Acids and bases
1/74
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
What is a Bronsted-Lowry acid?
A proton donor.
What is a Bronsted-Lowry base?
A proton acceptor.
H3O+ What is the name of this ion?
Oxonium ion.
True or false? H+ ions are protons.
True.
True or false? Water is fully dissociated.
False. WATER IS SLIGHTLY DISSOCIATED.
Give the symbol equation showing that water is slightly dissociated.
H2O (l) →← H+ (aq) + OH- (aq)
OR
H2O (l) + H2O (l) →← H3O+ (aq) + OH- (aq)
What is Kw?
The ionic product of water.
Give the Kw expression.
Kw = [H+] [OH-]
True or false? In pure water [H+] = [OH-] so Kw = [H+]2
True.
What is the value of Kw at 298K?
1 × 10-14 mol2 dm-6
Give the expression for calculating pH.
pH = -log [H+]
True or false? The higher the pH, the higher the concentration of H+ ions.
False. THE LOWER THE pH, THE HIGHER THE CONCENTRATION OF H+ IONS.
Why is a logarithmic scale used for pH?
The concentrations of hydrogen ions in aqueous solutions covers a very wide range.
True or false? A difference of 1 in pH means a tenfold difference in [H+].
True.
Give the expression for calculating [H+] from pH.
[H+] = 10-pH
Talk me through the steps of calculating [OH-] when you know pH.
1) Calculate [H+] using this expression, [H+] = 10-pH
2) Rearrange the expression for Kw (assumed to be 1 × 10-14 mol2 dm-6) so that [OH-] is the subject, [OH-] = Kw / [H+].
3) Put the values you know into the expression to get [OH-].
What is a strong acid?
An acid that fully dissociates when in an aqueous solution.
True or false? In strong acids [H+] = concentration of strong acid.
True.
Why can you assume that [H+] = concentration of strong acid in a strong acid?
Strong acids dissociate fully.
Talk me through how to work out the pH of a strong acid when you know it’s concentration.
1) The concentration of the strong acid is equal to the concentration of H+ ions.
2) Put the value for [H+] into the pH expression, pH = -log [H+], this gives the pH value.
What is a strong base?
A base that fully dissociates when in an aqueous solution.
True or false? In a strong base [OH-] = concentration of strong base.
True.
Talk me through how to calculate the pH of a strong base.
1) Know that the concentration of the strong base is equal to [OH-].
2) Rearrange the Kw expression so that [H+] is the subject, [H+] = Kw / [OH-].
3) Put your known values into the rearranged expression, you will get the [H+] value.
4) Put the [H+] value into the pH expression, you will get the pH value.
Give three examples of strong acids.
Hydrochloric acid (HCl), sulfuric acid (H2SO4), nitric acid (HNO3).
Give a general example of a weak acid.
Carboxylic acids.
Give two examples of strong bases.
Sodium hydroxide (NaOH), potassium hydroxide (KOH).
Give an example of a weak base.
Ammonia (NH3).
What is a weak acid?
An acid that only slightly dissociates in an aqueous solution.
What is a weak base?
A base that only slightly dissociates in an aqueous solution.
True or false? Weak acids and weak bases exist in an equilibrium when each in an aqueous solution.
True.
What is Ka?
The acid dissociation constant.
Give the general symbol equation for the dissociation of a weak acid.
HA (aq) →← H+(aq) + A- (aq)
How do you work out the units for Ka?
The same way you work out the units for Kc.
Talk me through how to work out the pH of a weak acid when you know it’s concentration and Ka.
1) Rearrange the Ka expression so that [H+]2 = Ka x [HA].
2) Square root the value of the answer to (1) to get [H+].
3) Use pH = -log [H+] to calculate pH, this is the pH.
Give the Ka expression.
Ka = [H+] [A-] / [HA]
OR
Ka = [H+]2 / [HA]
Why can you assume that [H+] = [A-] in a weak, monoprotic, pure acid?
Each acid molecule that dissociates dissociates into one H+ ion and one A- ion, so [H+] = [A-].
What is a monoprotic acid?
An acid that can donate one proton per molecule to an aqueous solution.
What is a diprotic acid?
