Instrumental Analysis Test 1 Review
1/116
Earn XP
Description and Tags
Hehe (THE ONLY FLASHCARDS YOU COULD EVER NEED)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
117 Terms
What is the order of electromagnetic spectrum from lower to higher energy?
Radio, Micro, Infrared, Visible, Ultraviolet, X-ray, Gamma Ray, Cosmic Rays
Radiation that interacts with molecular rotations
Microwaves
Radiation that interacts with the nuclear/electron spin
Radio Waves
Radiation that interacts with bond vibrations
Infrared waves
Radiation that interacts with inner core electrons/ionizes
X-rays
Radiation that interacts with valence electrons
UV and Visible light
Equation for absorption given extinction coefficient
A = ecl
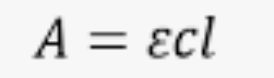
Equation for % Transmittance
%T = P/Po
Equation for absorption given transmittance
A = -Log(%T)
Why is A more useful than %T
Absorbance allows for a linear form of data and has a direct relationship to the concentration of the substance measured.
What are some common UV-Vis materials?
Plastic (Polystyrene and PMMA) and Glass (Quartz and Borosilicate)
For Beer’s Law, what is the most common source of error?
Sample Prep
For Beer’s Law, what is the absorptivity coefficient dependent on?
Wavelength
For Beer’s Law, The range of the monochromatic light needs to be ___ than the absorption band
Shorter
For Beer’s Law, the wavelength of light should be selected at what value?
at the highest absorbance (lambda max)
What concentration should solutions be for UV-Vis?
equal or less to 0.01 M
UV-Vis can analyze which states of matter…?
Solids, Liquids and Gases
For UV-Vis, what are some reasons why wavelength should be selected at the maximum sample absorbance
To minimize error that can occur by choosing a reliable position. If the wavelength is off a little near the peak, there is little change in measured absorbance.
Can measure lower concentrations due to the light being most absorbed at the specific wavelength, and thus decreasing interference.
For UV-Vis measurements, what absorbance range is ideal precision?
0.3-2
For UV-Vis, why does the compartment need to be closed?
To prevent stray light
What types of energy in a Jablonski diagram are considered when a molecule absorbs a photon?
Rotational, Electronic and Vibrational
Fluorescence emission occurs at ___ wavelengths than the excitation wavelength?
longer wavelengths
How does Phosphorescence compare with Fluorescence in terms of energy?
Fluorescence emits a higher energy while Phosphorescence is a lower energy emission
The excited state of a molecule in which electron spins are parallel?
Triplet
Lowest energy state of a molecule?
Ground State
Emission from an excited T1 to ground S0 state?
Phosphorescence
Excited state of a molecule in which electron spins are opposite
Singlet
Emission from excited S1 to ground S0 state
Fluorescence
Radiation emitted from a heated object (incandescence)
Blackbody radiation
What are the lifetime ranges for Phosphorescence and Fluorescence?
P = 10^-4 to 10^2 s and F = 10^-8 to 10^-4 s
What is the typical light source for IR?
SiC Globar
What are some types of UV-Vis light sources?
Xenon Arc Lamp, Tungsten Halogen lamp, Deuterium Arc Lamp, Lasers, and LEDs
what do modern UV-Vis specs use for separating light?
grating monochromator
What do modern IR spec’s use for separating light
Michelson Inferometer
what are common sample cell materials for IR?
Diamond, NaCl, Germanium, KBr and ZnSe
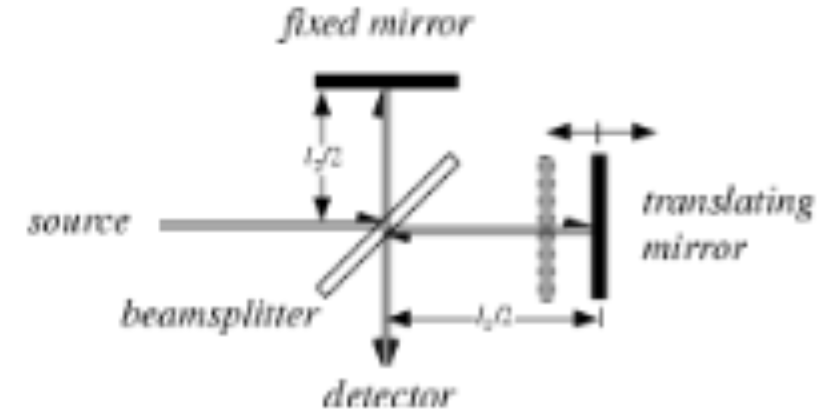
How does Michelson Inferometer work?
