Diagram of Y10 Periodic Table VMA | Quizlet
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
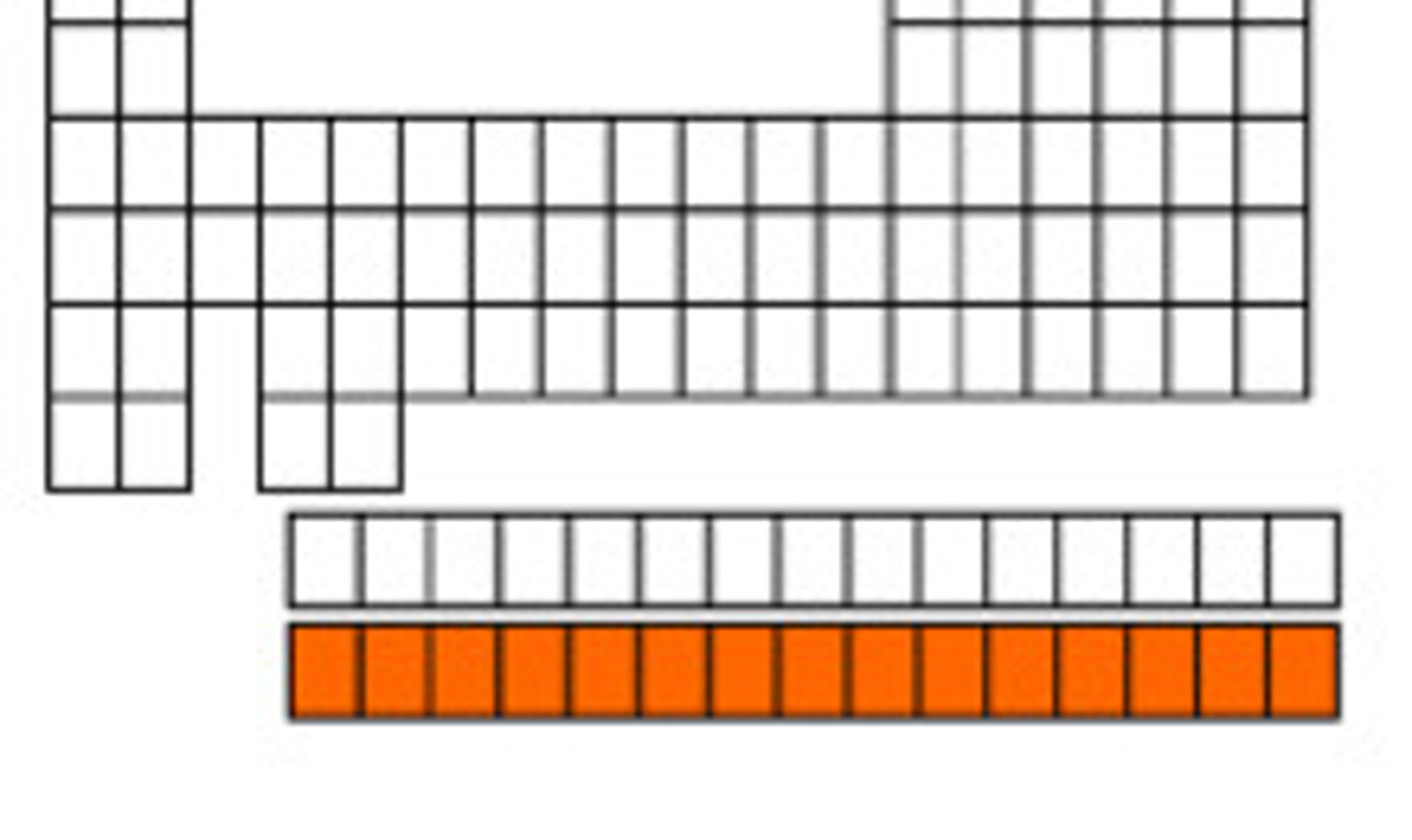
It is a table in which elements are arranged into a series of rows and columns that obey specific patterns.
The original design of the elements is credited to Dmitri Mendeleev in the 1800s.
