Lecture 8: Enzyme Kinetics (not reviewed)
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
True or false: the enzyme is a better fit for the transition state than for the substrate itself and thus lowers the activation energy
True
What is differential binding
enzymes binding to substrates through induced fit and thus contorting the substrate into a structure more similar to the transition state
What is binding energy and why is it important
Binding energy is the free energy released when an enzyme binds its substrate weak interactions
then stabilizes the transition state and lowers activation energy, speeding the reaction.
How does bidding energy influence enzyme specificity
it disallows the incorrect substrate from binding to an incorrect enzyme because the contacts necessary to achieve the necessary binding energy are to lower activation energy and stabilize the transition state
True or false: when a substrate is completely held in the active site, it take the characteristics of the transition state
True
True or false: Like ΔG, the rate of a reaction is constant
False, a rate is not constant, it changes over time
What are the phases in enzyme catalysis
Pre Steady State
Transition Point
Steady State
What is the pre-steady state
initial transient period during which the concentration of the enzyme-substrate complex builds up as they form
the enzyme is picking up substrates
What is steady state
the period in which the enzyme-substrates complex an other intermediates remain constant
the enzyme cannot pick up any more substrate
concentration of ES is constant
V0 (initial velocity)
What is the timescale of the pre-steady state
microseconds
What is the timescale of the steady state
seconds to minutes
Where does Micheal’s Menten live?
at the steady state!!!
What is initial velocity (V0)
the rate of the product formation when time is 0, and its so early before product accumulates
True or False: at the steady state the product can turn around and inhibit the formation of the ES complex
True
What happens other than velocity a we keep increasing the concentration of substrate
more ES complex is formed
What is Vmax
the maximum velocity the enzyme can achieve, where eery enzyme molecule is occupied
not a true max, due to eq it is not possible to MAX
initially btw
If the rate of increase of V0 (initial velocity) slows what does that mean?
free enzyme is becoming scarce
What is a first order reaction
Rate = k[s], where th rate is linear with concentration of substrate (inc substrate inc rate)
What is a zero order reaction
Rate = k, where the rate is independent of the concentration of substrate
no matter how much S you add, the rate is unfuckwitable
What is k1
the rate of the enzyme and substrate creating the enzyme-substrate complex, ES.
binding!!
what is k-1
the rate of the enzyme-substrate complex falling apart and disassociating into the E and S respectively
What is k2
the rate of the enzyme-substrate complex being catalyzed into the product.
What is Km
the michealis menten constant, that is unique to each enzyme and is independnet of enzyme concentration
tells us the affinity of an enzyme for its substrate
substrate concentration at Vmax/2
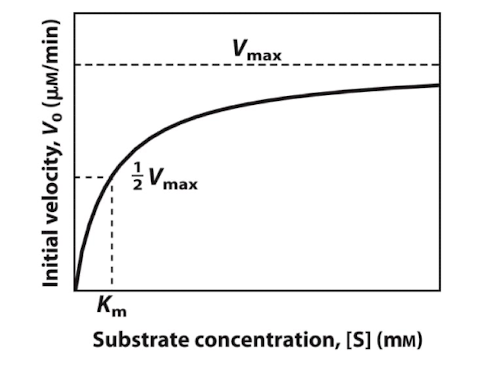
What is the michealist menten equation and what does it describe
It is the Velocity at time zero = Vmax (conc of S) over Km + conc of S
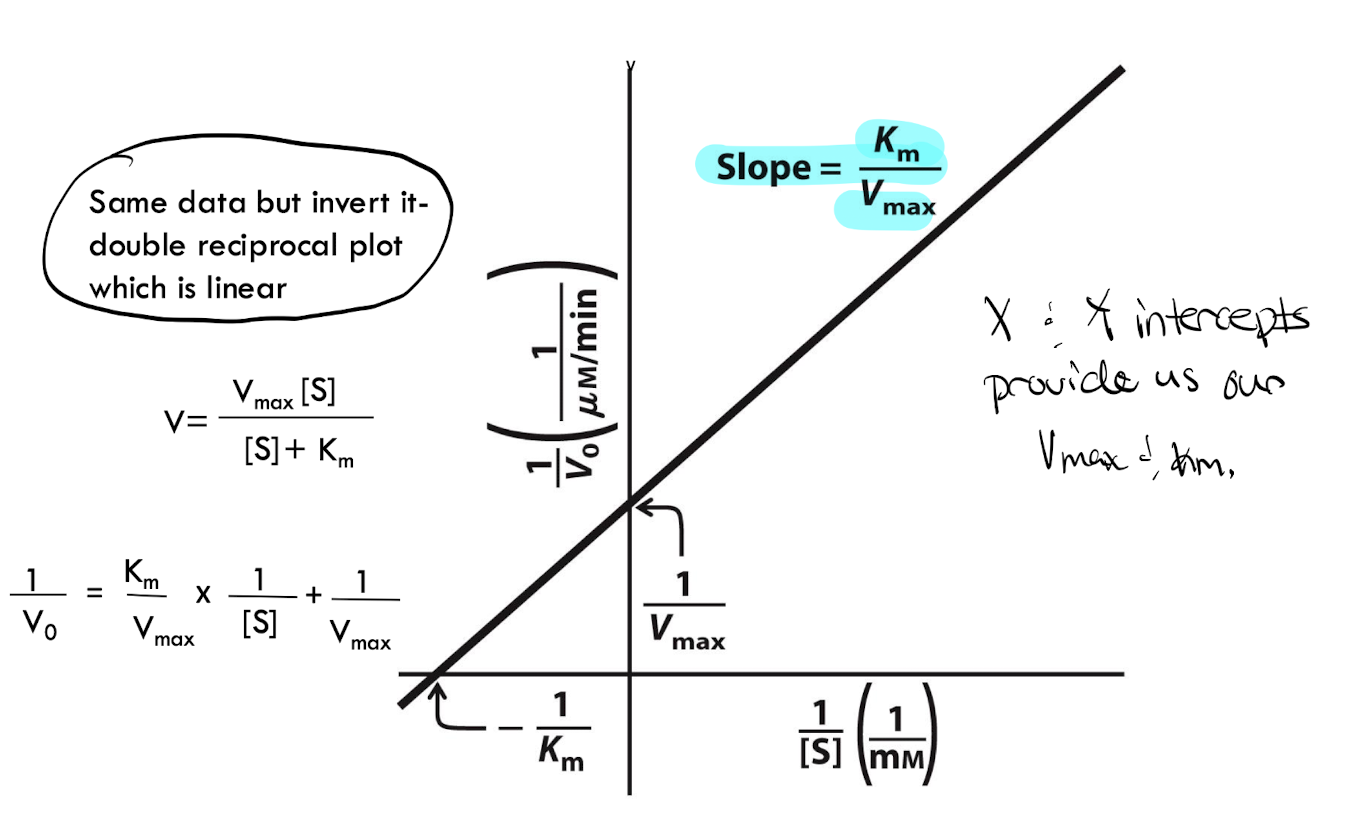
What is the Lineweaver Burk-Plot
it is the Michealis-Menten Equation plot but linearized by taking the reciprocal of both sides
What are assumptions being made for the michealist menten equation
the reverse reaction (P —> E + S) doesn’t matter since the initial rate velocity is what we care about and at that point there are no products
The ES complex is a steady state intermediate so the concentration of ES stays relatively stable
Remember, with nichealis mentin, you’re looking at V0 which is th initial velocity where time is ZERO
ok past Asare
What is the slope of the Lineweaver-Burk Plot?
Km/ Vmax
What does the Y-intececpt of the Lineweave-Burk Plot tell you?
1/Vmax