Light source from SiC rod goes through slit to beam-splitter. Splits light 50/50 to both a movable and stationary mirror. Light reflects and recombines at the beam-splitter and hits sample and detector. Recombined bean is either out or in-phase and creates a interferogram.
Equation to find frequency given wavelength
c = λv
Equation to find energy given wavelength
E = hv —> E = hc/λ
What is up with absorbed vs appearing colors and stuff?
Color absorbed is complementary to the color that appears. This is because the eye perceives the remaining combination of non-absorbed light that is reflected.
What are the common UV-Vis detectors?
CCD(charge-coupled device), PDA(photodiode array) and PMT(photomultiplier tube)
What are the common IR detectors?
Ferroelectric(DTGS) and Pyroelectric(MCT)
What are the advantages of PDA compared to PMT?
Fast scanning
records entire spectrum at one time
more rugged due to no moving parts
Cheaper
What are the advantages of PMT compared to PDA?
Lower detection limit/Better resolution
Less stray light
Name of the cathodes in PMT
dynodes
how much can a PMT multiply electrons?
A MILLION!!!!
What is used in PDA to disperse light
Grating polychromator (because entire spectrum is emitted onto the array)
Why are CCD detectors becoming more popular in spectroscopy?
Combines the advantages of both PDA and PMT detectors in being fast and having low detection limits
What are the different ranges in light sources and give examples?
Line (Diode laser), Narrowband (LEDs), Broadband (Lamps and SiC globar)
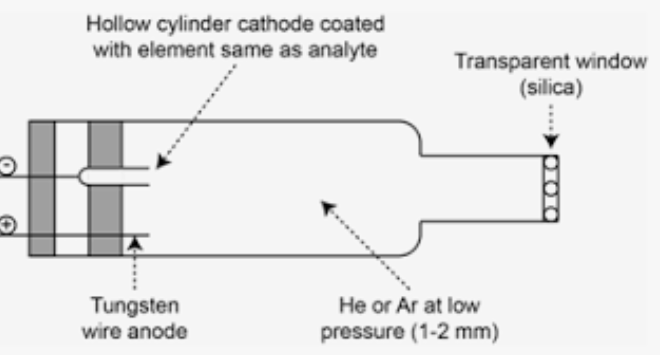
How does a hollow cathode lamp work?
A cathode, made from the element whose emission line is being tested, is sent into the gas phase through the use of He or Ar cations to then emit an atomic emission through the collision of high energy electrons.
Symmetrical vibrations are better seen with which instrument?
Raman
Asymmetrical vibrations are better seen with which instrument?
IR
what is a risk involving the high power of a Raman laser?
The laser can decompose the sample if the power is too high.
Is fluorescence a problem for Raman Spectrums?
Yes, indeed.
What kind of samples can be used for Raman?
Solid and liquid samples
Can Raman instruments use glass cells?
Yes
What must be considered regarding concentration of sample when using Raman over IR?
Sample concentration must be higher for Raman than IR
What range of light sources does Raman usually use?
visible light
Is fluorescence a problem for IR Spectrums?
No!!!!!!!!!!!!!!!!!!!!!!!!!!
What needs to be taken into account regarding water samples for both IR and Raman?
IR will generally detect water and mask important functional groups within that region of the spectrum.
For XRF, which electrons are involved?
Inner shell electrons
Which frequencies of light are used in XRF?
X-Rays
What are two main sources of information that XRF and XRD provides?
crystal structure (XRD)
elemental composition (XRF)
What type of samples can XRF instruments use?
Solid and liquid Samples
What needs to be taken in consideration regarding XRF samples?
they must be homogeneous when determining concentration. (hetero not allowed)
What is the typical range in detection limit for XRF?
ppm to %
Can XRF measure all the elements at the same time, or one at a time?
All at the same time
are XRF and XRD destructive techniques?
NO!! They wouldn’t hurt a fly
What is XRF information in terms of qualitative or quantitative?
