4.8- Organic Synthesis and analysis
1/101
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
How can you increase the chain length of a compound?
By reacting a halogenoalkane with KCN (potassium cyanide) in nucleophilic substitution
What can nitriles be converted into?
Hydrolysis to form a carboxylic acid (warming with dilute acid)
Reduced by LiAlH4 to form an amine
What's another method of increasing the length of the carbon chain?
Nucleophilic addition of HCN to an aldehyde or ketone
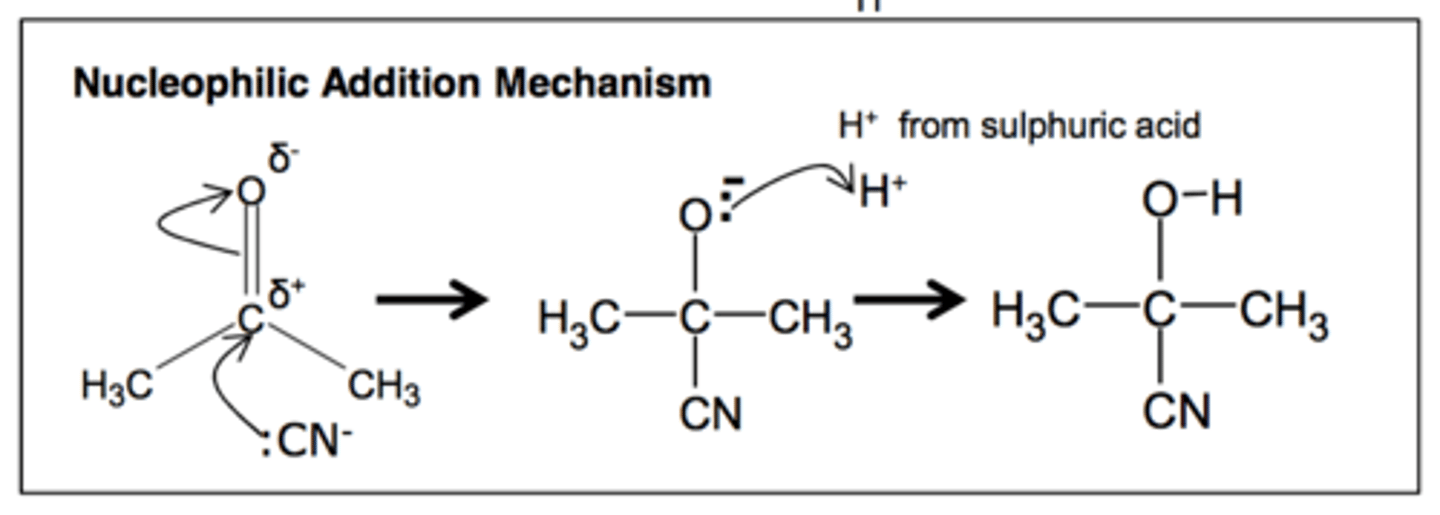
What do you do afterwards?
Hydrolyse it by warming with dilute acid to form a carboxylic acid
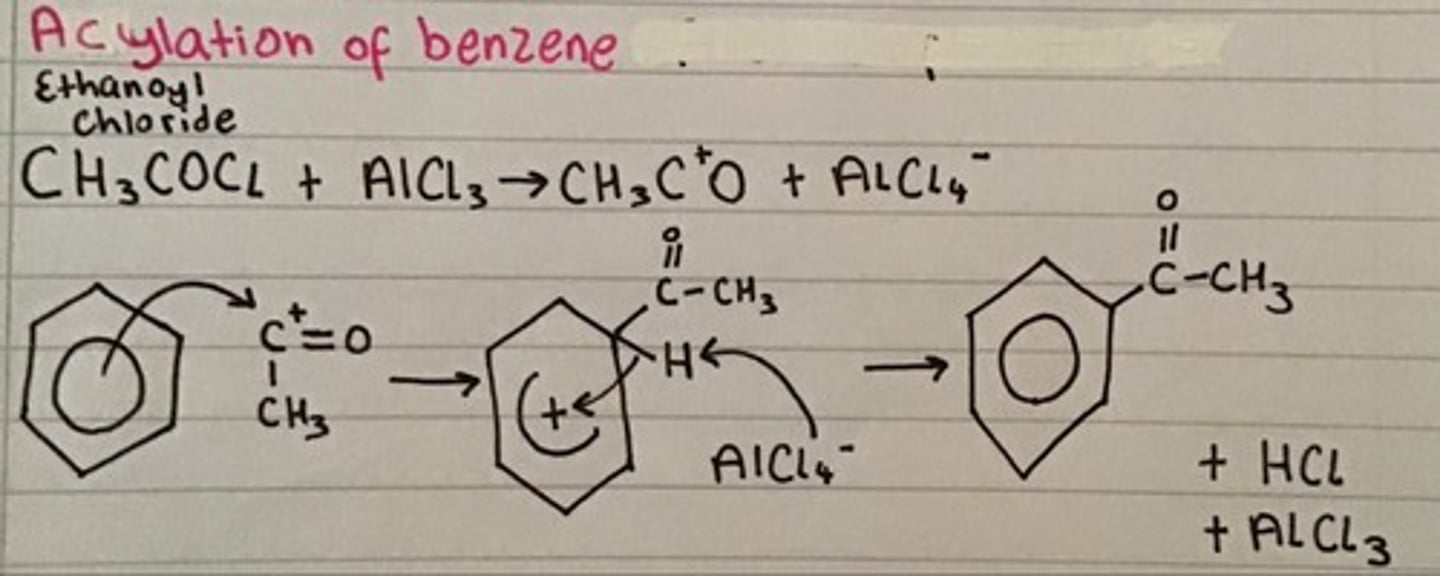
What can be done in aromatic systems?
Friedel-crafts alkylation using CH3Cl
or
Friedel-crafts acylation using CH3COCl (where the Cl bond gets replaced with bond to ring)
How can you decrease chain length?
Using soda lime Ca(OH)2
What will this produce? e.g. from butanoic acid?
CH3CH2CH2COOH + Ca(OH)2 -> CH3CH2CH3 + CaCO3 + H2O
Suggest a way of obtaining benzene from C6H5CH2Cl?
Reflux with NaOH (aq) to form primary alcohol
Oxidise using acidified potassium dichromate (III) to form a carboxylic acid
Decarboxylate using soda lime Ca(OH)2
What does miscible mean?
Liquids are completely soluble in each other at all concentrations
What can be used to separate miscible liquids?
Simple distillation if the boiling point of the product differs by 20 degrees C or more from the boiling points of the other compounds present
What are the other conditions?
If the product does not decompose at or below its boiling temp and the boiling temp is not too high
What can simple distillation also be used to separate?
A volatile liquid from a non-volatile one
What is used if the boiling temps are closer together?
Fractional distillation (fractionating column)
What does the fractionating column enable?
A more efficient separation of the products to occur
What are two examples in which fractional distillation may be used?
