PATH 381 - Module 2: Blood Gases
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
blood gas analysis allows for assesment of (3)
1. pulmonary gas exchange
2. blood oxygenation
3. acid-base balance
pulmonaru gas exchange (arterial ad venous blood)
arterial blood: (heart→arteries→microvasculature of tissues)
- O2-rich
venous blood:
(microvasculature of tissues→veins→heart)
- CO2-rich
gas exchange
- gas exchange at alveolar-capillary membrane between alveolar membrane in lungs and blood microcapilary endothelium (thin wall = free diffusion of O2 an d CO2 between alveoli and blood)
partial pressure of gas exchange
gases move from high → low partial pressure:
- pO2 alveolar air higher (100 mmHg) than venous blood (40 mmHg) = O2 diffuses from alveoli to blood
- pCO2 alveolar air lower than venous blood = CO2 diffuses from blood to alveoli
Oxyhemoglobin Dissociation Curve (p50, venous blood, arterial blood)
relationship between oxygen saturation of Hb
- p50: pressure where Hb 50% sat
- venous blood: Hb ~75% sat
- arterial blood: Hb 100% sat (100 mmHg)
Shifts of Hb dissociation curve
Left: Hb has increased affinity for O2
= Hb holds O2 tighter = LESS O2 to tissues
Right: Hb has decreased affinity for O2
= Hb holds less tightly = MORE O2 to tissues
Carbon monoxide and Hb dissociation curve
- CO has much higher affinity for Hb than oxygen
= o2 holds tightly to Hb
= curve shifts LEFT
Bicarbonate Buffering System
CO2 + H2O <-> H2CO3 <-> H+ + HCO3-
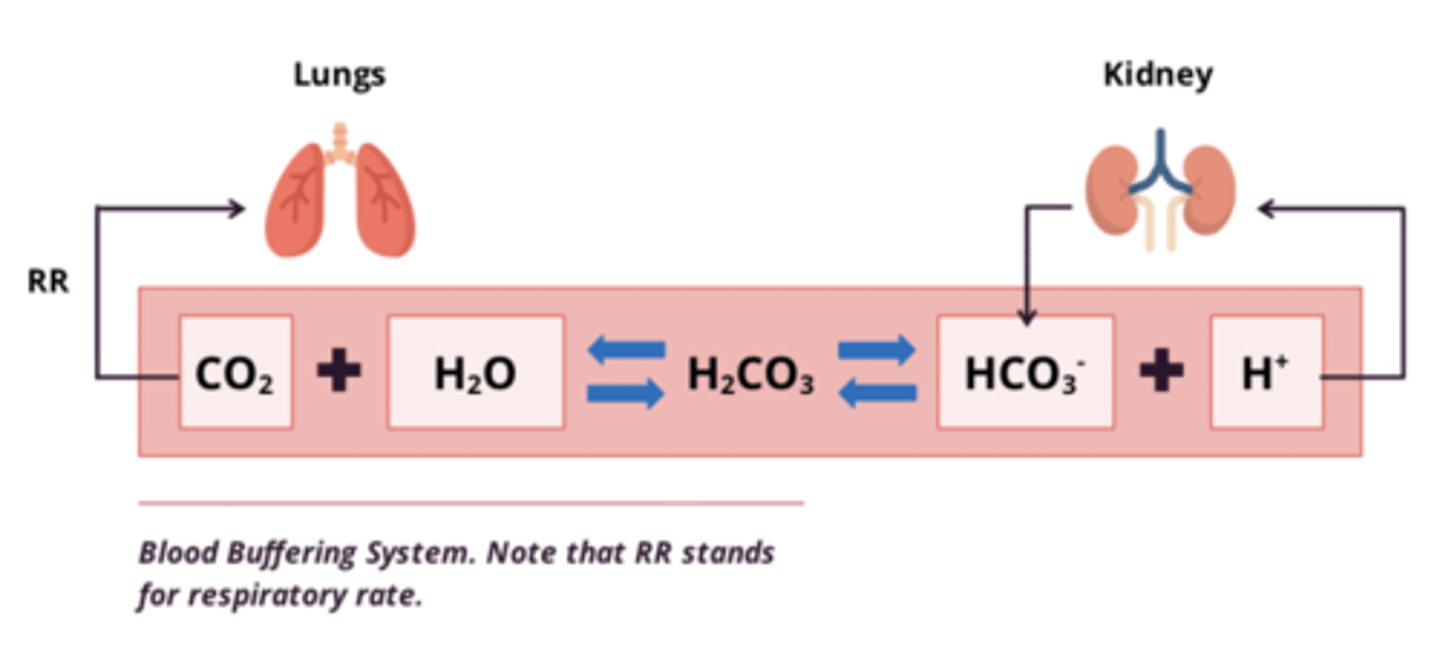
buffering system: changes in CO2 and HCO3-
Changes in CO2:
- when CO2 exhaled = eq shift LEFT = H+ removed from blood
- [CO2] dependent on respiratory rate and [H2CO3]
- increased H+ = increase exhalation of CO2 = shift eq Left = maintain eq
Changes in HCO3-:
- HCO3 produced by kidneys
-H+ (acid) removed by kidneys in urine as NH4+
- eg. too much H+ excreted in urine = shift RIGHT to maintain eq
alterations to blood pH
- pH<7.35: acidosis
- pH>7.45: alkalosis
- changes in HCO3- : METABOLIC
- changes in pCO2: RESPIRATORY
types and characteristics of acidemia
Respiratory acidosis: ↑ pCO2
Metabolic Acidosis: ↓ [HCO3-]
types and characteristics of alkalemia
Respiratory alkalosis: ↓ pCO2
Metabolic alkalosis: ↑ [HCO3-]
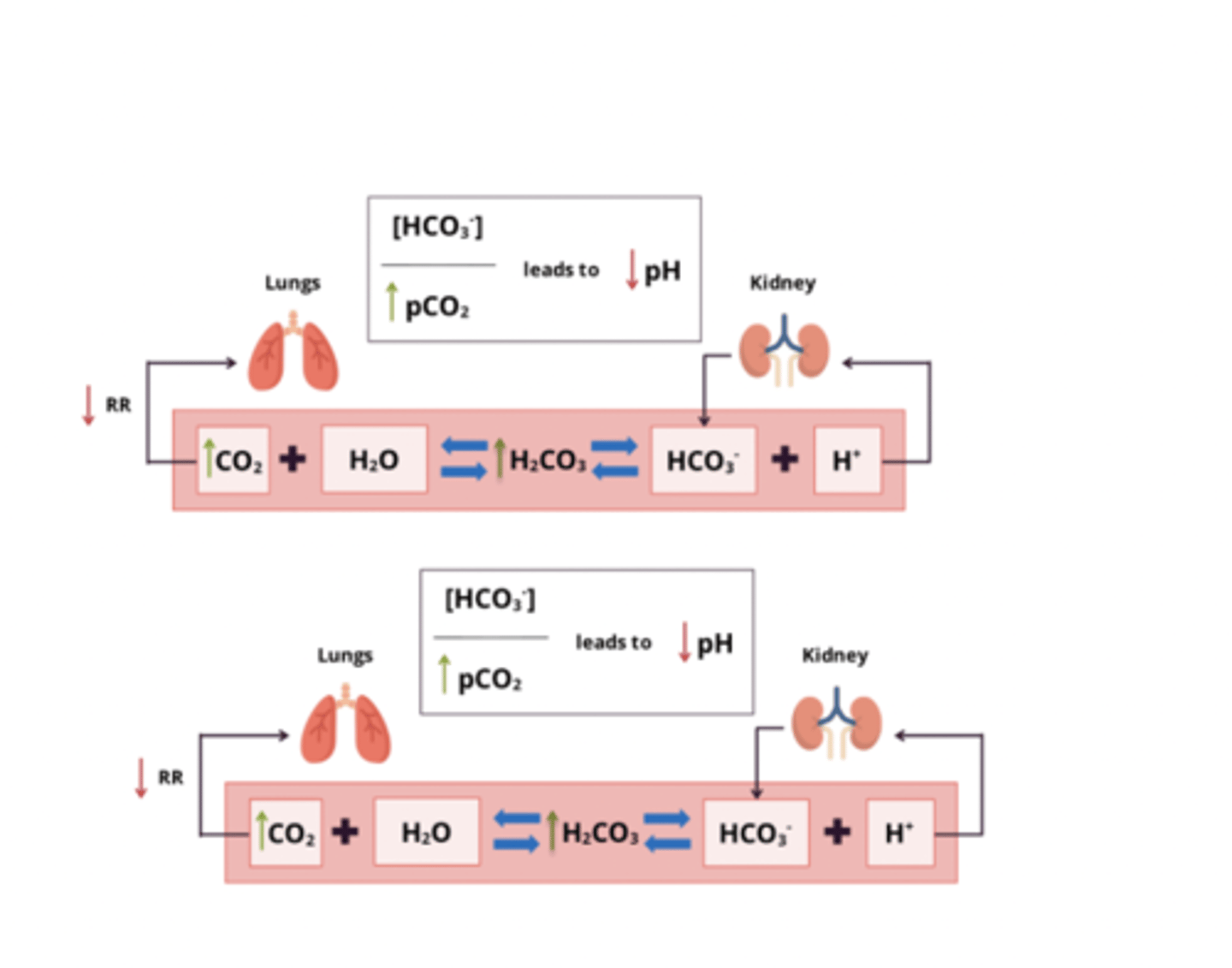
respiratory acidosis: pathology and compensation
pathology:
