CHAPTER 13 – CHEMICAL KINETICS
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
rate it is expressed as a…
change per unit time
Speed Formula
(Change in distance / Change in Time ) = (delta X) / (delta t)
In chemistry, we are concerned with:
rate of decrease [reactant] and increase in [product]
Chemical kinetics is the area of chemistry concerned with…
reaction rates and the sequence of steps by which a reaction occurs.
For chemical reactions, the reaction rate is measured as the….
change in the amounts of reactant/product divided by the change in time.
Rate equation
(delta concentration)/(delta t)
RATES OF CHEMICAL REACTIONS 5 For the general reaction:
𝑎𝐴 + 𝑏𝐵 → 𝑐𝐶 + 𝑑𝐷
The reaction rate can be determined with any of the reaction species:
correct
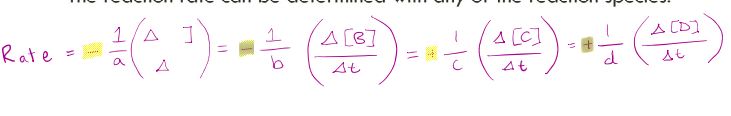
For the following reaction, the rate may be measured in three ways.
𝑁2 + 3𝐻2 → 2𝑁𝐻3
correct
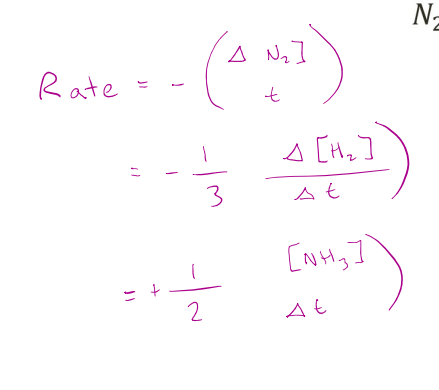
Changes of reactant and product concentrations over time can be monitored over time by….
measuring a property of the species that is proportional to its concentration
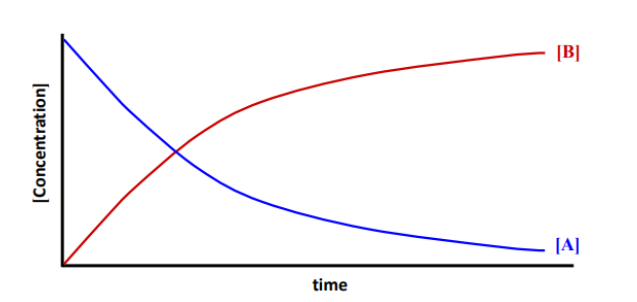
The graph shows the concentration vs. time for the following reaction:
𝑁2 + 3𝐻2 → 2𝑁𝐻3
Which curve best represents nitrogen?
b)
The average rate of a reaction can be calculated between any…
two times during the reactions progress.
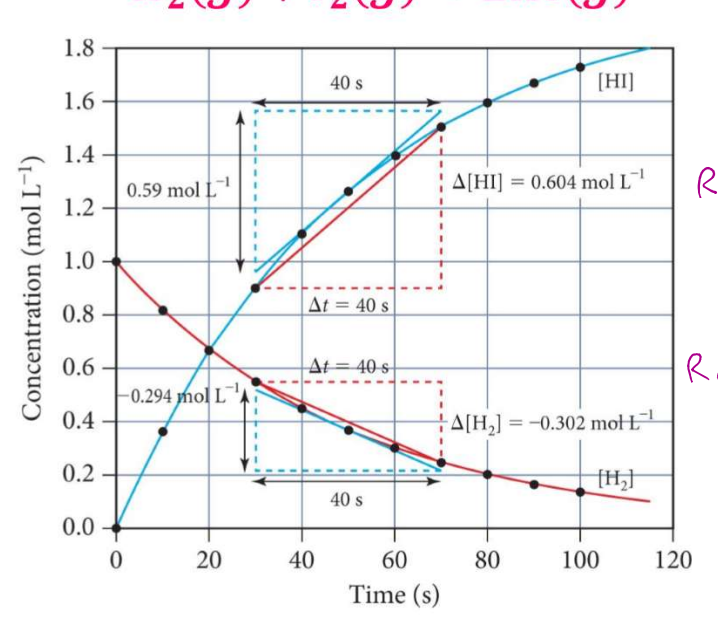
Calculate AVERAGE RATES OF REACTION:
𝑯𝟐(𝒈) + 𝑰𝟐(𝒈) → 𝟐𝑯𝑰(𝒈)
correct
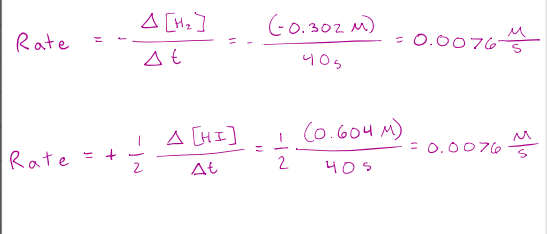
The instantaneous rate of a reaction for a specific point in time is given by….