Periodic table of elements
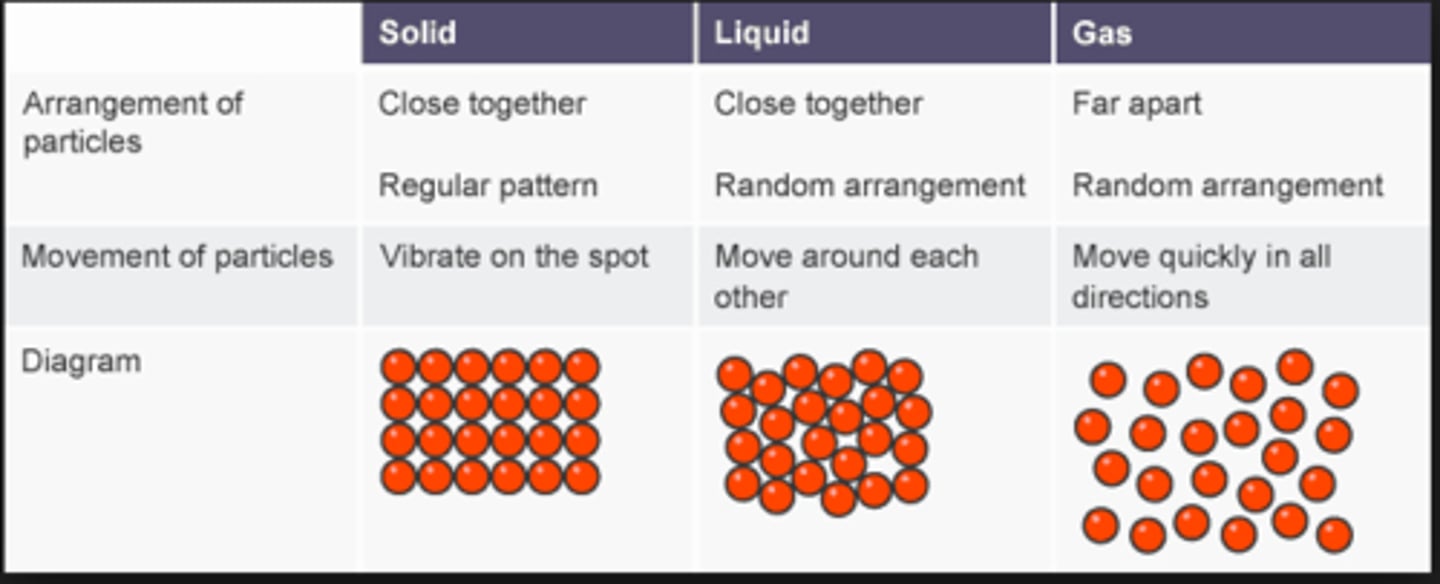
An illustration of the different states of matter like solids, liquids and gases.
particle diagram
A molecule composed of one kind of atom; cannot be broken into simpler units by chemical reactions.
Elements

Groups of two or more atoms held together by chemical bonds
Molecules
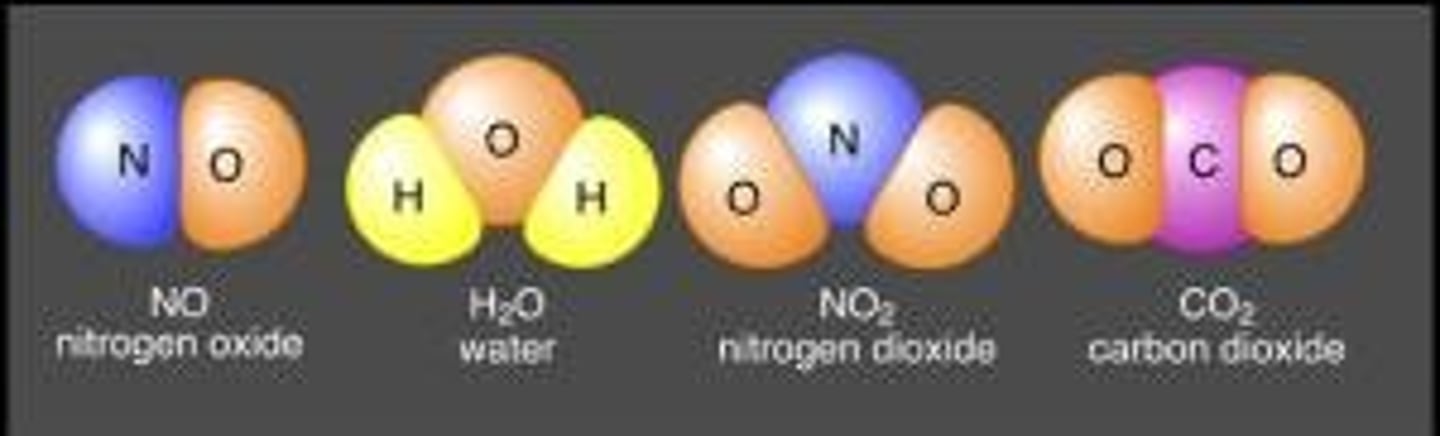
A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds.