The separation of ethanol obtained from the fermentation of sugars
The primary separation of the products present in crude oil
What can be used for compounds that decompose just before or at their boiling temps or have high boiling temps?
Distillation under reduced pressure
or
Steam distillation
What does the reduction in pressure in vacuum distillation enable?
Compounds to boil at lower temps than when distillation occurs at atmospheric pressure
What is steam distillation also used for?
To separate immiscible liquids
What industry is it often used in and why?
Perfume industry- essential oils extracted from plants may decompose if heated to their boiling temps at atmospheric pressure
How does steam distillation work?
Steam is passed into the reaction mixture and the volatile compounds present pass over with the steam and condense in the receiving flask
How does solvent extraction work?
Depends on the differing solubility of a compound in two immiscible solvents
Eg.
iodine is 90 times more soluble in tetrachloromethane than water is
if tetrachloromethane is added to an aqueous solution of iodine and the mixture shaken, most of the iodine is extracted into the tetrachloromethane layer
These two layers can then be separated
What can two immiscible liquids be separated using?
A separating funnel

Which solvent goes above?
The less dense one e.g. oil floats on water because it is tense dense
What can be used to separate an insoluble solid from a liquid?
Filtration
How can this be carried out?
Using filter paper and a funnel
Why is fluted filter paper quicker?
The filtrate only needs to travel through one layer of filter paper and the paper touches the funnel at the folds
What else could be used?
a Buchner funnel (vacuum filtration)
Once the solid is in the funnel, what needs to happen?
The solid needs to be washed with an appropriate solvent and dried
Where dried and what temp?
In the air, or a drying oven at a temp below its melting temp
How can you separate a soluble solid from solution?