the slope of the tangent to the curve at that time.
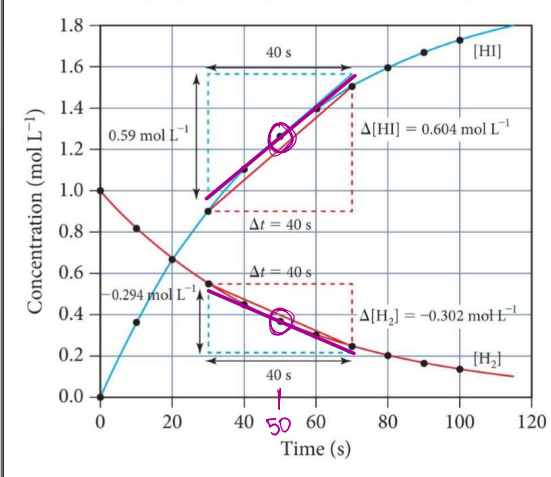
Calculate INSTANTANEOUS RATES OF REACTION 10
𝑯𝟐(𝒈) + 𝑰𝟐(𝒈) → 𝟐𝑯𝑰(𝒈)
correct
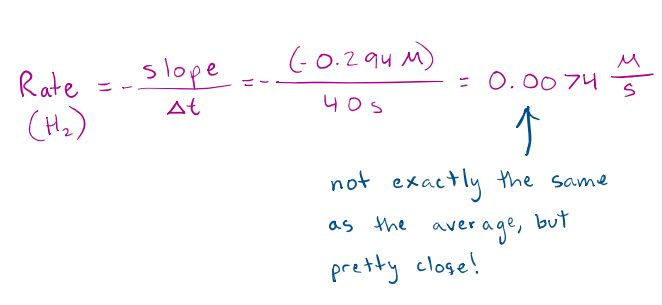
For the first 10.0 s of the following reaction, the concentration of I dropped from 1.000 M to 0.868 M.
𝐻2𝑂2 𝑎𝑞 + 3𝐼 − 𝑎𝑞 + 2𝐻 + 𝑎𝑞 → 𝐼3 − 𝑎𝑞 + 2𝐻2𝑂(𝑙)
Calculate the average rate of reaction over this interval.