compounds
The property that describes how readily a substance combines chemically with other substances
Reactivity
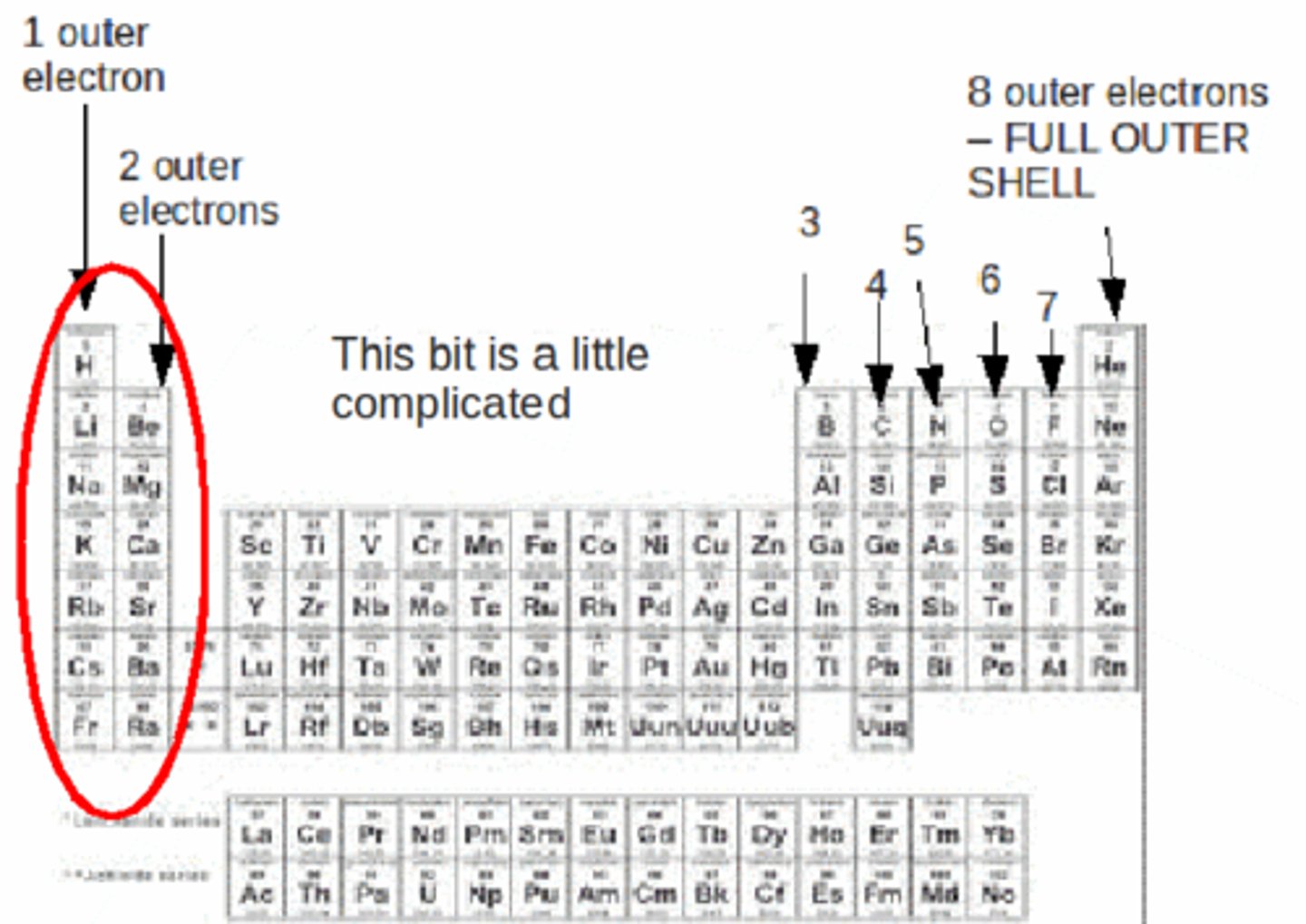
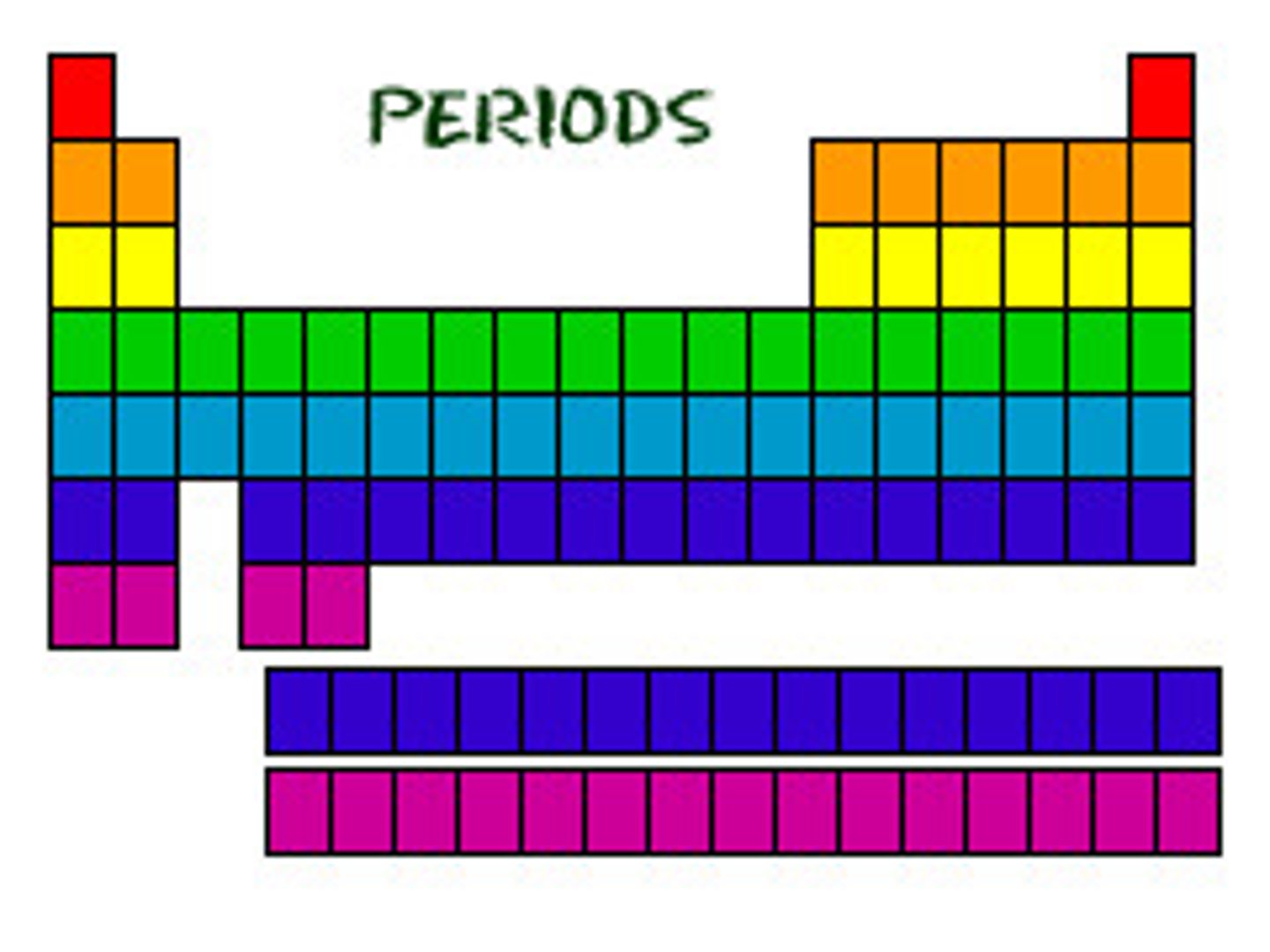
The horizontal rows on the periodic table.