XRF is both qualitative (which elements are present) as well as quantitative (how much of each element)
What is the difference between Line spectra, Band spectra, and Continuum spectra in atomic spectroscopy
Line Spectrum: sharp, well-defined spectral lines caused by the excitation of individual atoms (emission and absorption spectrums)
Band spectrum: closely spaced lines that are difficult to separate and end up making “bands” of light. Caused by small molecules or radicals
Continuum spectrum: any radiation produced when solid is heated (blackbody radiation and continuous spectrum)
when a heat source heats up a cold gas, you get this spectrum
Absorption spectrum
when a gas is heated and allowed to cool, you get this spectrum
Emission spectrum
In Atomic spectroscopy, this broadening occurs due to the collisions of molecules deactivating the excited state
Collisional (pressure) broadening
In atomic spectroscopy, this occurs from the rapid motion of atoms as they emit or absorb radiation
Doppler broadening
In atomic spectroscopy, this occurs by the lifetime of excited states
Natural broadening
What are the benefits/properties of ICP?
Simultaneous multi-element analysis
Low detection limits
Wide sensitivity range
How is the sample heated in a Graphite Furnace AAS?
heated electrothermally in a graphite tube
Both FAAS and GAAS use this lamp (and you should too!)
hollow cathode lamp
GFAAS advantages over FAAS
detection limits in ppb
less sample required
Name at least 3 detectors used on UV-Vis instruments
Phototubes
Photomultiplier tubes
Silicon photodiodes (diode arrays)
photoconductive cells (CCDs)
Name at least 2 IR detectors (preferably the important ones cause Eklund wants it like that I guess idk guys this class is evil and I want my degree now)
Thermocouples
Pyroelectric devices
bolometers
pneumatic cells
What is the detector used in FAA/GFAA instruments?
PMTs
Describe how FTIR interferometer works by using a diagram
hehe, you got that my King/Queen/Monarch
Why would you be tempted to use Raman rather than FTIR?
1.If you need visible light source
can see those symmetric vibrations
can use glass cells in case you hate pellets
can use water samples
can use SERS attachment to enhance signal by a MILLION!!!
Why would you be tempted to not use Raman over FTIR?
If you have a low concentration of sample = Bad
Fluorescence (but you can use 1064 nm laser to minimize it)
Sample will probs decompose due to that high power laser
Not that groovy for asymmetric vibrations because Raman only wants the homo ones
What is Bathochromic shifts?
Shift to a longer wavelength in energy (red shift)
What is a Hypsochromic shift?
Shift to a shorter wavelength (blue shift)
If your UV-Vis instrument is a scanning type, what type of detector does it have?
Photomultiplier tube (PMT):
What is Electron Ionization? IS IT HARD OR SOFT?
hits sample with 70 ev, causing sample to break into fragments “fingerprint” of molecule. HARD method
What is chemical Ionization? IS IT HARD OR SOFT?
uses CH5 to collide with molecules to add that extra H and ionize the parent molecule. SOFT method
In mass spec, this is when you add up all the lowest isotopic masses
Monoisotopic Mass
In mass spec, this is when you add up the most abundant masses, and round to nearest integer (common method)
Nominal Mass
In mass spec, this is when you add all the average masses weighed for abundance
Average Mass
What is the most common use for a mass spectrometer?
To be a detector in chromatography, either gas or liquid, or to a lesser extent with an ICP
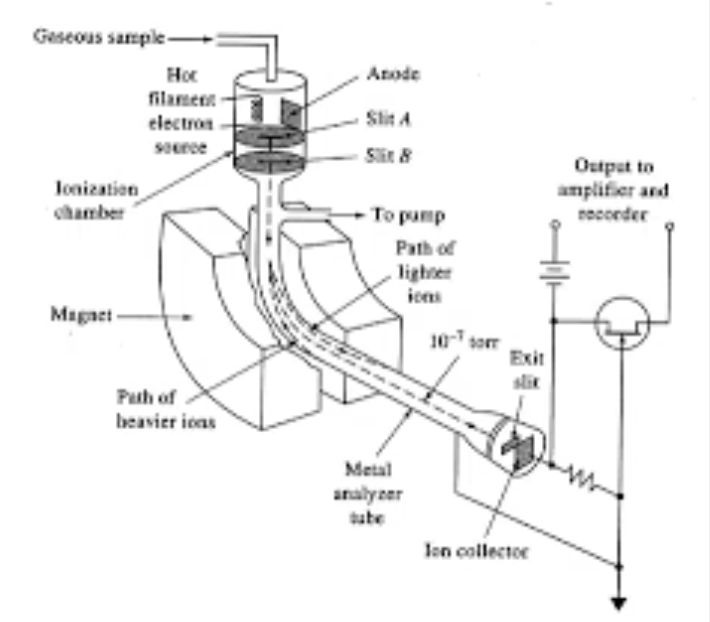
What is this Mass Spec?