What does the X intercept of the Lineweaver-Burk Plot tell you?
the -1/Km
what is turnover #
the number of S converted to P by E when fully saturated
What is an enzyme reaction involving a ternary complex ? (sequential)
A SEQUENTIAL kind of enzymatic reaction where there are multiple reactants and all substrates bind to their enzyme before any product is released.
Also, a ternary complex of the enzyme and both substrates form
What are the two kinds of enzymatic reactions involving a ternary complex?
ordered ( E+S eq ES1 eq E1S2 —→E + P1 + P2
non-ordred same as above but circular
What happens to the lineweaver-burk plot when the enzymatic reaction involves ternary complexes
there will be INTERSECTING lines telling us that the reaction is sequential
What is the enzymatic reaction in which NO ternary complex is formed?
Ping Pong/Double Displacement mechanism, a reaction having multiple substrates in which one or more products are released before all substrates bind the enzyme
crank em out as quick as possible
What is a ternary complex
a complex with three parts, enzyme, substrate, and substrate
seen in sequential enzymatic reactions
What happens to the Lineweaver-Burk plot one the enzyme and substate participate in ping pong/double displacement mechanisms
the lines become parallel
True or False: Michelias Menten enzymes are not passive and can be fine tuned signaling molecules
False, micheal’s menten genetic are passive and rely solely on concentration and in turn cannot be fine tuned via signaling molecules
True or false: Michealis Menten genetics do not apply to allosteric enzymes
True, allosteric enzymes are under different kinetic rules
True or False: Michealis Menten Genetics are ubiquitous, applying to all enzymes
FALSE
What differentiates allosteric enzymes from Michealis Menten enzymes
allosteric enzyme have as secondary regulator separate from the active site that allow the cell to turn enzyme activity up or down while MM enzymes cannot be regulated outside of concentration changes
What is an allosteric enzyme?
a enzyme with an active site and allosteric site(s) that when bound changes the confirmation of the enzyme making it high or low affinity thus regulating enzymatic activity
What are homotropic enzymes
homotropic enzymes are allosteric enzymes where both th active site and allosteric site bind to the same substrate
What are heterotropic enzymes?
heterotropic enzymes are allosteric enzymes where the active site and allosteric site bind to different substrates
What is feedback inhibition
a mechanism in allosteric enzyme pathways where the product(s) of the pathway inhibits one enzymatic reaction and thereby regulates the pathway
What are the two kinds of allosteric enzyme regulation
reversible and non reversible
where non covalent binding is reversible but covalent modifications can be irreversible OR reversible
Allosteric enzymatic pathways depend on alteration in the quaternary strcutre
ok!
What are the two models for cooperative binding
Concerted Model
Sequential Model
What is cooperative bidding
something that occurs only in allosteric enzymes
when something bids to the allosteric site taking the confirmation from Tense ( T, not enzymatically active) to Relaxed (R, enzymatically active)
What is the concerted model for cooperative binding
the model for cooperative ending that says that all active site must be in the same state at the same time
all R or all T no hybrid
What are the R and T confirmations and what do they mean?
R is relaxed and allows for pro eenzymatic activity, while T is tense and disallows enzymatic activity
What is the sequential model for cooperative binding
the model for cooperative ending intact says that allotter centimes can undergo sequential changes in structure so you can have hybrids of R and T
You see a signmoid curve when you have what type of enzyme?
allosteric
What are the chacateristcis of a allosteric enzyme graph plot?
one curve showing low activity (tense), one showing high activity (R)
What are allosteric regulators
small molecules that can be positive or negative thus shift the sigmoidal curve