- due to failure of ventilation → accumulation of CO2 in blood
- primary disturbance: ↑ pCO2 => ↓ HCO3:pCO2 ratio = ↓ pH
Compensation:
- kidneys excrete more H+ (and NH4+) and reabsorb more HCO3-
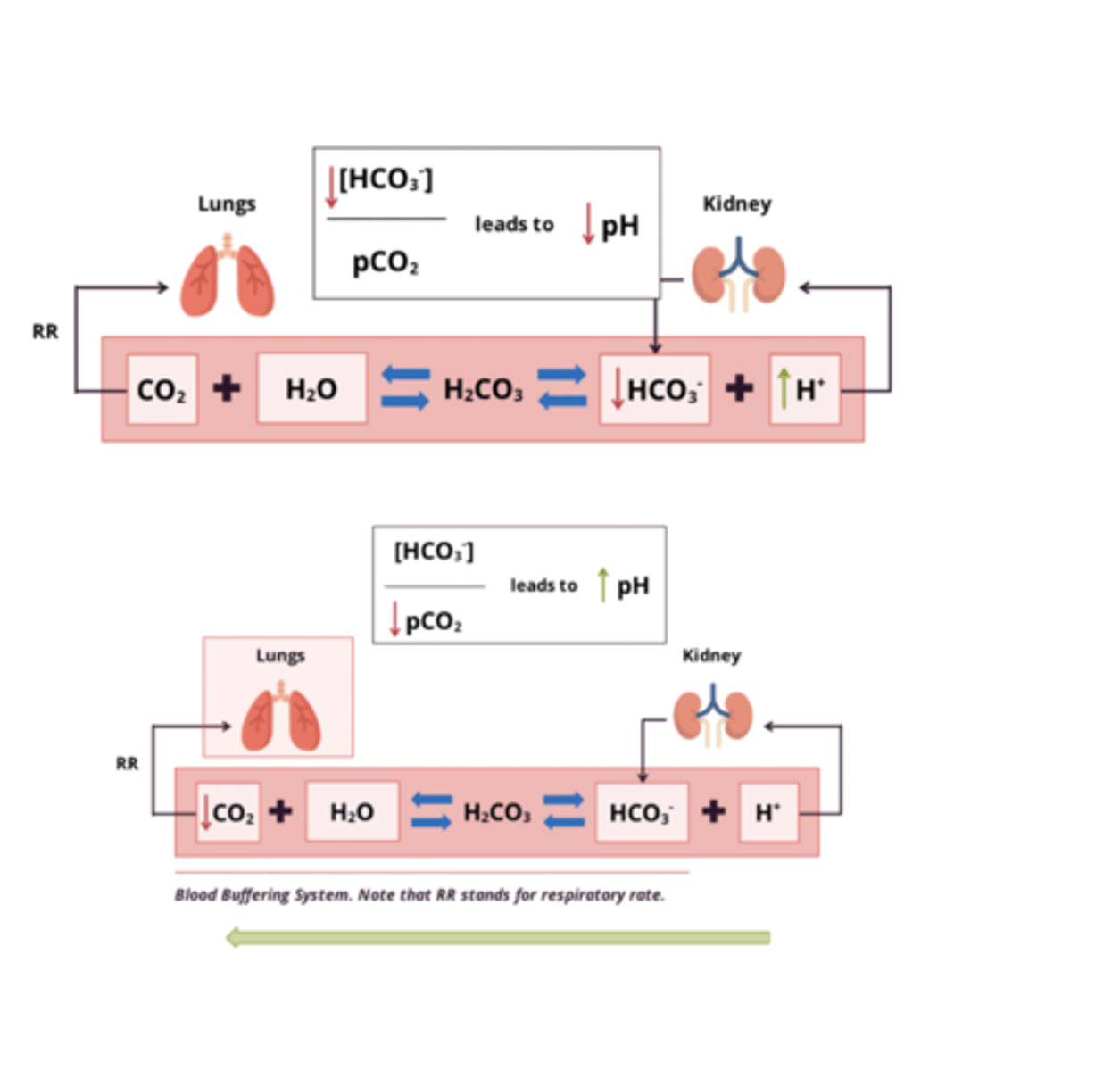
metabolic acidosis: pathology and compensation
pathology:
- increased free H+ in systemic circulation → low serum HCO3- levels (<21 mmol/L)
- ↓ HCO3:pCO2 ratio = ↓ pH
Compensation:
- hyperventilation → expel more CO2 → restore pH
metabolic acidosis: etiology (4)
1. increased acid production
- overproduction of endogenous acid (eg. lactic acid) = ↓ pH
2. decreased acid excretion
- kidney failure = decreased acid excretion = ↓ pH
3. renal GI bicarbonate losses
- diarrhea = increased excretion of renal GI HCO3- = ↓ pH
4. acid ingestion
- ingestion of ethylene glycol (metab to toxic acid) = ↓ pH
evaluation of acid-base disturbances (5)
1. acid-base stats: acidemia or alkalemia?
2. primary disturbance: metabolic, respiratory, or both?
3. calculate range of compensatory responses to determine presence of mixed disorder
4. calculate anion gap for cause of metabolic acidosis
5. do clinical signs align with acid-base analysis?