correct
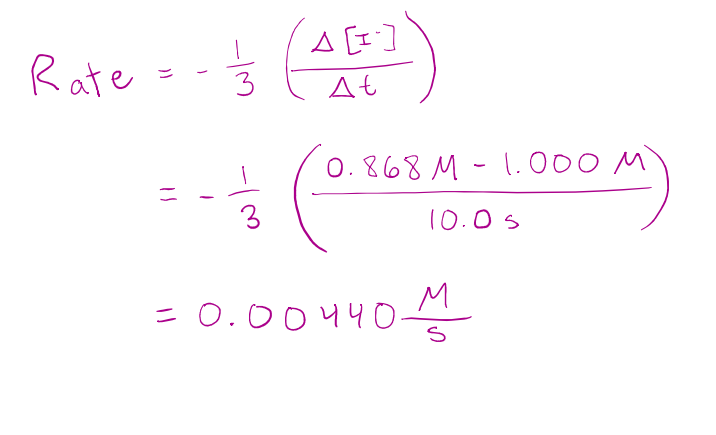
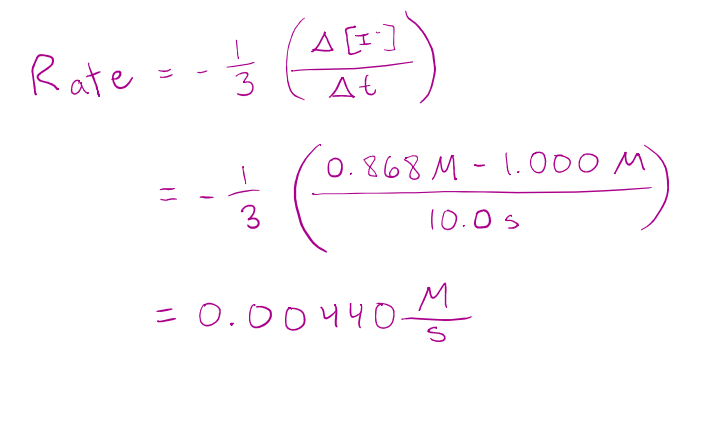
Using your answer from the previous question, predict the rate of change in concentration for the H+.
𝐻2𝑂2 𝑎𝑞 + 3𝐼 − 𝑎𝑞 + 2𝐻 + 𝑎𝑞 → 𝐼3 − 𝑎𝑞 + 2𝐻2𝑂(𝑙)
correct
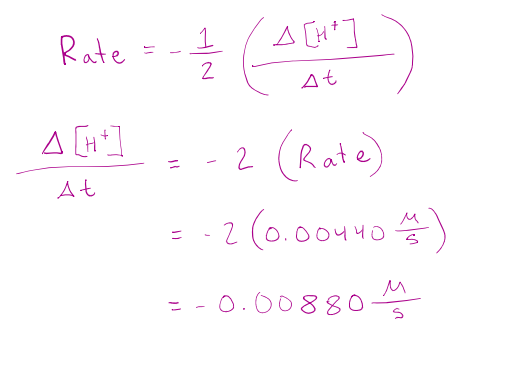
The rate of a reaction often depends on the concentration of….
one or more of the reactants and slows as the reactant concentrations fall.
The rate law describes….
the relationship between the concentrations of the reactants and the rate of the reaction.
The rate law
correct

ZERO-ORDER REACTIONS: RATE=
K[A]^0
ZERO-ORDER REACTIONS: RATE=K[A]^0 14 The rate of the reaction is…
independent of the concentration of reactant species A
What does slope look like for K[A]^0
straight line
FIRST-ORDER REACTIONS: RATE=
K[A]^1
FIRST-ORDER REACTIONS: RATE=K[A]1 15 The rate of the reaction is directly…..
proportional to the concentration of reactant species A
What does slope look like for K[A]^1
straight slope
FIRST-ORDER REACTION: if you doubled [A]…
Rate doubles
SECOND-ORDER REACTIONS: RATE=
K[A]^2
SECOND-ORDER REACTIONS: RATE=K[A]^2 The rate of the reaction is…
proportional to the square of the concentration for reactant species A (quadratic relationship)
What does slope look like for K[A]²
Quadratic
THE RATE CONSTANT (K) (3 factors)
Specific to rxn as written, Temperature dependent, units vary with rxn order.
Consider the reaction below 2𝐴 → 2𝐵 + 𝐶 If the rate of reaction is directly proportional to the concentration of A, this reaction is ____
D. A first-order reaction
The order of a reaction can be determined only through…
experiment!
