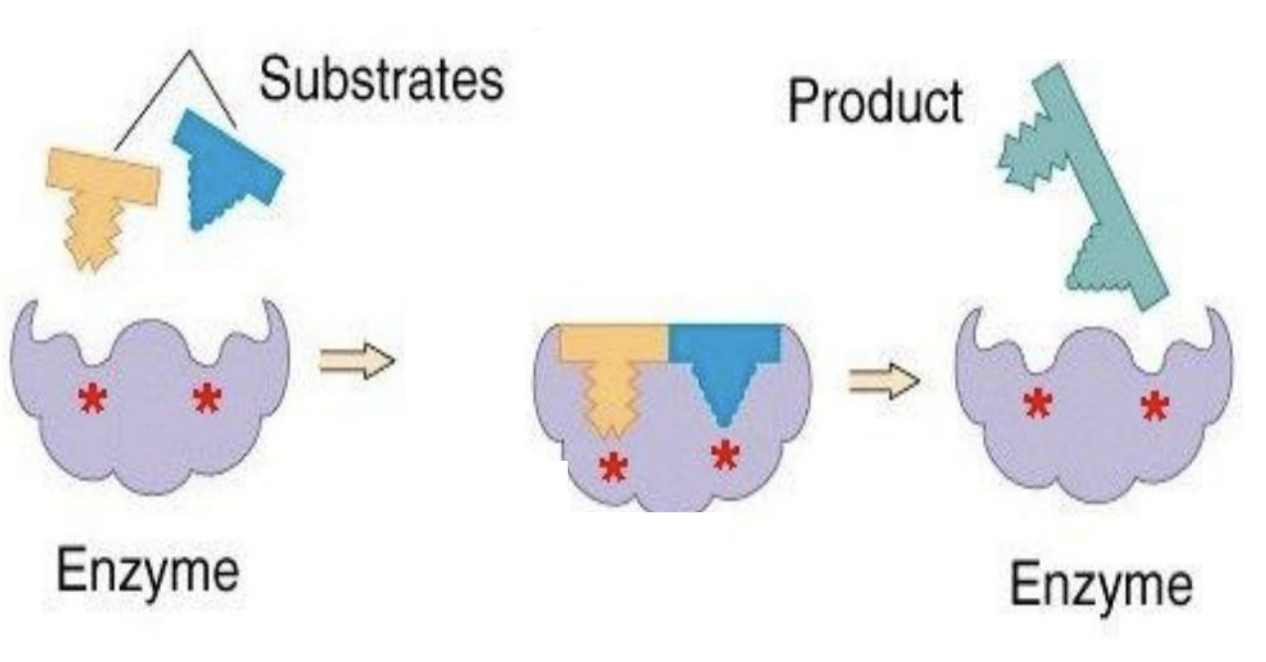
Enzyme catalysis
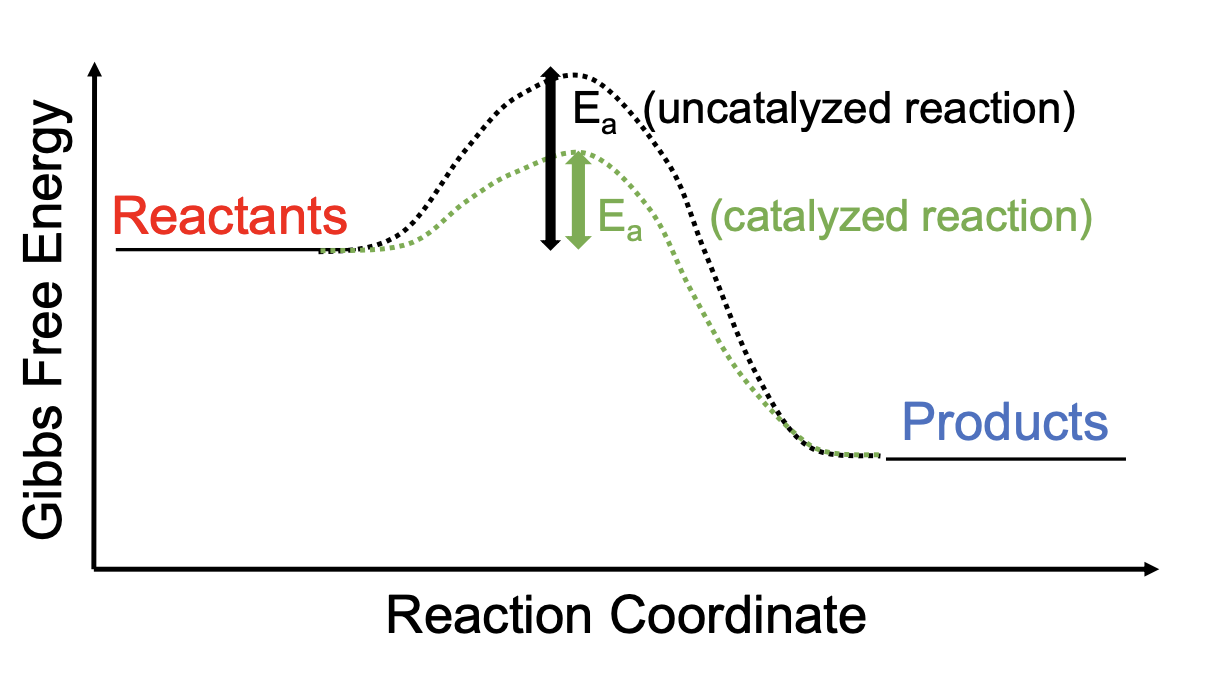
Reaction Coordinate diagram of an uncatalyzed reaction vs. a catalyzed reaction
Equilibrium
point at which forward rate = reverse rate and the concentrations of reactants and products remains constant
Equilibrium Expression
Reaction: aA + bB ←→ dD + eE
Lower-case letters are meant to represent coefficients
Upper-case letters are meant to represent molecules
Keq = [D]d [E] e / [A] a [B] b (Products/Reactants)
For solutions (aq), use molar concentration (mol/L)
For gases (g), use molar concentration or the partial pressure in atm
Solids (s) and liquids (l) are omitted from the equation
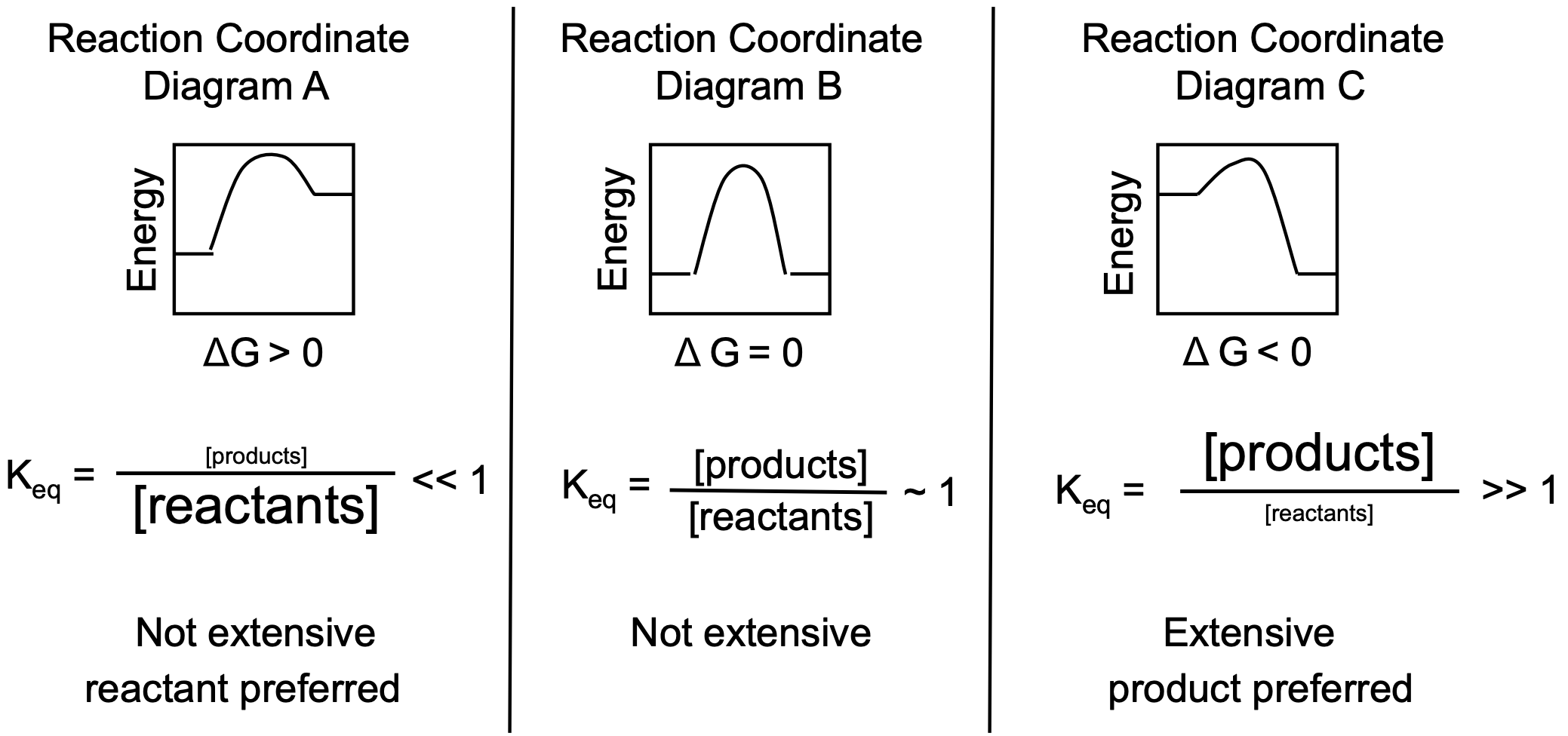
Keq ~ 1
There is a significant amount of both reactants and products present at equilibrium
Same amount of reactants and products being transferred
Keq >>1 (generally greater than 1.0x103)
A very large K means an “extensive” reaction, or the reaction goes “completely” to products
Large amount of product
Keq << 1 (generally smaller than 1.0x10-3)
A very small Keq means that products are not favored. The solution contains mainly reactants
A large amount of reactants
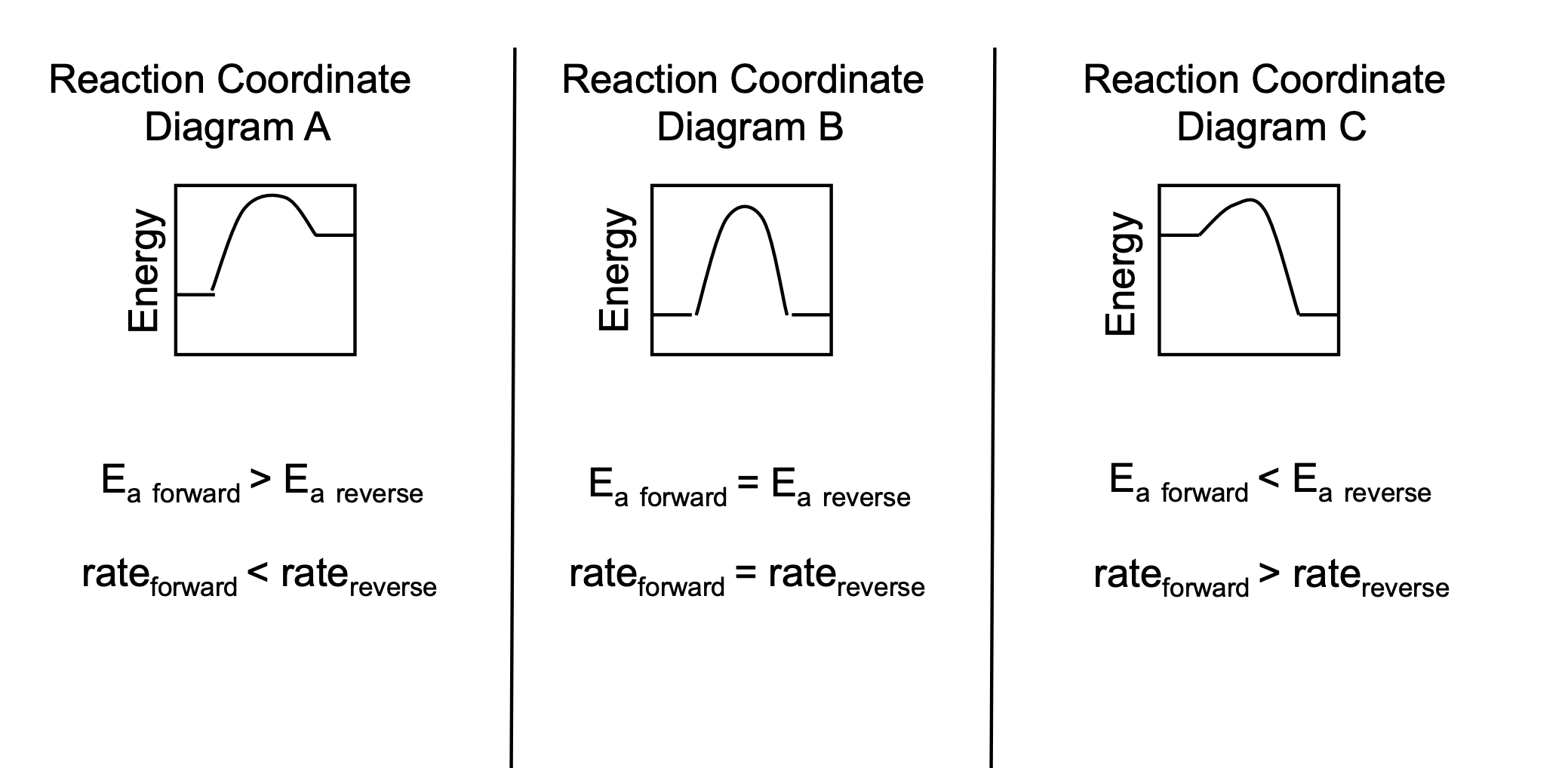
Comparing and Contrasting the Kinetics of Various Reactions
Comparing and Contrasting the Thermodynamics of Various Reactions
Le Chatelier’s Principle
If a system at equilibrium is disturbed by changing the conditions, the system will respond to counteract that change and reestablish equilibrium