1. determining acid-base status
pH<7.35: acidemia
pH>7.45: alkalemia
2. nature of primary disturbance
- check pCO2 and HCO3- levels
pCO2: RESPIRATORY
- average pCO2 within 35-45 mmHg
- pCO2 low = alkalosis, high = acidosis
[HCO3-]: METABOLIC
- average [HCO3-] = 22-30 mmol/L
- [HCO3-] low = acidosis, high = alkalosis
example cases:
1. pCO2 = 40 mmHg, [HCO3-] = 10 mmol/L
2. pCO2 = 55 mmHg, [HCO3-] = 25 mmol/L
1. metabolic acidosis (low HCO3-)
2. respiratory acidosis (high pCO2)
mixed acid-base disorder
COPD:
- damage to alveoli and airways = compromised gas exchange, hypercapnia
- altered resp exchange = CO2 retention
- but heart and renal failure associated with COPD → other acid-base disturbances
COPD examples:
1. pCO2 = 80 mmHg, [HCO3-] = 38 mmol/L, pH=7.29
2. pCO2 = 80 mmHg, [HCO3-] = 15 mmol/L, pH=6.88
1. metabolic ALKalosis (high HCO3) AND Respiratory ACidosis (high pCO2)
2. metabolic AND respiratory ACIDosis (low HCO3-, high pCO2)
3. assess appropriateness of compensation
respiratory acidosis:
- defect: ↑ pCO2
- compensation: ↑[HCO3 -] 1 - 3.5mmol/L per 10 mmHg ↑ pCO2
respiratory alkalosis:
- defect: ↓pCO2
- compensation: ↓ [HCO3 -] 2-5 mmol/L per 10 mmHg ↓ pCO2
metabolic acidosis:
- defect: ↓[HCO3 -]
- compensation: ↓pCO2 1.3 mmHg per 1 mmol/L ↓[HCO3 -]
metabolic alkalosis:
- defect: ↑[HCO3 -]
- compensation: ↑ pCO2 0.7 mmHg per 1 mmol/L ↑[HCO3 -]
![<p>respiratory acidosis: </p><p>- defect: ↑ pCO2</p><p>- compensation: ↑[HCO3 -] 1 - 3.5mmol/L per 10 mmHg ↑ pCO2</p><p>respiratory alkalosis:</p><p>- defect: ↓pCO2</p><p>- compensation: ↓ [HCO3 -] 2-5 mmol/L per 10 mmHg ↓ pCO2</p><p>metabolic acidosis: </p><p>- defect: ↓[HCO3 -]</p><p>- compensation: ↓pCO2 1.3 mmHg per 1 mmol/L ↓[HCO3 -]</p><p>metabolic alkalosis:</p><p>- defect: ↑[HCO3 -]</p><p>- compensation: ↑ pCO2 0.7 mmHg per 1 mmol/L ↑[HCO3 -]</p>](https://knowt-user-attachments.s3.amazonaws.com/52cd766d-3d36-4abc-8f2a-88ed050c233f.png)
when assessing compensation, what do inadequate/exaggerated responses indicate?
mixed disorder (another primary acid-base disturbance)
- eg. pH normal, but abnormal pCO2 and/or [HCO3-]
- change in pH greater than could be from one alone
- pCO2 and [HCO3-] change in opposite direction
4. determine presence of anion gap (metabolic acidosis)
- classification of metabolic acidosis based on presence/absence of anion gap
- if gap too big/small → disorder of lungs, kidneys, other organ systems (must identify cause)
- if normal gap but signs of acidosis → non-anion gap metabolic acidosis