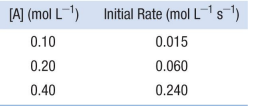
Use the data provided below to determine the order of reaction for the simple reaction 𝐴 → 𝑝𝑟𝑜𝑑𝑢𝑐𝑡s
correct

REACTION ORDER FOR MULTIPLE REACTANTS Consider the following reaction which has multiple reactant species:
𝑎𝐴 + 𝑏𝐵 → 𝑐𝐶
Rate =
k[A]^n[B]^m
n → order for A
m → order for B
overall rxn order = m+n
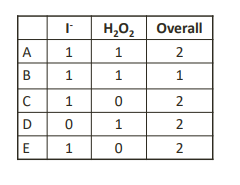
For the reaction:
𝐻2𝑂2 + 3𝐼− + 2𝐻+ → 2𝐻2𝑂 + 𝐼3 −
The rate law is: 𝑟𝑎𝑡𝑒 = 𝑘 𝐻2𝑂2 𝐼 −
What is the order for each reactant and the overall reaction order?
2
![<p>Determine the reaction order for H2 and NO, as well as the overall reaction order for the following reaction. </p><p>2𝐻2 + 2𝑁𝑂 → 2𝐻2𝑂 + 𝑁2 </p><p>What is the value of the rate constant, k, and its units?</p><p>(To determine the order with respect to H2 we must hold the concentration of the other reactant constant. This way, the NO is not affecting the reaction rate)</p><p>(Rate 1/ rate2) = (k[H2]^n[NO]^m / ibid.)</p>](https://assets.knowt.com/user-attachments/34804840-df59-42be-887a-7ef5bd7bcf89.png)
Determine the reaction order for H2 and NO, as well as the overall reaction order for the following reaction.
2𝐻2 + 2𝑁𝑂 → 2𝐻2𝑂 + 𝑁2
What is the value of the rate constant, k, and its units?
(To determine the order with respect to H2 we must hold the concentration of the other reactant constant. This way, the NO is not affecting the reaction rate)
(Rate 1/ rate2) = (k[H2]^n[NO]^m / ibid.)
n = 1 , m = 2, rate = 1250 M^-2 S^-1
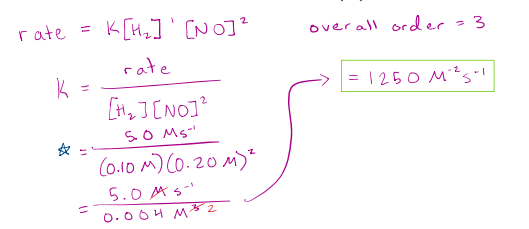
Experimental data has shown that the rate law for the reaction below is Rate = k[HgCl2 ][C2O4 2- ]^ 2
2𝐻𝑔𝐶𝑙2 𝑎𝑞 + 𝐶2𝑂4 2− 𝑎𝑞 → 2𝐶𝑙− 𝑎𝑞 + 2𝐶𝑂2 𝑔 + 𝐻𝑔2𝐶𝑙2(𝑠)
How will the rate of reaction change if [HgCl2 ] is doubled and [C2O4 2- ] is tripled?
D. The rate will increase by a factor of 18
Integrated rate laws provide the relationship between…
reactant concentration and time.
Suppose the reaction A → products is zero-order. The rate does not depend on…. only….
[A], only k.
A plot of ___ vs. time : ZERO-ORDER REACTIONS
[𝑨]𝒕
![<p>[𝑨]𝒕</p>](https://assets.knowt.com/user-attachments/6ac66d3f-a57d-42a7-8b54-ccddbafa202c.png)
Suppose the reaction A → products is first-order. The rate is directly proportional to…
[A].
FIRST-ORDER REACTIONS: plot of ___ vs. time
𝐥𝐧[𝑨]𝒕
![<p>𝐥𝐧[𝑨]𝒕</p>](https://assets.knowt.com/user-attachments/9560ac30-4cf2-42cd-8403-5755ae416d77.png)
Cyclopropane rearranges to form propene gas: 32 The reaction is first-order in cyclopropane and has a measured rate constant of 3.36 x10-5 s -1 at 720K. If the initial cyclopropane concentration is 0.0445 M, what will the cyclopropane concentration be after 235.0 minutes?
correct

Suppose the reaction A → products is second-order. The rate is proportional to….