Crystallisation
Crystallisation
Heat solution gently over a water bath to boil off solvent
Crystals will form upon cooling
Filter to isolate the crystals
To purify these crystals:
Dissolve the crystals in the minimum volume of hot solvent.
Filter the hot solution to remove any insoluble impurities.
Allow the solution to cool to form crystals.
Filter the mixture to isolate the crystals.
Wash the crystals with a small volume of cold solvent to remove soluble impurities.
Dry the product between two pieces of filter paper in an oven set at a temperature below the product's melting temperature.
Define polymerisation
the joining together of a large number of monomer molecules
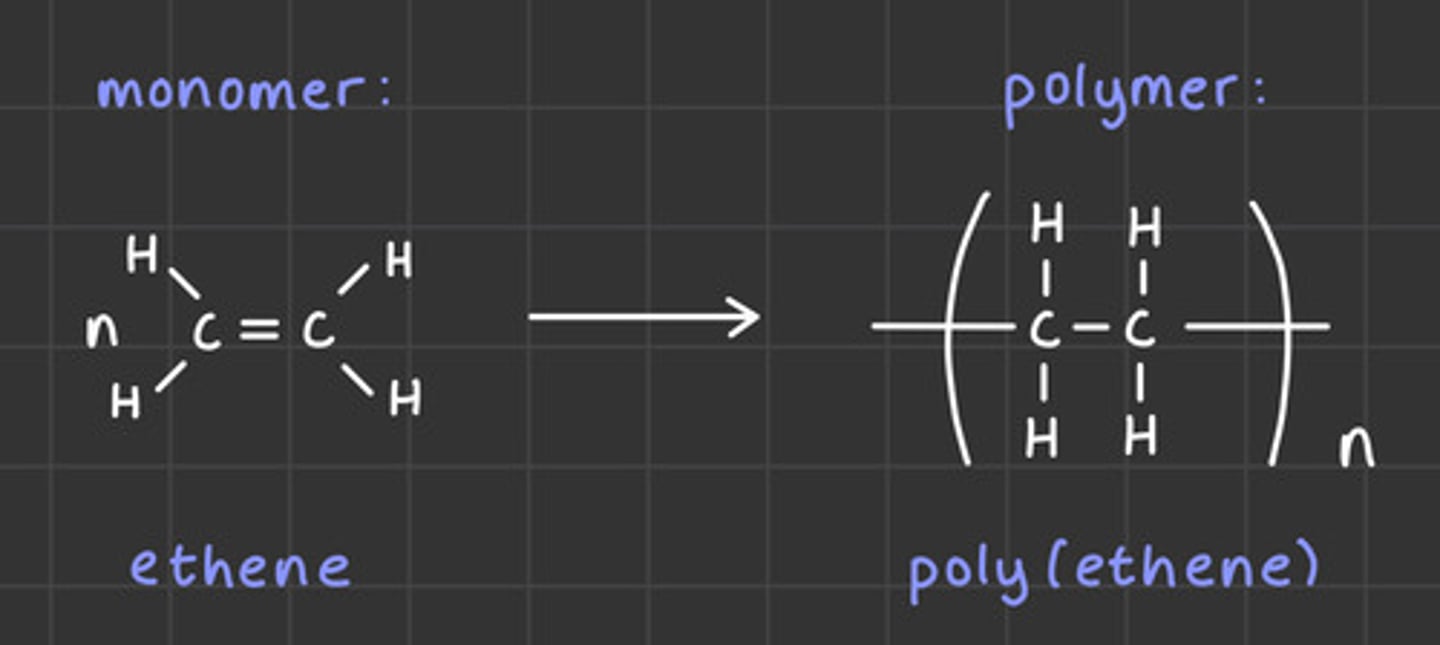
What is addition polymerisation?
When lots of unsaturated monomer molecules - alkenes - open up their double bonds and join together to form polymer chains
How would you draw a polymer of ethene?
What is condensation polymerisation?
Where monomers with two functional groups join together, usually losing small molecules such as water
What does condensation polymerisation occur between?
Two amino acids (between the -OH off one COOH group and the NH off another)
What is the difference between addition and condensation polymerisation?
Addition:
If an alkene
No small molecule is lost
The polymer is the only product
The chain only consists of carbon atoms
Condensation:
The monomer(s) contain functional groups such as -NH2 -COOH or -OH
The chain has an amide link -C(O)N(H)- or an ester link -OC(O)-
Main chain has atoms other than carbon
What is a polyester?