anion gap
Anion gap = [Na+] - ([Cl-] + [HCO3-])
normal value: 12
5. evaluating clinical signs (2)
brain to brain loop
1. patient history:
- use history and determine potential causes of acid-base disturb, like vomiting, diarrhea, meds, overdose, chronic conditions predisposed to acidosis (ie. diabetes millitus)
2. physical examination:
- physical exam where symptoms unique to each cause may be present
case study: patient came in with metabolic acidosis. explain results of low pCO2 upon arrival, and then high pCO2 later on.
- low [HCO3-] => compensate via hyperventilation (↓ pCO2)
- hyperventilating too long = fainting = no longer adequately compensating
- limit to lungs ability to compensate = respiratory muscle fatigue
- acidosis worsened = respiratory muscles fatigued = can't sustain hyperventilation = CO₂ retention (↑ pCO2) and therefore acute respiratory acidosis
electrochemical sensors
- measures potential/voltage or current to maintain potential at known values
- quickly measure dissolved o2, co2, ionized Ca, K+ in blood
- eg. ion-selective electrode and oxygen sensors
electrochemical sensors involve
measures potential/voltage or current to maintain potential at known values
electrochemical sensors consist of 2 electrodes:
1. reference electrode:
- commonly Ag/AgCl electrode with saturated KCl in solution as internal electrolyte:
( AgCl <-> Ag + Cl- )
2. indicator electrode:
- has a very thin membrane at bottom that can take up the desired ion species
- membrane in contact with sample and is the source of signal generated by ion-selective electrode
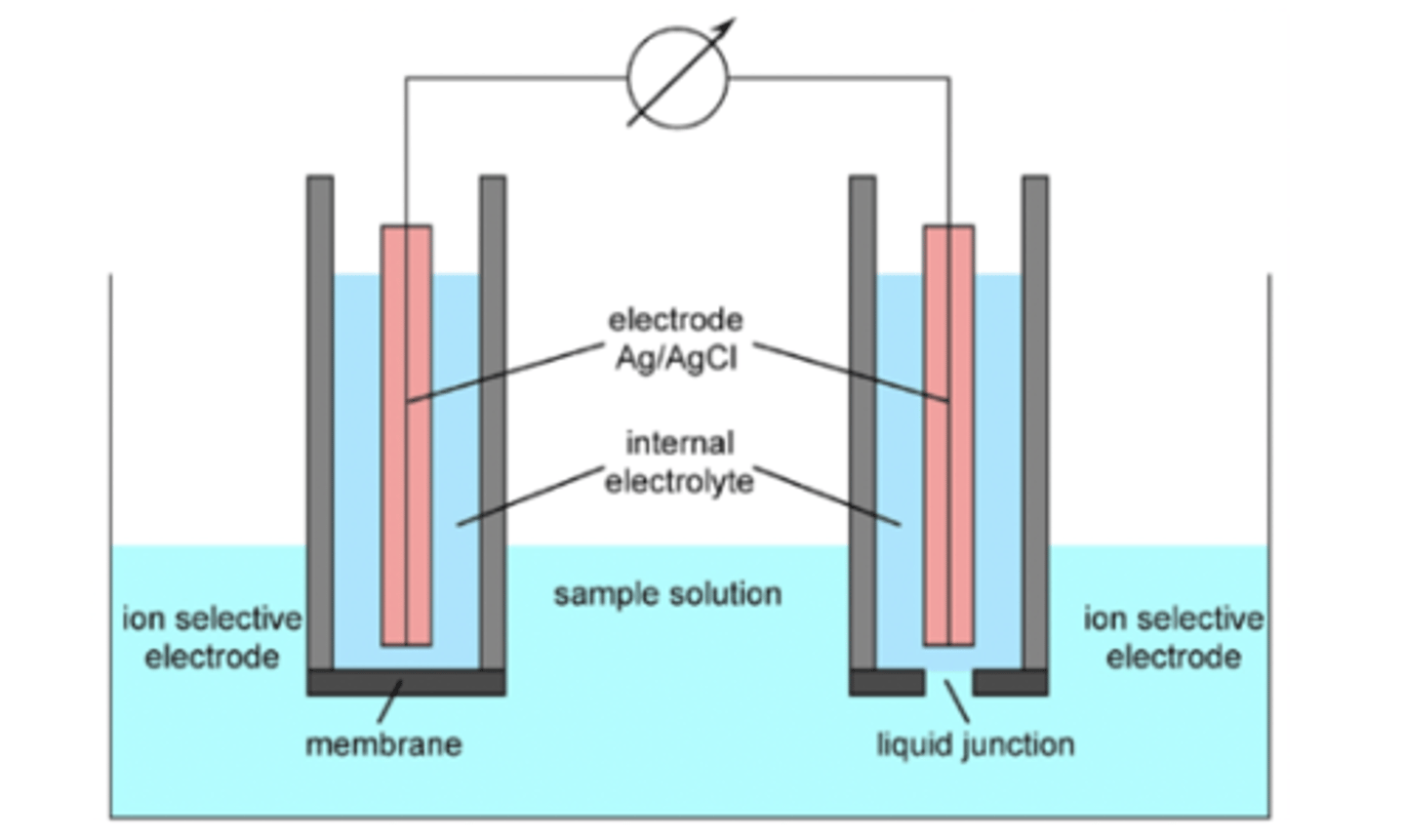
2 types of ion selective electrodes
1. pH electrode
2. polymer membrane ion-selective electrode
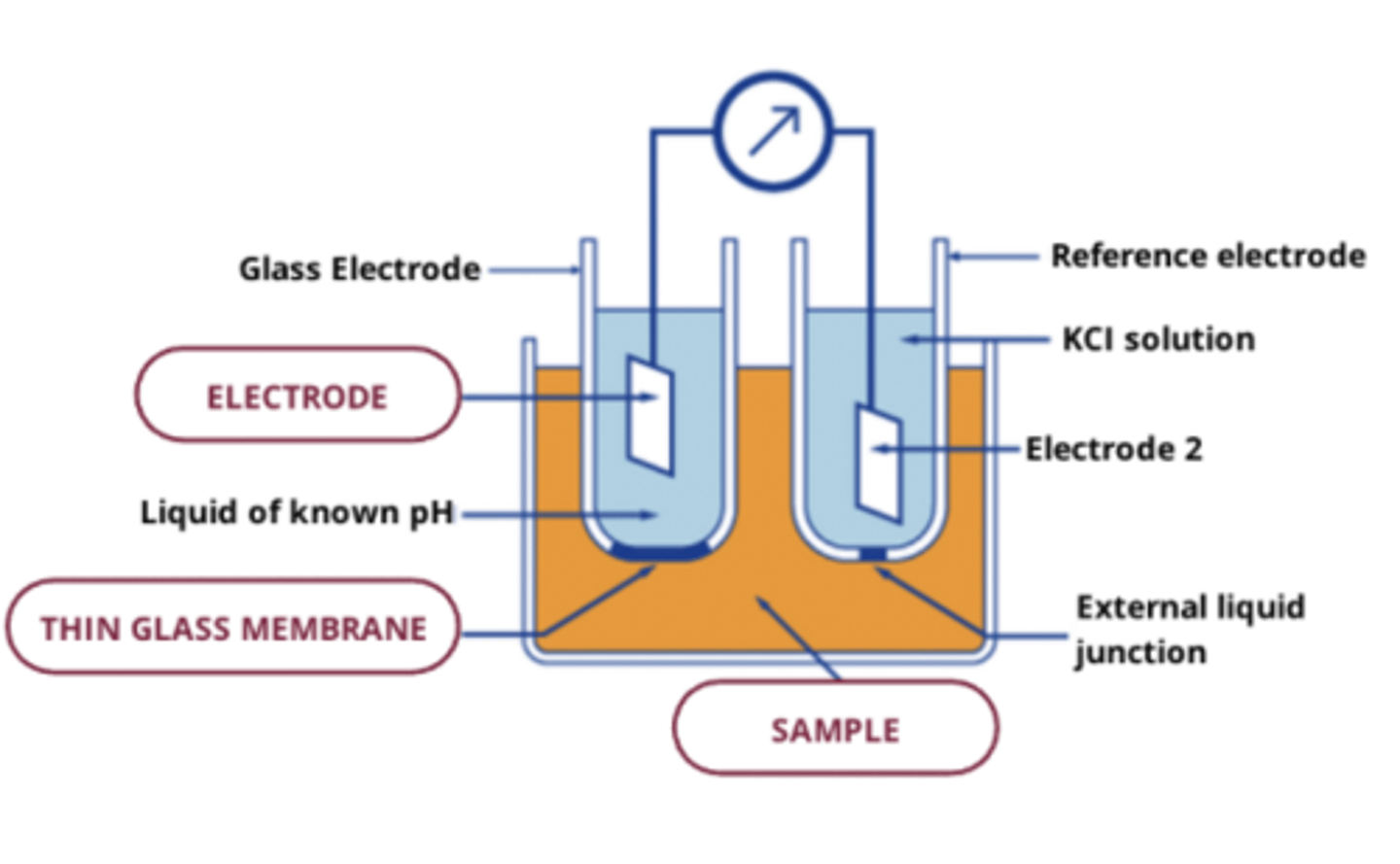
pH electrode
- oldest kind of ion-selective electrode, used for measuring pH
- use thin film of glass => ion exchange between H+ ions in sample and internal electrolyte of known pH in indicator electrode
- commercial pH electrodes are combination where reference electrode built into into sensor
polymer membrane ion-selective electrodes
- detects variable chemical species
- includes analytes,bio/gas ensors, polyemer, ionophores
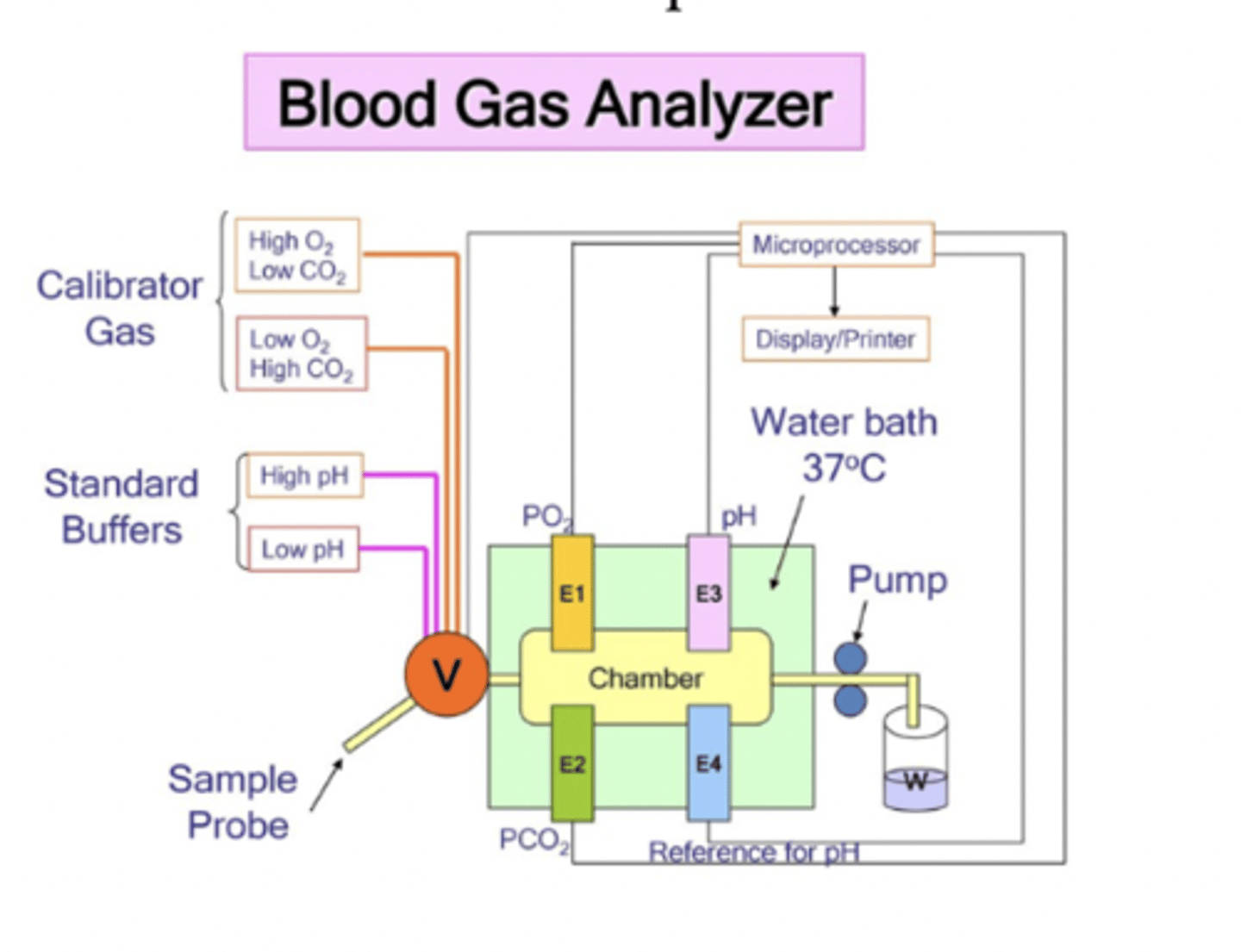
ion-selective electrode: blood-gas analyzer
- detect ions and partial pressure of gases in blood
- arterial blood gas (ABG) analyzer uses combination of ion-selective electrodes and oxygen sensors to measure pO2, pCO2, electrolytes