Elements in the same row have the same number of electron shells/energy levels.
Period
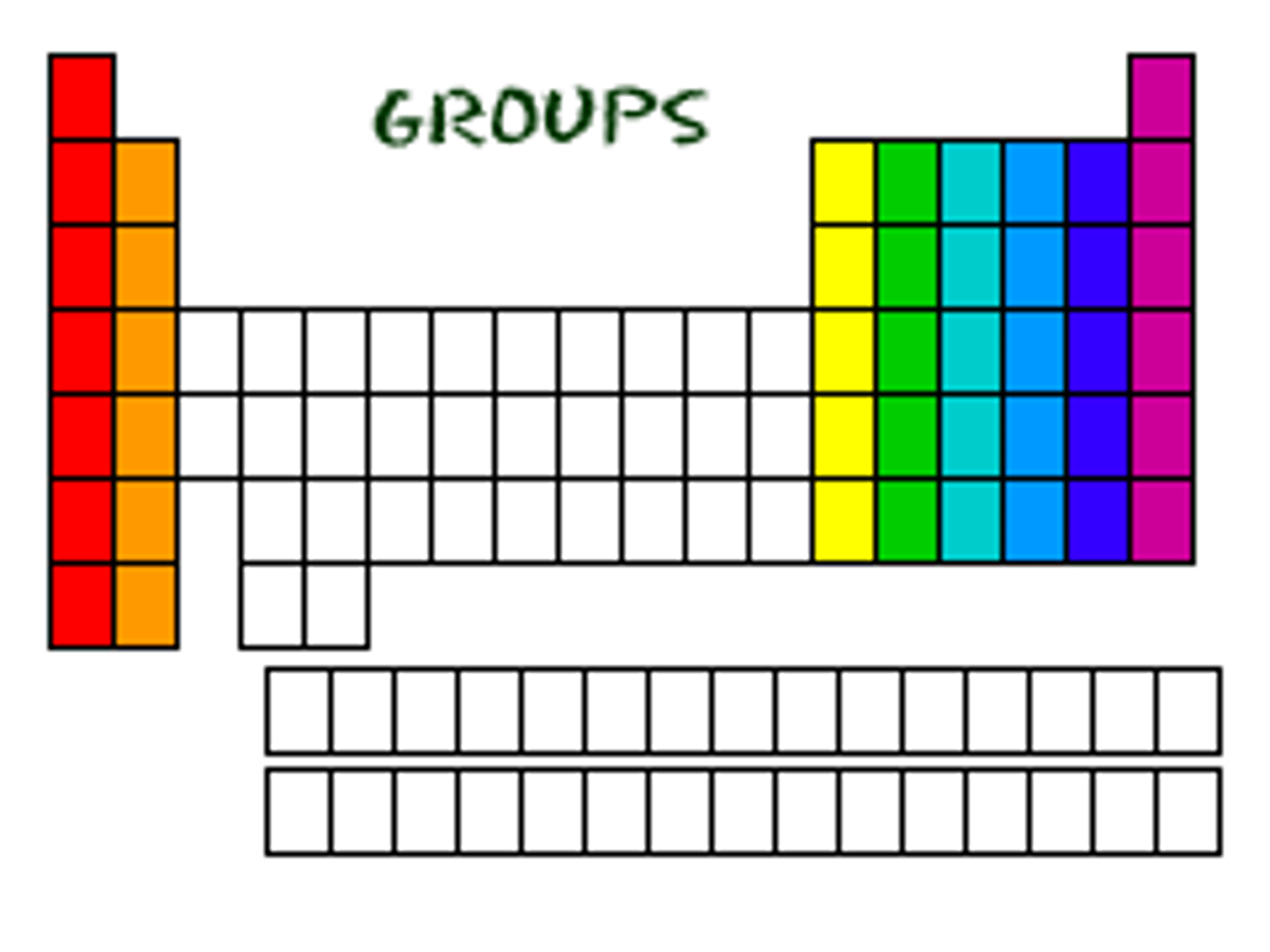
The vertical columns on the periodic table.