Magnetic/electrostatic Sector
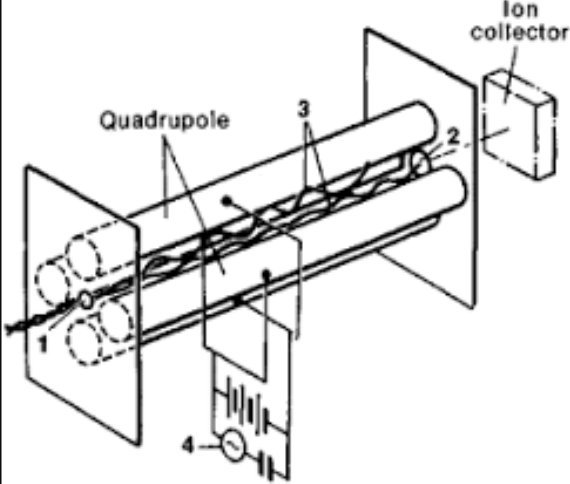
What is this Mass Spec
Quadrupole
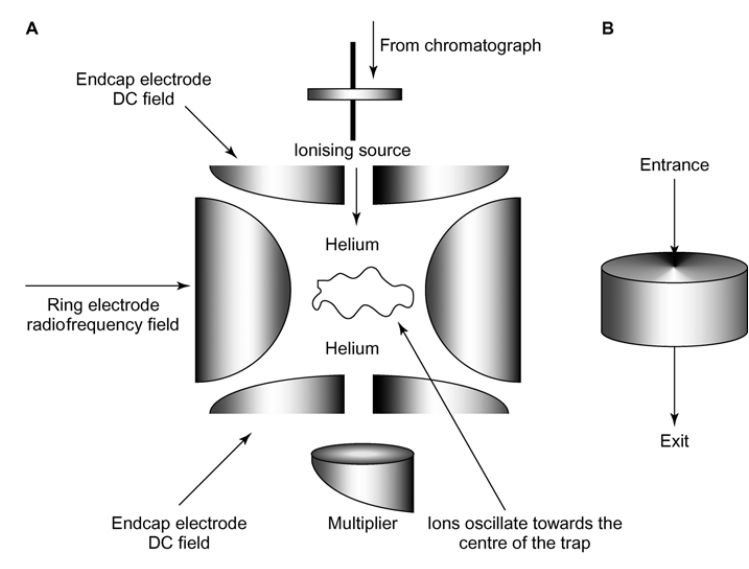
What is this Mass Spec?
Ion-Trap
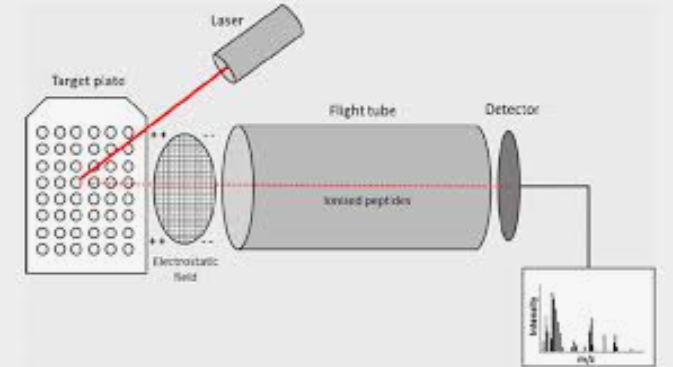
What is this Mass Spec?
MALDI-TOF
HELP!!!, tell me rn what Electronspray is and what it is used in mass spec or Lunch Money will get depressed
Electronspray is a method that produces charged droplets that de-solvate and are directed into a mass analyzer. Used to deliver samples from a LCC to a MS. (If you answered correctly, you saved Lunch Money from having to be medicated)
HEREY HEREY, WHAT DO BE FAAS, GFAAS AND ICP CALLED AND WHAT DO BE THEIR HEAT SOURCES???
FAAS (Flame Atomic Absorption Spectrometry) uses air/acetylene flame
GFAAS (Graphite Furnace Atomic Absorption Spectrometry) uses small graphite tube
ICP (Inductively Coupled Plasma) uses Argon plasma flame
HEREY HEREY, WHAT DO BE FAAS, ICP and GFAAS TECHNIQUES BE BASED ON (absorption or emission)?
ICP: emission
FAAS, GFAAS: absorption