how do ion-selective electrodes differ from oxygen sensors
ion-selec electrodes: measure potential
oxygen sensors: measure current
oxygen sensors
- measures current
- O2 = reduction = used to measure pO2
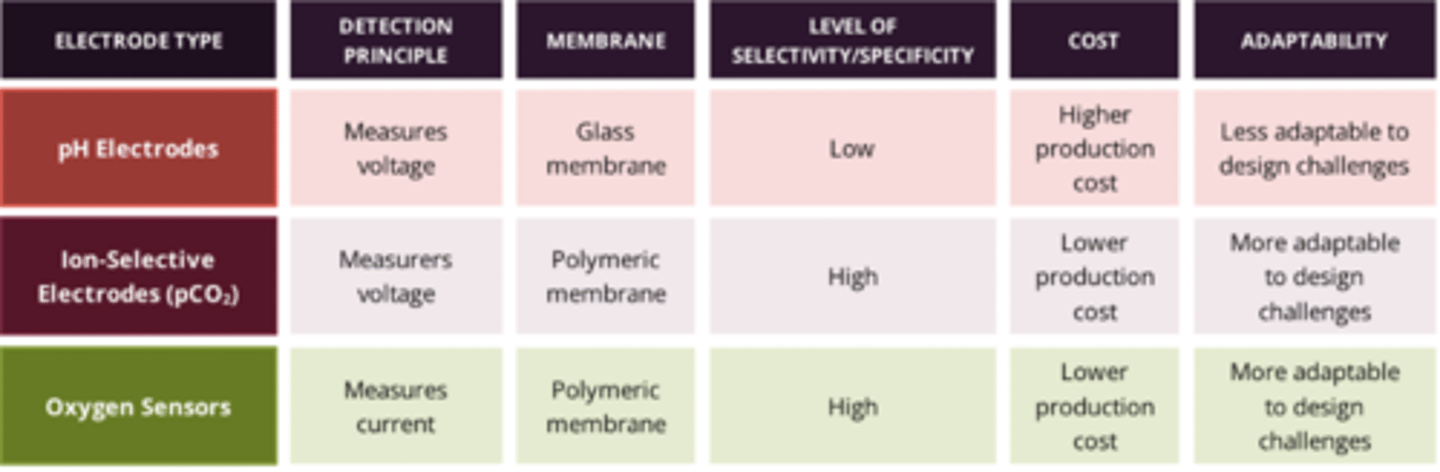
comparison of electrochemical sensors (detection principle, membrane, level of selectivity, cost, adaptability)
pre-analytical challenges in blood gas testing
- different types of blood samples => different results
- arterial samples = high O2, vs venous - low O2
- can be mixed arterial-venous
Arterial Blood Sampling (standard samples, measurement, methods, risks)
standard sample via:
- indwelling arterial catheter
- arterial puncture
measurements:
- blood pH, pCO2, pO2
- along with HCO3 and base = evaluation of acid-base status and ventilation/oxygenation
method:
- arterial catheter = painful, difficult
- so most done via arterial puncture (needle and syringe), usuall in radial artery
risk:
- catheter = invasive = risk of complications: systemic infection, hemorrhage, thrombosis, ischemia
capillaries (pressure and ateriovenous difference)
pressure difference: due to high pressure on arterial side of circ = more arterial than venous blood
ateriovenous difference: pH, pCO2, and pO2 gradient across capillary network due to exchange of O2 and other nutrients (CO2, etc)
capillary blood sampling
- can be taken by skin puncture or using lancet or automated incision device
- least invasive, safest collection technique
- collects small volumes, substitutes for arterial blood
- but clinical values dependent on extent that pH, pCO2, pO2 of capillary blood accurately reflect that of arterial blood
venous blood sampling
- most common type of blood collected
- but less useful than arterial blood for blood gas assessments
- comparable values to arterial except for O2 and CO2
- still invasive but less than arterial
factors affecting blood analyte stability
1. cellular metabolism
2. air, diffusion, evaporation
3. tube storage, position, spinning
blood analyte stability: cellular metabolism
- RBC will continue to metabolize in collected sample
- metab is temp-dep and will DECREASE as temp decreases
- for blood samples testing acid-base disturb, if cells left to sit (and metabolize), they will consume glucose in sample = produce lactic acid = change pH of blood
- needs to be delivered on ice or immediately after collection
blood analyte stability: air, diffusion, evaporation
- pH and conc of blood gases (pCO2, pO2) rapidly change in sample exposed to air, so cannot be exposed to air