Elements in the same group/family have the same number of valence electrons. (Except Helium which is in Group 8, but only has 2 valence e-)
Group
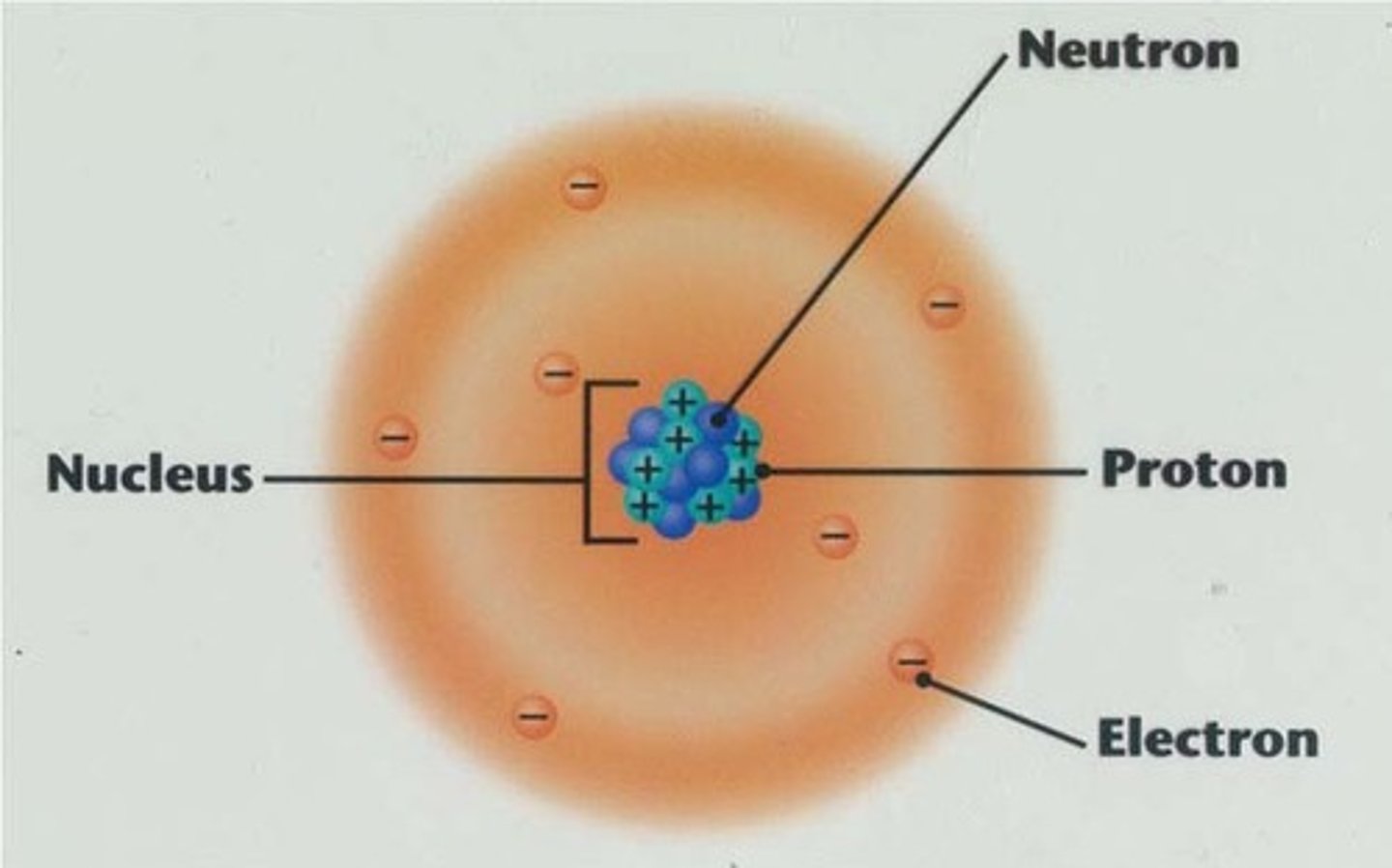
Number of protons and neutrons
Atomic mass
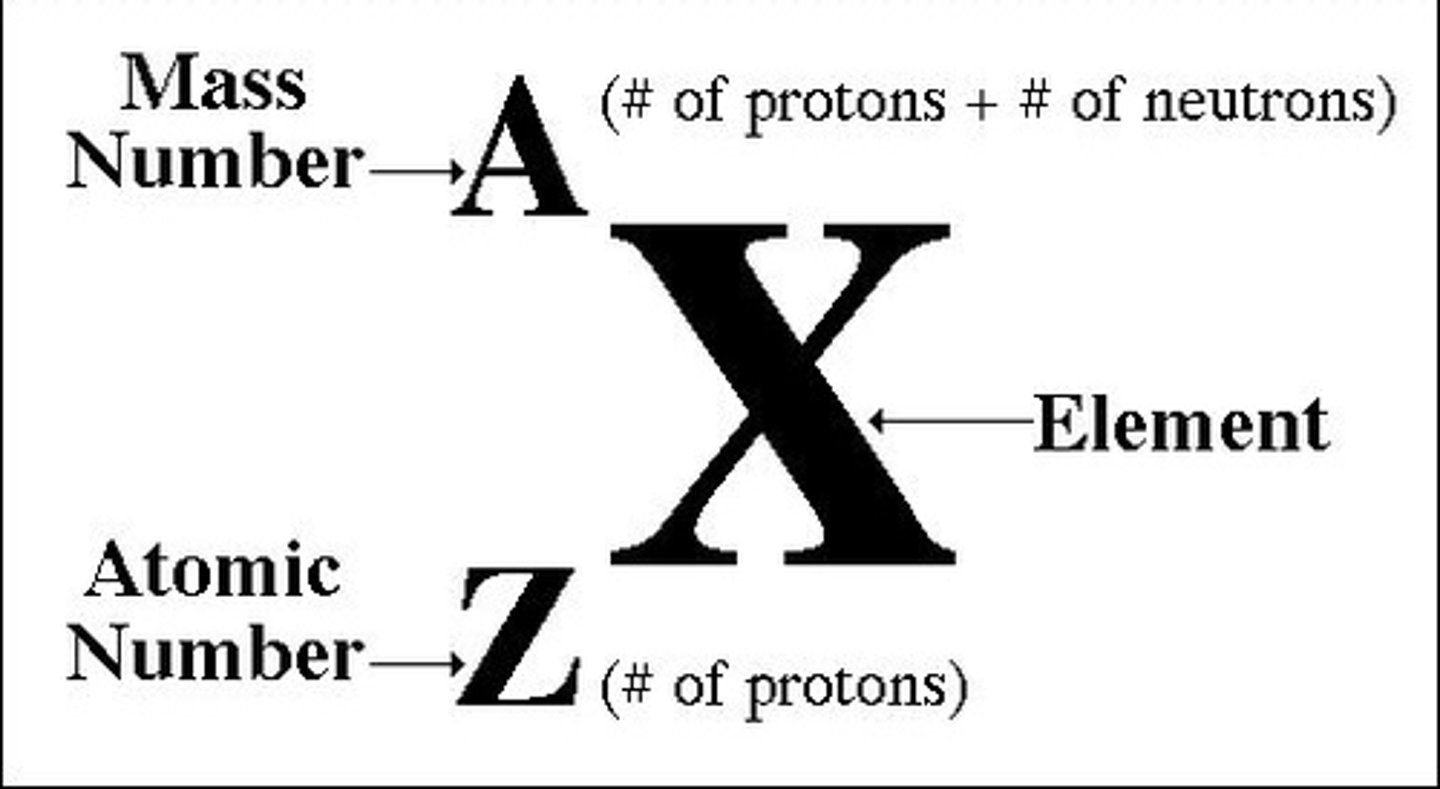
The number of protons in the nucleus of an atom
Atomic number
positively charged particles in the nucleus of an atom