A polymer linked by ester groups
What are polyesters formed from?
A dicarboxylic acid and a diol, forming water as the small molecule that is eliminated
What are the uses of polyesters?
Clothing, packaging and plastic bottles
What is the most common polyester?
PET polyethylene terephthalate
What are the diol and dicarboxylic acid used to make PET?
ethane-1,2-diol and benzene-1,4-dicarboxylic acid
What is PET made into?
synthetic fibres
good insulator so can be used in the making of blankets and as a filling material for duvets
Why may PET not be used?
Not easily biodegradable and there is a need for polyesters that will degrade quickly in landfill
What is one of these?
PLA poly(lactic acid)
What is an added advantage of using PLA over PET?
It can be derived from renewable resources such as corn starch or sugar cane
What is the main difference in their structures?
PLA is made from one type of monomer whereas PET requires two different types of monomer
What are polyamides a product of?
Condensation polymerisation
What are polyamides formed from?
The reaction between a dicarboxylic acid and a diamine
What is the small molecule eliminated?
Water
What are the monomers joined together by?
An amide group/ peptide link
What do they therefore have in their main chain?
Nitrogen
What does a polyamide look like?
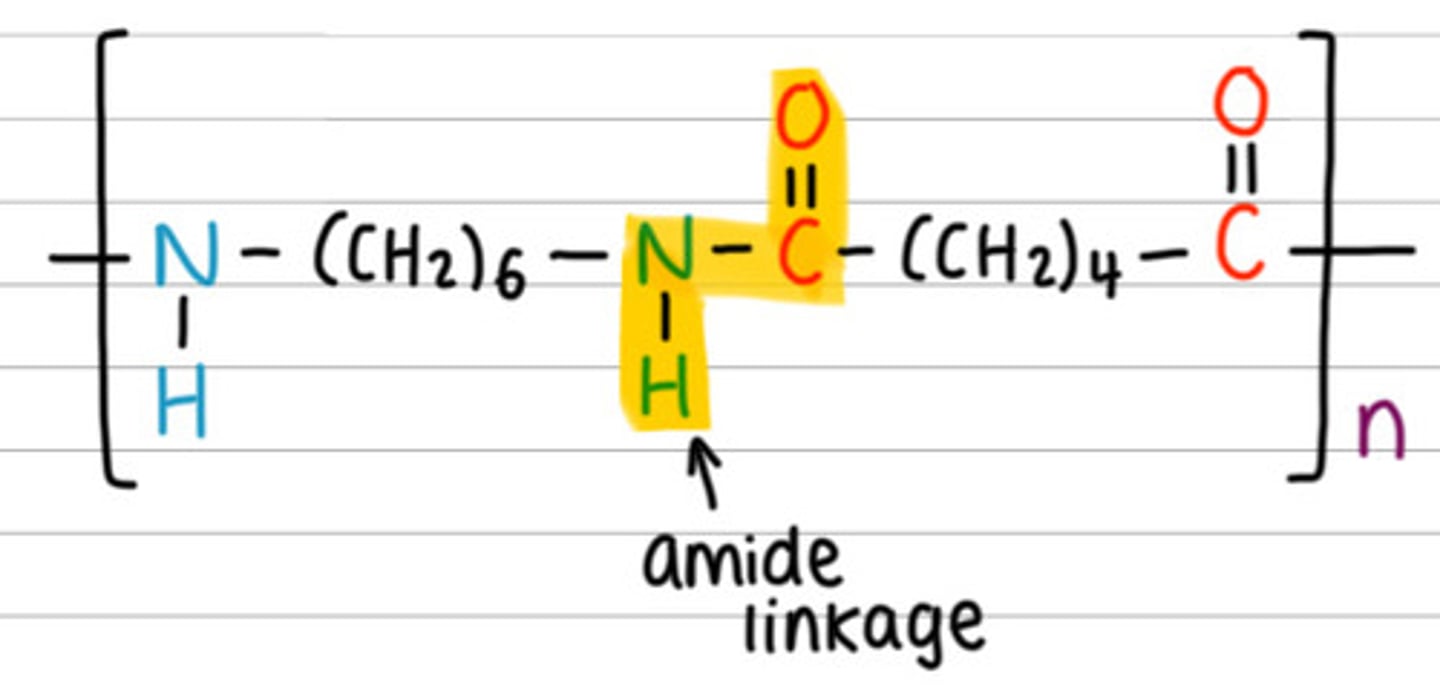
Why is nylon-6,6 called that?