[A]^2 .
SECOND-ORDER REACTIONS: A plot __ vs time
1/[A]t
![<p>1/[A]t</p>](https://assets.knowt.com/user-attachments/76371c6e-4e29-4dd6-bb14-83ab5b0d03ed.png)
For the following reaction, a plot of 1/ [𝑁𝑂2] vs. time yielded a straight line with a slope of 0.255 L mol-1 s -1 .
𝑁𝑂2(𝑔) → 𝑁𝑂 𝑔 + 𝑂(𝑔)
If the concentration of NO2 after 5.0 minutes is 0.00567 M, what was the initial NO2 concentration?
correct

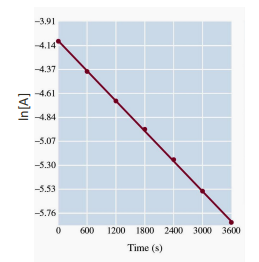
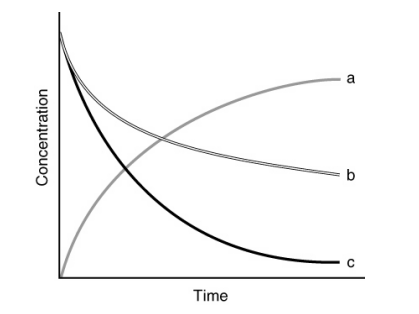
Kinetics data was measured for the decomposition of A and is shown in the graph below. What is the order of reaction with respect to A?
B) First-order
A student prepares a graph of the concentration of A vs. time for the reaction:
𝐴 → 𝐵 + 𝐶
The graph is a straight line with a negative slope. What can you conclude about the order of the reaction?
C. It is a zero-order reaction
The half-life (𝒕𝟏/𝟐) of a reaction is the time it takes…
for a reactant to fall to one-half its original value.
The half-life is dependent on…
the order of the reaction.
integrated rate law:
𝒍𝒏 [𝑨]𝒕 = −𝒌𝒕 + 𝒍𝒏 [𝑨] 0
The decay times for first-order reactions are…
constant.
The decay times for first-order reactions are constant. The decay does not depend on….
the concentration of the reactant.
FIRST-ORDER HALF-LIVES: Fraction remaining =
[A]t / [A]0 = 0.5^n
n = Time elapsed / (t 1/2)
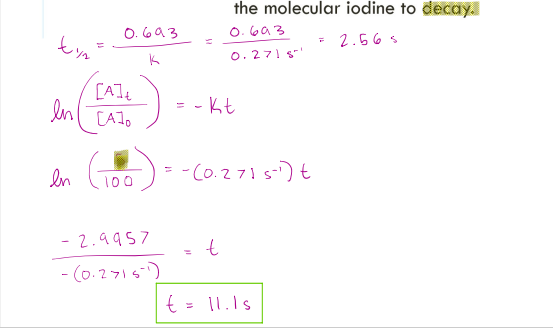
Molecular iodine dissociates at 625 K with a first order rate constant of 0.271 s-1 .
Calculate the half-life of the reaction and the time for 95% of the molecular iodine to decay.
correct
SECOND-ORDER REACTIONS Integrated rate law: 𝟏 [𝑨]𝒕 = 𝒌𝒕 + 𝟏 [𝑨]�
correct

ZERO-ORDER REACTIONS Integrated rate law: [𝑨]𝒕= −𝒌𝒕 + 𝑨
correct

A decomposition reaction, with a rate that is observed to slow down as the reaction proceeds, is found to have a half-life dependent on the initial reactant concentration. Which of the following best describes this reaction?
C) A doubling of the initial concentration of the reaction results in a quadrupling of the rate.
Generally, as temperatures are increased, we see increased…
rates of chemical reactions.
The Arrhenius equation shows…
the temperature dependence of reaction rates through the rate constant.
