Unit 7
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Reactants
the substance(s) that undergo a change during a reaction.
Products
the substance(s) that are formed during the reaction.
Diatomic Elements
Pure elements that naturally exist as molecules composed of two atoms bonded together.
Law of Conservation of matter
matter cannot be created or destroyed in a chemical reaction; it can only change forms.
Activity series
ranking of cations that participate in single-replacement reactions.
Precipitate
a solid that forms from a solution during a chemical reaction.
Soluble
Will dissolve in water.
Insoluble
Products will not dissolve in water, instead forming a solid precipitate.
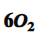
Subscript of oxygen
2 in chemical formulas indicates that there are two oxygen atoms present.
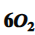
coefficient of oxygen
6 indicates there are six separate molecules of diatomic oxygen.
what does aqueous (aq)
Aqueous, meaning a substance is dissolved in water.
Single replacement
A single-replacement reaction occurs when a metal takes the place of another metal within an ionic compound.
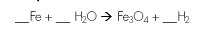
Balance the equation
3:4:1:4
Decomposition
Decomposition reactions take a large molecule and break it down into two or more smaller ones.
Opposite of synthesis.
Synthesis Reactions
A synthesis reaction occurs when two or more elements or compounds combine to form a single, larger molecule.
Double Displacement Reactions
In a double displacement reaction, the cations of two ionic compounds will switch places.
Combustion
Combustion is a reaction between a carbon-based fuel and oxygen, producing carbon dioxide and water vapor.
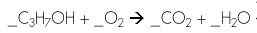
Identify the following type of reaction
Combustion
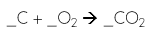
Identify the following type of reaction
Synthesis