- glood gas analysis best done using blood gas syringe (blood collected to sealed syringe, sample injected into blood gas instrument via syringe)
blood analyte stability: tube storage, position, spinning (sample, protocol, consequences)
sample type:
- majority testing on serum sample that was first allowed to clot (30 min) OR a plasma sample that has not clotted
- plasma sample usually preferred, but addition of anticoagulant has consequences/may interfere with method, so not always preferred
tube spinning protocol:
- spinning of sample needs standard protocol
- must ensure separation and minimal lysis of cellular elements from serum/plasma (supernatant)
- once spun, supernatant must be removed ASAP
- challenges with thixotropic component (gel barrier)
tube spinning consequences:
- at same time blood gases tested, often also tested for amount of electrolytes (Na, Cl, K)
- if speed/slow centrifuge to quickly = excess shear force = RBC either break or get release of K+ into serum being tested
impact of preservatives on blood gas lab test results: heparin
- sample should be collected in anticoagulated (plasma state) = ensures sample can be infused to instrument and flow is not interrupted by clots
- done via heparin
- but addition of liquid heparin into tube can affect accuracy for pCO2, pH, and other analytes bc multicharged molecule
- so use balanced heparin
- when heparin balanced with correct amt of cations, used as a anticoag that minimally impacts analysis
Point of Care Testing (POCT)
providing lab testing at patient's location (taking test/instrument to patient)
- eg in situations where time barriers impact delivery of care to patient
examples of POCT devices
- hormone pregnancy test (test hCG)
- blood glucose monitoring (diabetes)
- malaria antigen test
- ultrasound
- blood gas testing through blood gas analyzer
2 challenges with POCT
1. compact size can be limiting
2. fast testing not always better
why is compact size limiting? (3)
taking same method in large analyzers and compact into small portable device with same quality of result
Compromise:
- some compromise dependent on cost/changes in method
-considered in decision touse POCT (should only be used when minimal impact on test)
sensitivity and specificity:
- often require simplifying to fit into compact device, may not be as specific/sensitive as lab results
limited capacity:
- limited range of results, so likely need to follow up with lab test after initial POCT