Arrhenius equation
correct

Even though a reaction may energetically favourable, _____ is needed for the reaction to take place.
an input of energy
The activation energy (Ea ) is…
the energy barrier that must be overcome for the products to form.
So, why do activation energy barriers exist?
Ea is E required to reach the transition state. Larger Ea leads to slower Rxn rates.
Reactions proceed through a _____ state that is ….
transition state that is higher in energy than either the reactants or products.
ARRHENIUS EQUATION – THE EXPONENTIAL FACTOR: represents the…
fraction of molecules with enough energy to make it over the activation barrier.
ARRHENIUS EQUATION – THE EXPONENTIAL FACTOR: k =
𝐴𝒆^( −𝑬𝒂 /𝑹T)
As Ea increases, exponential factor k… Rate…
decreases, decreases
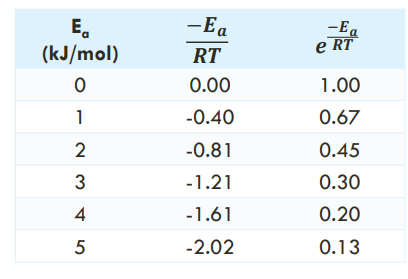
For a reaction performed at 25 °C (298 K): THE EXPONENTIAL FACTOR AND EA (on the table, when is Ea increasing?)
top to bottom Ea increases
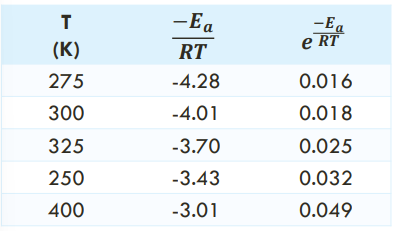
THE EXPONENTIAL FACTOR AND TEMPERATURE : For a reaction with an Ea of 10.0 kJ/mol: (K and Rate)
As T increases, exponential factor and k increase. Rate increases as T increases.
THERMAL ENERGY DISTRIBUTIONS: The higher the temperature… (3)
the wider the energy distribution and the greater the average energy.The fraction of molecules with enough energy to surmount the activation energy barrier and react increases
Arrhenius plots can be used in… (2)
he analysis of kinetic data to measure the frequency factor (A) and the activation energy (Ea)
Arrhenius plot formula
lnk = -(Ea)/R * (1/T) +lnA
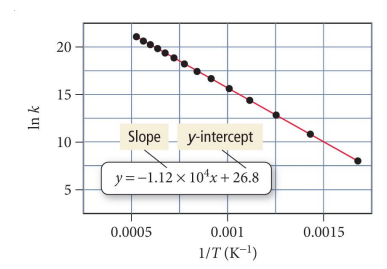
Using the data from the Arrhenius plot for the decomposition of ozone, determine the rate constant at 298 K.
𝑂3 𝑔 → 𝑂2 𝑔 + 𝑂(𝑔)
correct
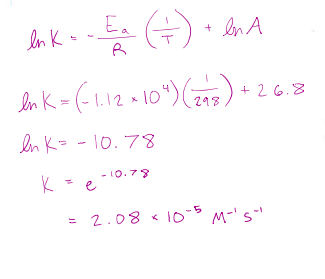
TWO-POINT ARRHENIUS EQUATION =
correct
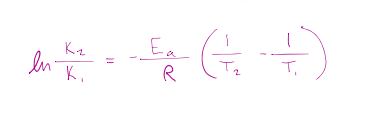
Consider the following reaction:
𝑁𝑂2 𝑔 + 𝐶𝑂 𝑔 → 𝑁𝑂 𝑔 + 𝐶𝑂2(𝑔)
The rate constant is measured to be 2.57 L mol-1 s -1 at 701 K and 567 L mol-1 s -1 at 895 K.