An acid that can donate two protons per molecule to an aqueous solution.
Give the pKa expression.
pKa = -log Ka (same relationship as pH and [H+]).
What is the general concept of a titration?
Finding the unknown concentration of a solution, of which you know it’s volume, by adding it to another solution of which you know the volume and concentration.
What is the general concept of an acid-base titration?
Adding an acid to a base (or vice versa) until the indicator in the solution with the known concentration changes colour (showing that the acid and base have been neutralised).
Sketch a strong base - strong acid titration curve.
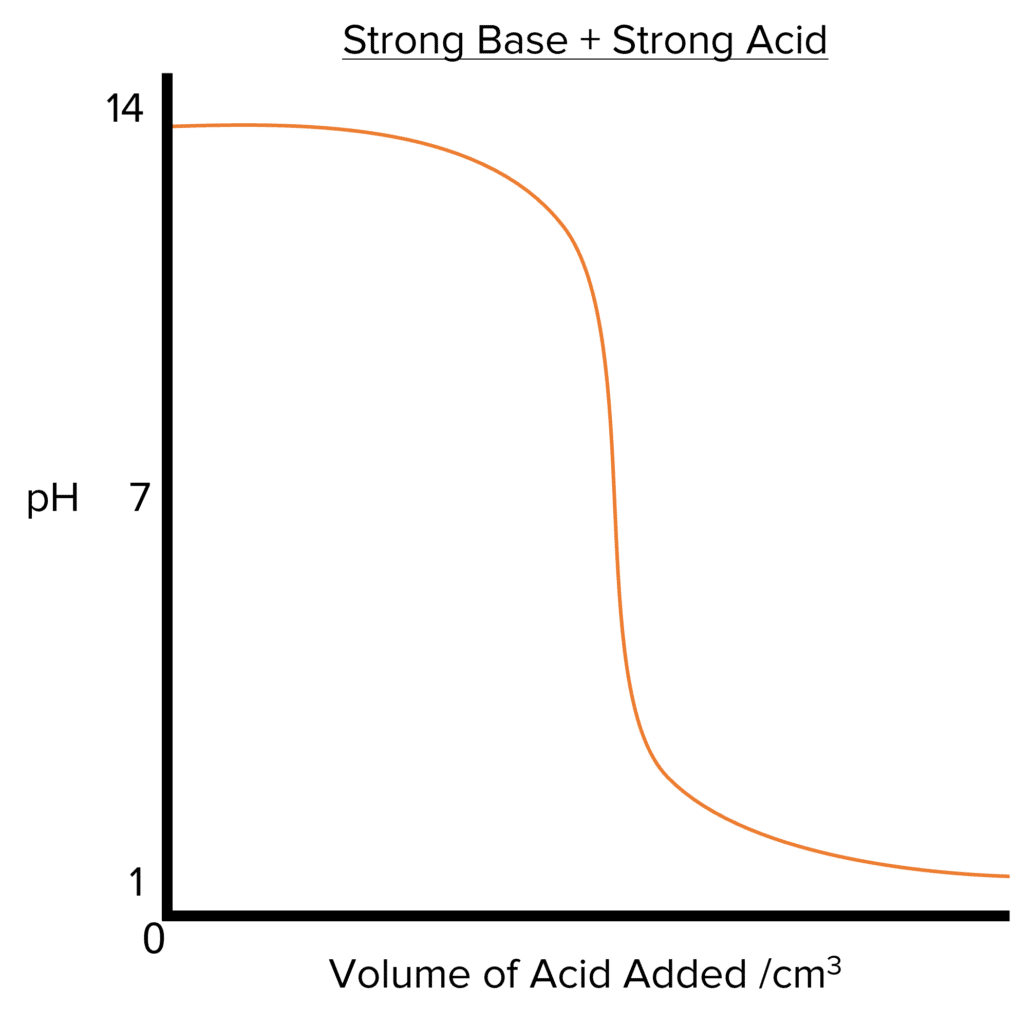
Sketch a strong acid - weak base titration curve
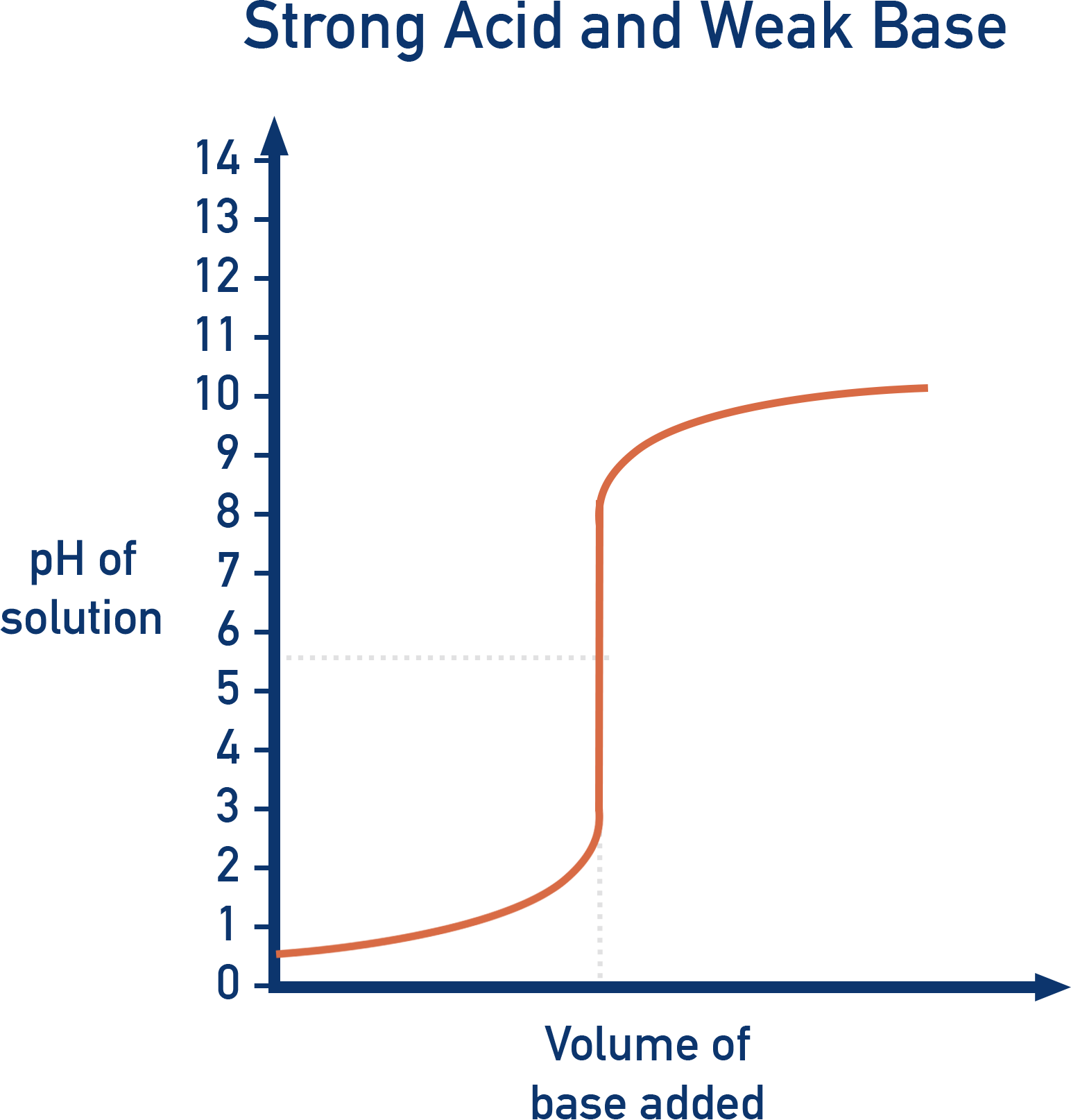
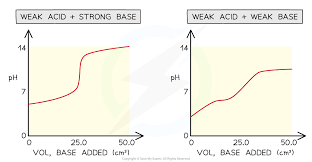
Sketch a weak acid - strong base titration curve. Then, sketch a weak acid - weak base titration curve.