why is fast testing with POCT not always better? (4)
rate limiting factors:
- lab tests not usually rate limiting factor in ED
- usually analyzing/interpreting lab results
routine verification:
- POCT needs routine verification and monitoring (labour intensive, time consuming)
test selection:
- must consider which POCT is most relevent/accurate
specific benefits:
- may only decrease length of stay for small subset of patients
more considerations of POCT
- not error-proof
- patients must carefully follow test directions and be familiar with test system
- inconsistent POCT techniques => inconsistent results
- if not performed properly, can = serious consequences
- when not consistently documented/managed = not incorp into health records = clinician cannot monitor health changes
hospital-grade POCT
- big difference between patient- vs hospital-grade POCT device
- hospital-grade uses quality framework for clinical lab testing
- consistency in use and quality metrics
pros of POCT
1. accessibility
- provides test results in locations without clinical labs (rural, developing worlds, airplanes, etc)
- accessibile, rapid screen for infectious diseases
- useful in natural disaster
- eliminates concern ppl won't return for treatment if they have to go home and wait for test results
2. efficiency:
- gets rid of ER overcrowding by reducing time takes to treat = useful in ER/operating room
- eg. if someone having stroke, must quickly measure prothrombin before stroke meds
- faster treatment for infectious disease=prevent spread (eg. rapid COVID tests)
futue of POCT
- may include testing for outbreaks, pandemics, endocrine testing for surgical therapy, sepsis testing, stroke markers, DNA testing
- will co-exist with central lab testing
- there will always be a cost in providing POCT (financial cost and accuracy)
Contact Lenses POCT
- new contacts to measure/monitor glucose in people with diabetes
- biochemical tear film sensing tech used to detect and monitor eye diseases (tears contain wide range of biomarkers)
- hope that they can eventually diagnose other diseases like cancer, hypertension, Alzheimer's
limitations of contact lenses for POCT
- tears may not have enough info for useful diabetes monitoring
- concern bc lack of consistency between blood and vitreous levels (in eye)
- no study provided treatment goal for use of vitreous glucose and its relation to blood glucose
- biochemical sensors in lenses don't match accuracy of other analysis tools