What is the activation energy for the reaction, in kJ/mol?
correct
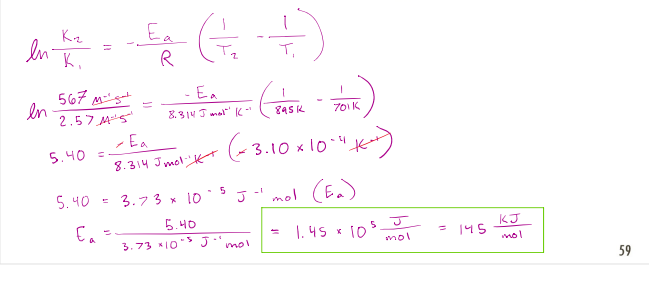
The frequency factor (A) is…
the number of approaches to the activation barrier per unit time.
he exponential factor tell us if a collision has…
enough energy.
THE COLLISION MODEL: The frequency factor tells us….
how many of the right kind of collision occur
The orientation factor (p) is the…
fraction of collisions which have the proper orientation for reaction.
Since the orientation factor is a probability, the values usually range from…
0-1.
THE ORIENTATION FACTOR, P: If p ≈ 0,
few collisions are effective
THE ORIENTATION FACTOR, P: If p ≈ 1
nearly all collisions are effective
Which of the following reactions would you expect to have the smallest orientation factor?
C) 𝐻𝐶𝑙 𝑔 + 𝐻𝐶𝑙 𝑔 → 𝐻2 𝑔 + 𝐶𝑙2(𝑔)
REACTION MECHANISMS
describes the series of steps that occur.
The individual steps in a mechanism are known as…
elementary steps
Each elementary step has its own…
Ea and rate constant.
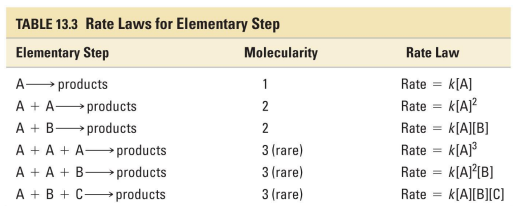
Molecularity refers to…
how many species react together in an elementary step.
unimolecular
1 reactant particle
bimolecular
2 reactant particles (which may or may not be the same)
termolecular
3 reactant particles (rare)
The following reaction occurs by the set of elementary steps listed below:
2𝑁𝑂 + 𝑂2 → 2𝑁𝑂2
Step 1: 𝑁𝑂 + 𝑂2 𝑁𝑂3 (fast)
Step 2: 𝑁𝑂3 + 𝑁𝑂 → 2𝑁𝑂2 (slow)
What is the rate law for the slow step?
D) rate = k[NO3 ][NO]
The reaction cannot occur faster than the…
slowest step.
The rate law of the slow step determines…
the rate of the overall reaction.
rate determining step
The slowest step in the mechanism
This slowest step will have the
largest activation energy.
Determine the overall reaction, the rate determining step, and the rate law for the following mechanism.
Step 1: 𝐴 + 𝐵2 → 𝐴𝐵 + 𝐵 (slow)
Step 2: 𝐴 + 𝐵 → 𝐴𝐵 (fast)
correct
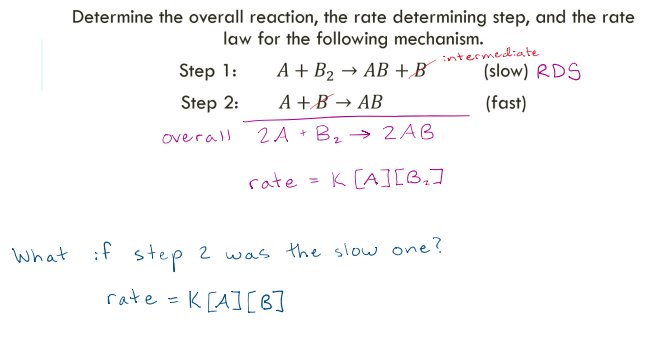
If the rate determining step is not the first step, the rate law will include…
an intermediate.
We want to express the rate law in terms of..
reactants only as intermediates are very hard to measure.
For a fast elementary step at equilibrium, the rate of the forward reaction is equal….
to the rate of the reverse reaction.