Because each of the two monomers has six carbon atoms
What is another example of a polyamide?
Kevlar
What is this produced from?
Benzene-1,4-dioic acid and benzene-1,4-diamine
What is it used for and why?
Good fire retardant properties and five times stronger than steel, used in bulletproof vests
What is melting temperature?
The temperature at which the solid begins to change into a liquid
What can melting temp be affected by?
The presence of impurities so the value obtained give an indication of a compound's purity
What does the presence of impurities do to the melting temp?
Lowers the expected melting temp and compound melts over a wider range of temperature rather than at a fixed value
Why is it necessary to react aldehydes and ketones with 2,4-DNPH to determine their melting temps from one another?
Difficult to obtain an accurate melting temp for them, whereas derivatives with 2,4-DNPH melting temps are easier to measure
How can melting temps usually be found?
Using a heating bath method
Electrical heating method
Steam distillation- use for something prone to decomposing
What does a heating bath look like?
Stirring rod
Capillary tube
Sample
Heating bath
Heat
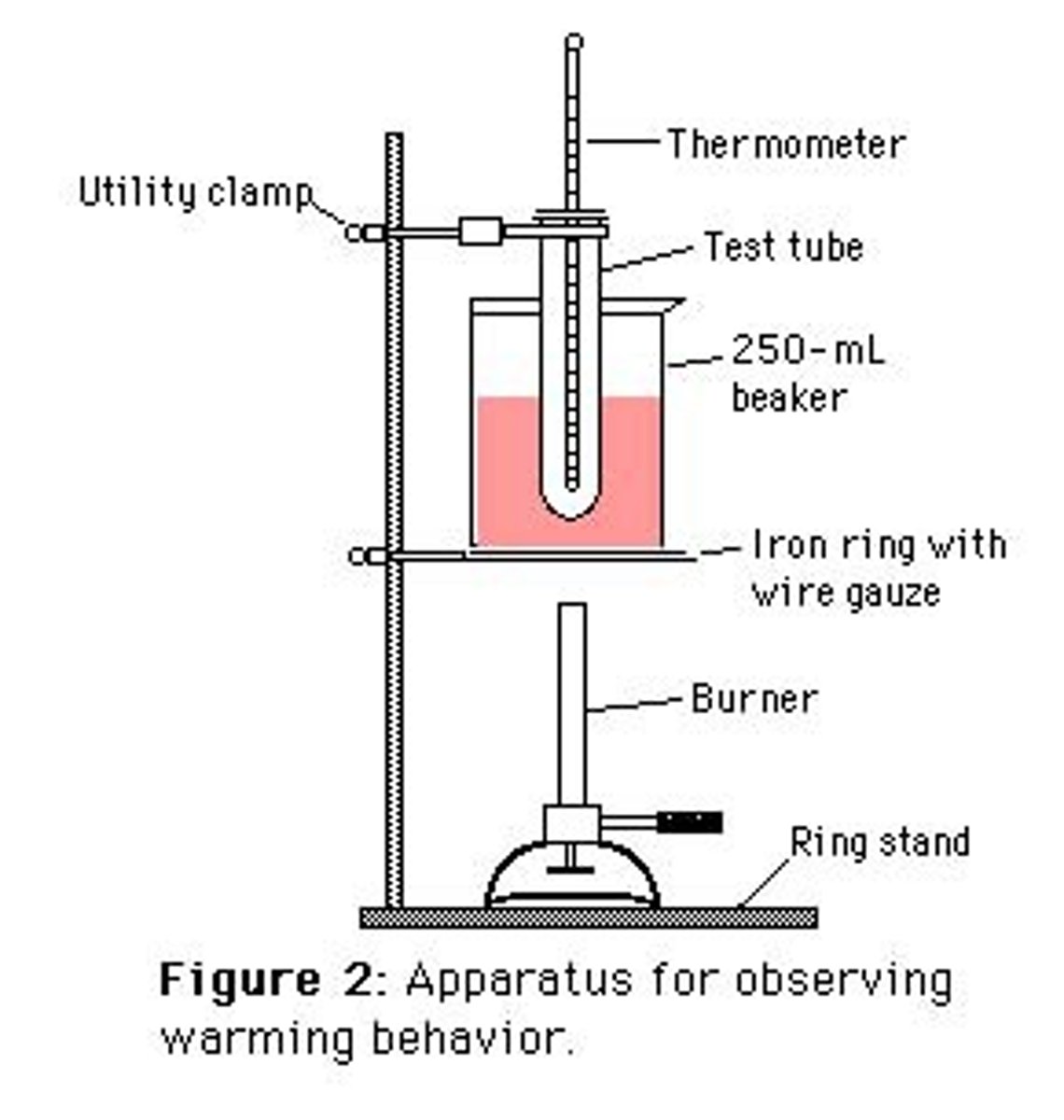
Why do you stir?