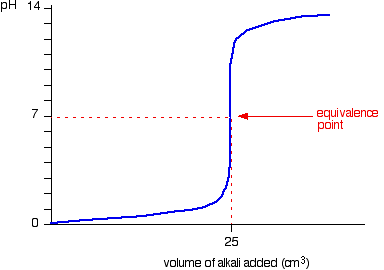
What is the equivalence point?
The point at which sufficient acid has been added to the base to just neutralise it (or vice versa).
Label the equivalence point on a generic titration curve.
True or false? The change in pH is smallest when a weak acid is reacting with a strong base.
False. THE CHANGE IN pH IS SMALLEST WHEN A WEAK ACID IS REACTING WITH A WEAK BASE.
Where is the half-neutralisation point on a titration curve?
The point halfway between zero and the equivalence point.
What does the half-neutralisation point mean in terms of H+ ions?
When half of the H+ ions have reacted with A- forming HA.
True or false? At the equivalence point all the H+ ions from the acid have reacted with the OH- ions from the base.
True.
True or false? At the half-neutralisation point [HA] = [A-]
True.
Explain why [HA] = [A-] at the half-neutralisation point.
If half of the H+ ions have reacted with A- to form HA, that means that half of the solution if HA. The other half of the solution contains A-, they are halves so they are equal concentrations so [HA] = [A-].
Explain (with equations) why Ka = [H+] at the half-neutralisation point.
Ka = [H+] [A-] / [HA] normally, at the half neutralisation point [HA] = [A-] meaning they are both cancelled out in the Ka expression leaving Ka = [H+].
Explain (with equations) how pH = pKa at the half-neutralisation point.
At the half neutralisation point [HA] = [A-] so Ka = [H+], so -log Ka = -log [H+], so pH = pKa.
What is the equivalence point in terms of hydrogen ions and hydroxide ions?
The volume at which the same number of moles of hydrogen ions has been added as the number of hydroxide ions in the solution (or vice versa).
Give three things you must find when choosing an indicator.
Sharp colour change (enough for one drop of acid/base to change it’s colour).
The colour change taking place at a range of pH that are involved with the equivalence point of the titration.
Distinct colour change.
True or false? All indicators are suitable for all acid-base titrations.
False. NOT EVERY INDICATOR IS SUITABLE FOR EVERY ACID-BASE TITRATION.
What is a buffer solution?
A solution that can resist pH changes when small amounts of acid or alkali are added to them.
In general, how does a buffer work?
They are based on an equilibrium that will shift to oppose any changes made in the H+ ion or OH- ion concentration.
What makes up an acidic buffer?
A weak acid and it’s salt.
Give a symbol equation to show the equilibrium with the weak acid in an acidic buffer. In what position does equilibrium lie in this reaction and why? What does this mean about the concentration of the components of this reaction?
HA →← H+ + A-
Equilibrium lies to the left because weak acids dissociate weakly in solution.
Higher concentration of HA, lower concentration of H+, lower concentration of A-.
Give a symbol equation to show the equilibrium with the salt of the weak acid in an acidic buffer. In what position does equilibrium lie in this reaction and why? What does this mean about the concentrations of the components of this reaction?
A-X+ →← A- + X+
Equilibrium lies to the right because salts dissociate strongly.
Lower concentration of A-X+, higher concentration of A-, higher concentration of X+.
Talk me through what happens when you add H+ ions to an acidic buffer.
1) The extra H+ ions react with the A- ions (that are in a high concentration from the dissociation of the salt) in the solution.
2) This causes more HA to be produced meaning equilibrium has shifted left (in the equation for the weak acid dissociation).
Talk me through what happens (with an equation) when OH- ions are added to an acidic buffer solution.
1) The added OH- ions react with HA to form water and A- , which decreases the concentration of OH- ions.
HA + OH- →← H2O + A-
2) Equilibrium shifts right to oppose the change and produce more H+ ions.
OR (another way of explaining it in terms of H+ ions)
1) The OH- ions react with H+ ions forming water.
2) This decreases the H+ ion concentration.
3) This causes equilibrium in the weak acid reaction to shift right to produce more H+ ions (change counteracted).
What are basic buffers made up of?
A weak base and it’s salt.
Give an example of a basic buffer.