(Examples of “disturbances” include changing [reactant], [product], and/or temperature)
Adding reactants OR after removing products: “Shift equilibrium to the right.”
forms more products
Decreases reactants
Removing reactants OR after adding products: “Shift equilibrium to the left”
Forms more reactants
Decreases products
Endothermic (Reactant + heat ←→ product)
After heating: “Shift equilibrium to the right”
After cooling: “Shift equilibrium to the left”
Exothermic (Reactant ←→ product + heat)
After heating: “Shift equilibrium to the left”
After cooling: “Shift equilibrium to the right”
Brønsted acid
H+/proton donor
Partial positive atom
Brønsted base
H+/proton acceptor
Lone pair on negative or partial negative atom that can be used to form new covalent bond
Brønsted acid-base reaction
The transfer of a proton
Brønsted bases and conjugate acids differ by 1 proton
Brønsted acids and conjugate bases differ by 1 proton
Conjugate Base
Accepts H+ in the reverse reaction
forms after Brønsted acid donates proton
Conjugate acid
Donates H+ in the reverse reaction
Forms after Brønsted base accepts H+
The 7 “Strong Acids”
The acids that dissociate completely in water and undergo Brønsted reactions
Take into account: The nomenclature of conjugate acid and conjugate base is still the same, even if the parent Brønsted acid is weak
HCl + H2O → H3O+ + Cl-
HBr + H2O → H3O+ + Br-
HI + H2O → H3O+ + I-
HNO3 + H2O → H3O+ + NO3-
HClO3 + H2O → H3O+ + ClO3-
HClO4 + H2O → H3O+ + ClO4-