So heat is even in water
What is used instead of water for melting temperatures over 100 degrees C?
Silicone oil
What will all of the hydrogen protons be in in ethane on the H NMR spectrum?
Equivalent environments
How many signals are seen?
1 (singlet)
How many environments are seen in chloroethane?
2 as the -CH3 protons are in one environment and the -CH2- protons are in another
What will the peak areas be?
3:2
In 1,2 dichloroethane, what are all of the protons in and what does this do?
The same environment
Single peak (singlet)
How does this differ from that seen in 1,1-dichloroethane?
The hydrogens are in 2 different environments so 2 peaks occur
What happens if the H NMR spectrum of 1,1 dichloroethane is measured using a high resolution spectrometer?
The two peaks are seen to be split
Why?
Because the magnetic environment of a proton or protons in one group is affected by the magnetic environment of neighbouring groups
1,1-dichloroethane
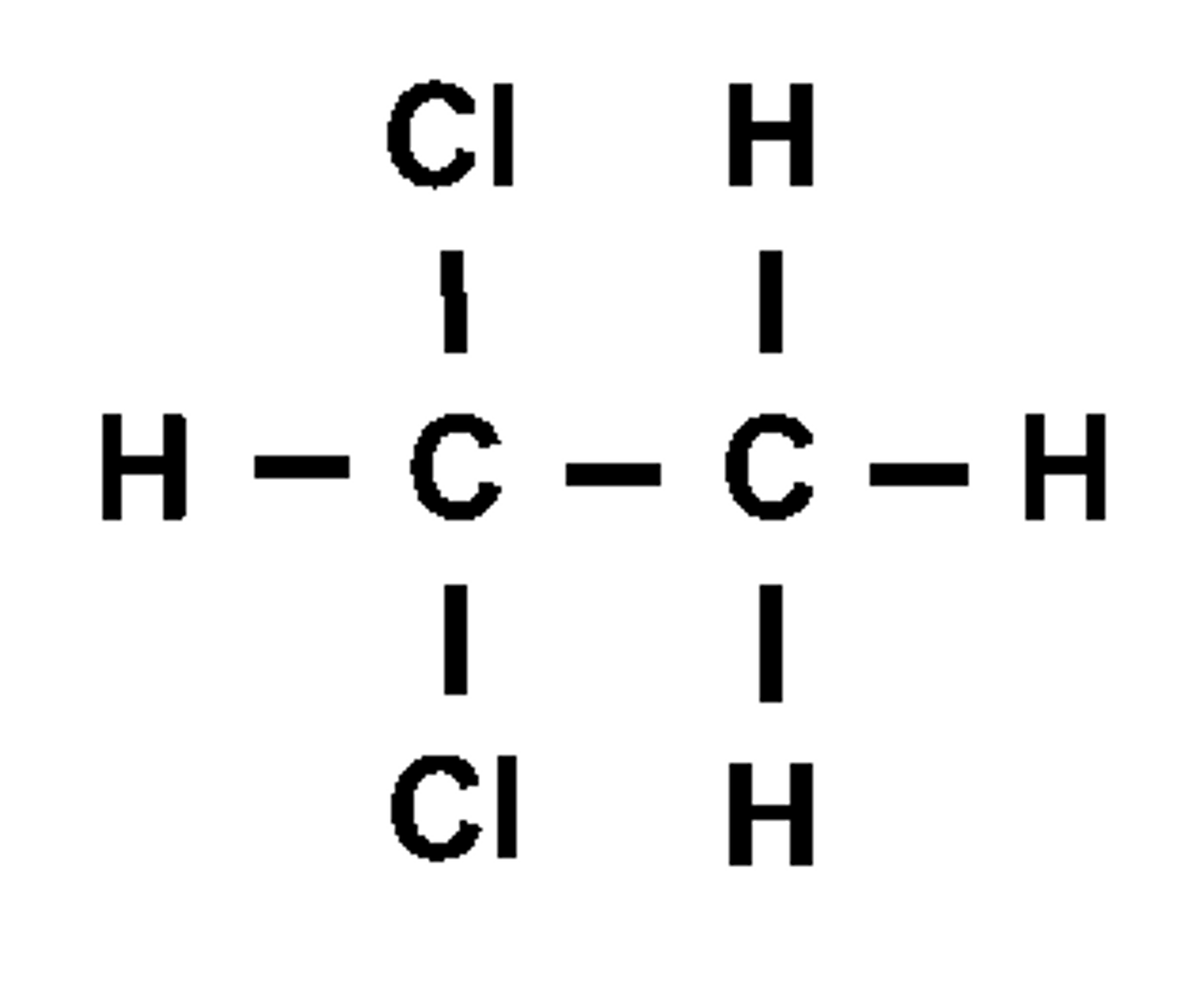
What will the splitting pattern look like?