Ammonia (NH3) (weak base), ammonium chloride (NH4 + Cl-) (salt of weak base).
True or false? The two equilibrium equations in a buffer coexist in the same beaker.
True.
Using ammonia and ammonium chloride buffer, give the symbol equation to show the equilibrium established with ammonia. What are the concentrations of the components of this equation? Why are the concentrations like this?
NH3 + H2O →← NH4+ + OH-
Higher concentration of ammonia, lower concentration of ammonium ions, lower concentration of hydroxide ions.
Ammonia is a weak base so weakly dissociates meaning equilibrium lies far to the left here.
Using ammonia and ammonium chloride buffer, give the symbol equation to show the equilibrium established with ammonium chloride. What are the concentrations of the components of this equation? Why are the concentrations like this?
NH4+Cl- →← NH4+ + Cl-
Lower concentration of ammonium chloride, higher concentration of ammonium ions, higher concentration of chloride ions.
Salts dissociate strongly so equilibrium lies far to the right.
Talk me through what happens when a base is added to an ammonia and ammonium chloride buffer.
1) The OH- ions from the base react with the NH4+ ions in the solution (ammonium ions in high concentration from strong dissociation of salt).
2) This produces NH3 and H2O so equilibrium has shifted left (change counteracted).
Talk me through what happens when an acid is added to an ammonia and ammonium chloride buffer.
1) H+ ions from the acid react with OH- ions from the weak base dissociation (not in high concentration because weak bases weakly dissociate).
2) High concentration of NH3 and H2O produced counteracts this decrease in OH- ions and replaces them, equilibrium has shifted right (change counteracted).
Talk me through how to calculate the pH change when an acid is added to an acidic buffer when you know: the concentration of the buffer solution, the volume of the weak acid in the buffer, the concentration of salt in the buffer, the volume of acid added, the concentration of acid added, Ka.
1) All the H+ ions from the added acid will react with the A- ions from the salt (that are in high concentration as salts dissociate strongly).
2) Calculate the number of moles of weak acid in the buffer, n = cv.
3) Calculate the number of moles of salt in the buffer, n = cv.
4) Calculate the number of moles of acid added, n = cv.
5) Add the moles of weak acid to the moles of added acid to get the new total moles of acid.
6) Subtract the moles of added acid from the moles of salt in the buffer to get the new total moles of salt.
7) Add up all the volume values to get the new volume of the mixture.
8) Work out the new concentration of acid ([HA]) with the new moles of acid and the new volume, n = cv.
9) Work out the new concentration of salt ([A-]) with the new moles of salt and the new volume, n = cv.
10) Rearrange the Ka expression so that [H+] is the subject.
11) Calculate [H+] using the rearranged Ka expression.
12) Put your [H+] value into the expression pH = -log [H+], this gives your new pH value.
Give two equations you can use to work out the pH of an acidic buffer solution.
1) [H+] = Ka x (moles of acid/moles of salt)
OR [H+] = Ka x (concentration of acid/concentration of salt) (only if there has been no addition of anything else to the buffer)
2) pH = -log [H+]
Talk me through how to calculate the pH of the solution formed when a strong acid is mixed with a strong base.
(What you know: volume of strong acid, concentration of strong acid, volume of strong base, concentration of strong base, Kw)
1) Know that initially [OH-] is the same as the concentration of the strong base and that [H+] is the same as the concentration of the strong acid.
2) Work out the volume of the solution formed by adding together the volume of the strong acid and the volume of the strong base.
3) Work out the moles of OH- and the moles of H+ with n = cv.
4) Work out how many moles of either OH- or H+ are in excess by subtracting the smaller mole value from the larger mole value.
(5) IF EXCESS MOLES OF H+
1) Use c = n / v to work out [H+] from the excess moles of H+.
2) Use pH = -log [H+] to get pH).
(5) IF EXCESS MOLES OF OH-
1) Use c = n / v to work out [OH-] from the excess moles of OH-.
1) Use [H+] = Kw / [OH-] to get [H+].
2) Use pH = -log [H+] to get pH).
What equation do you use to work out the concentration of something when you know it’s solubility and the volume?
c = m / V
(c = concentration, m = solubility, V = volume)
(treat solubility like mass).