H2SO4 + H2O → H3O+ + HSO4-
Different Definitions of Acids and Bases
Lewis definition:
More broad
Lewis acid: electron pair acceptor
Lewis base: electron pair donor
Brønsted defintion:
Brønsted acid: H+/proton donor
Brønsted base: H+/proton acceptor
Note: Brønsted reactions always meet the definition for Lewis reactions, but lewis reaction are not always Brønsted reactions
Mechanism arrows and how to determine where they are
Mechanism arrows represent how electrons (not atoms) are moved around
1) Identify bonds broken and bonds formed to determine if the reaction meets Lewis definition
2) Start with a molecule that is donating a pair of electrons
Start arrow at the source of electrons: lone pair
A bond to which atom? That’s what the arrow points to
How to know where the bond is being formed:
Lone pairs attach to a partial positive Hydrogen atom
3) Go to the molecule that takes in lone pairs
Start arrow at the source of electrons: bond breaking
When a bond breaks, lone pairs go to the atom that is not forming a bond
What atom? That’s what the arrow points to
How to know where to find where the lone pair is being formed
The atoms that are partially negative or negative that have a bond with the partially positive hydrogen atom
A higher energy species is
More reactive (wants to change)
Less stable
Analogous to a strong acid and/or strong base

A lower energy species is
Less reactive
More stable
Analogous to a weak acid and/or weak base

Ka – Acid Dissociation Constant