1 Quartet and 1 doublet
What is the rule followed to give this?
n+1 of adjacent hydrogens e.g. the -CH3 group causes the splitting pattern of the -CH to be a quartet
What is the process of signal splitting called?
Spin-spin coupling
Explain the splitting pattern for ClCH2- C(=O)-CH3
For the CH2Cl it is a singlet
for -CH3 it is a singlet
The hydrogens don't affect each other as they are separated by a Carbon atom that it not bonded to any hydrogen atoms
What is chromatography used for?
To separate substances from a mixture by their slow movement, at different rates, through or over a stationary phase
What is the stationary phase in paper chromatography?
Water trapped in the cellulose fibres of the paper
What is the stationary phase in thin layer chromatography?
A layer of silica (SiO2) or aluminium oxide (Al2O3) coated onto plastic or a glass plate
How are both paper chromatography and TLC carried out?
Spots of the starting materials in a suitable solution are placed at the bottom of a piece of chromatography paper of TLC plate, which are then placed in a suitable solvent with the initial solvent level below the spots
What does the solvent then do?
Rises up the paper/plate, separating the mixture into a series of spots
What happens when the solvent front has moved a suitable level?
The paper/plate is removed and dried
What can you then do?
Calculate the Rf value using :
the distance moved by spot from the starting line/ distance moved by solvent front
What is two way separation?
Sometimes the use of a particular solvent does not completely separate the spots
Overcome by rotating the dried chromatogram through 90 degrees and then using a different solvent
What do you do if the solute causes colourless spots to be produced?
The chromatogram is sprayed with a suitable reagent that causes the spots to become coloured
or
Shine UV light onto the plate. If the separated compounds are fluorescent materials then they show a colour.
or
The plate can have a coating of fluorescent materials and then be exposed to UV light- the spots then show up as dark spots on a fluorescent background
What are the advantages of TLC over paper chromatography?
It is faster and that the thin later on the plates can be made from a variety of materials
What is the most common type of gas chromatography?
gas-liquid chromatography
How does this work?
A gaseous mixture is passed 'through' liquid particles supported on an unreactive solid
The gaseous mixture is swept into the column by a carrier gas
What could the carrier gas be?
Hydrogen, argon or helium
What does the column contain?
Fine solid material or is a hollow column whose walls are coated with a solid on which there is a liquid stationary phase
What is the retention time?
The time taken from the sample entering the injection port until it reaches the detector
What does the efficient separation of the compounds in the mixture depend on?
The volatility of the compound itself, the column temperature, the length of the column and the flow rate of the carrier gas
For similar compounds, what is an important factor is separating the compounds by GLC?
Their boiling temperatures
What is done very often?
The separated compounds of the mixture are passed into a mass spectrometer, where a positive identification can be made
What comes off the column first?
The compound with the lowest boiling temperature
What is HPLC?
High performance liquid chromatography
What happens in this technique?
The column is packed full of solid particles and the mixture sample is dissolved in a suitable solvent. The solution is then forced through the column at high pressure
What is HPLC usually used for?
Compounds that vaporise at high temperatures where they might start to decompose