The equilibrium constant (Keq) for an acid reacting with water
A- = Conjugate base
H3O+ = Conjugate acid
HA = Brønsted acid
Water (the Brønsted base) is considered a pure liquid and doesn’t appear in the equilibrium expression
The stronger the acid, the larger the [A–][H3O+], the larger the Ka
The weaker the acid, the smaller the [A–][H3O+], the smaller the Ka
![<p>The equilibrium constant (K<sub>eq</sub>) for an acid reacting with water</p><ul><li><p>A<sup>- </sup>= Conjugate base</p></li><li><p>H<sub>3</sub>O<sup>+</sup> = Conjugate acid</p></li><li><p>HA = Brønsted acid</p></li><li><p>Water (the Brønsted base) is considered a pure liquid and doesn’t appear in the equilibrium expression</p></li><li><p>The stronger the acid, the larger the [A–][H3O+], the larger the K<sub>a</sub> </p></li><li><p>The weaker the acid, the smaller the [A–][H3O+], the smaller the K<sub>a</sub></p></li></ul><p></p>](https://assets.knowt.com/user-attachments/7e34ef6f-f0d9-45c4-907f-aab7d35e516d.png)
pKa
Simpler way to depict Ka
pKa = -log(Ka)
The stronger the acid, the smaller (more negative) the pKa
The weaker the acid, the bigger (more postitive) the pKa
In order to qualitatively compare the relative strength of acids:
Consider the stability of the conjugate base
The more stable the conjugate base (A-), the stronger the parent acid (HA)
Product (Conjugate base) formation is favored when: Products (Conjugate base) are lower in energy
Reactants (Parent acid) are more reactive when: Reactants (Parent acid) are higher in energy
How to qualitatively access relative acidity
May need to find the corresponding conjugate base first. If so, determine what the molecules would look like after they give away a partial positive proton.
1) Most important: An atom holding a negative charge in a conjugate base
Same row: Electronegativity
Same column: atom size
spread out—charge across a bigger surface area = lowering energy
2) 2nd most important: Consider resonance in the conjugate base
Equally preferred: Energy is blended out
Spread out charge = lower energy
The more variations of resonance structures, the more stable the conjugate base
3) 3rd most important: Consider e-donating and e-withdrawing groups in conjugate base
If the polar charge is facing away from the atom with the negative charge, then it’s more stable
If the polar charge is facing towards the atom with the negative charge, then it’s not stable
My assumption (not in notes): If both are facing away from the atom with the negative charge, then compare the electronegativity and size of the more electronegative atom.
The reaction is extensive if
the reacting acid is much stronger than the produced conjugate acid
The conjugate base relationship with the reactants preferred
More stable conj. base / Weaker base = reactants preferred / reverse reaction is effective
Less stable conj. base / Stronger base = reactants are even more preferred / reverse reaction is even more effective
Keq relationship with pKa
Keq = 10(pKa of conjugate acid - pKa of reacting acid)
pKa = -log(Ka)