protons

the particles of the nucleus that have no charge
neutrons

negatively charged particles found outside the nucleus
electrons
regions around the nucleus in which given electron or electron pair is likely to be found
orbitals
TERM
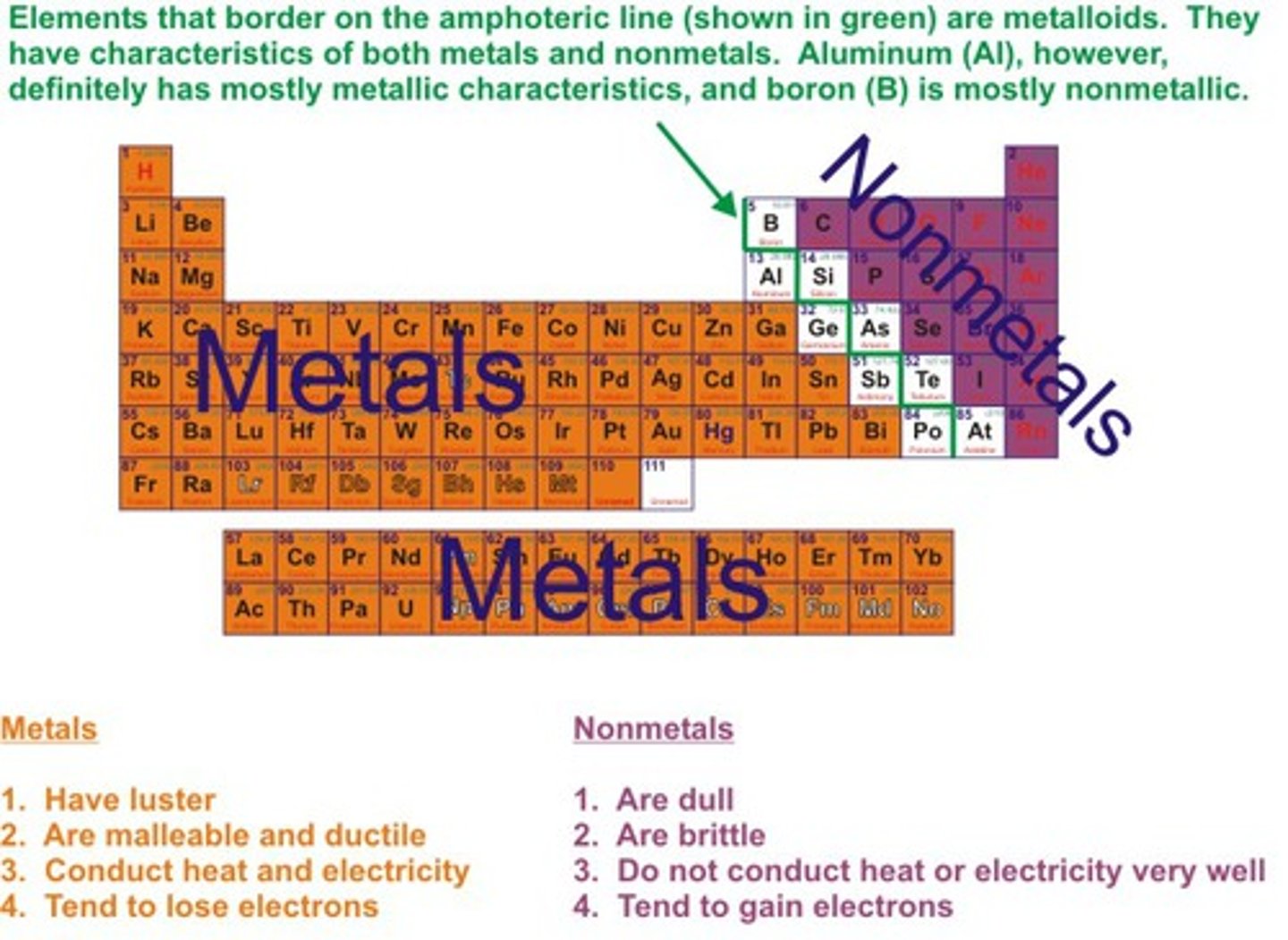
A fundamental grouping of elements such as sodium, gold, copper, and iron in the periodic table that have similar chemical and physical properties, (like Malleable,Ductile, Metallic luster, Solid at room temperature, are good conductors of electric current and heat.)
DEFINITION
Metals
Capable of being shaped.
Easy to shape or bend.
Malleable

Describes a material that can be pulled out into a long wire.
Ductile

TERM
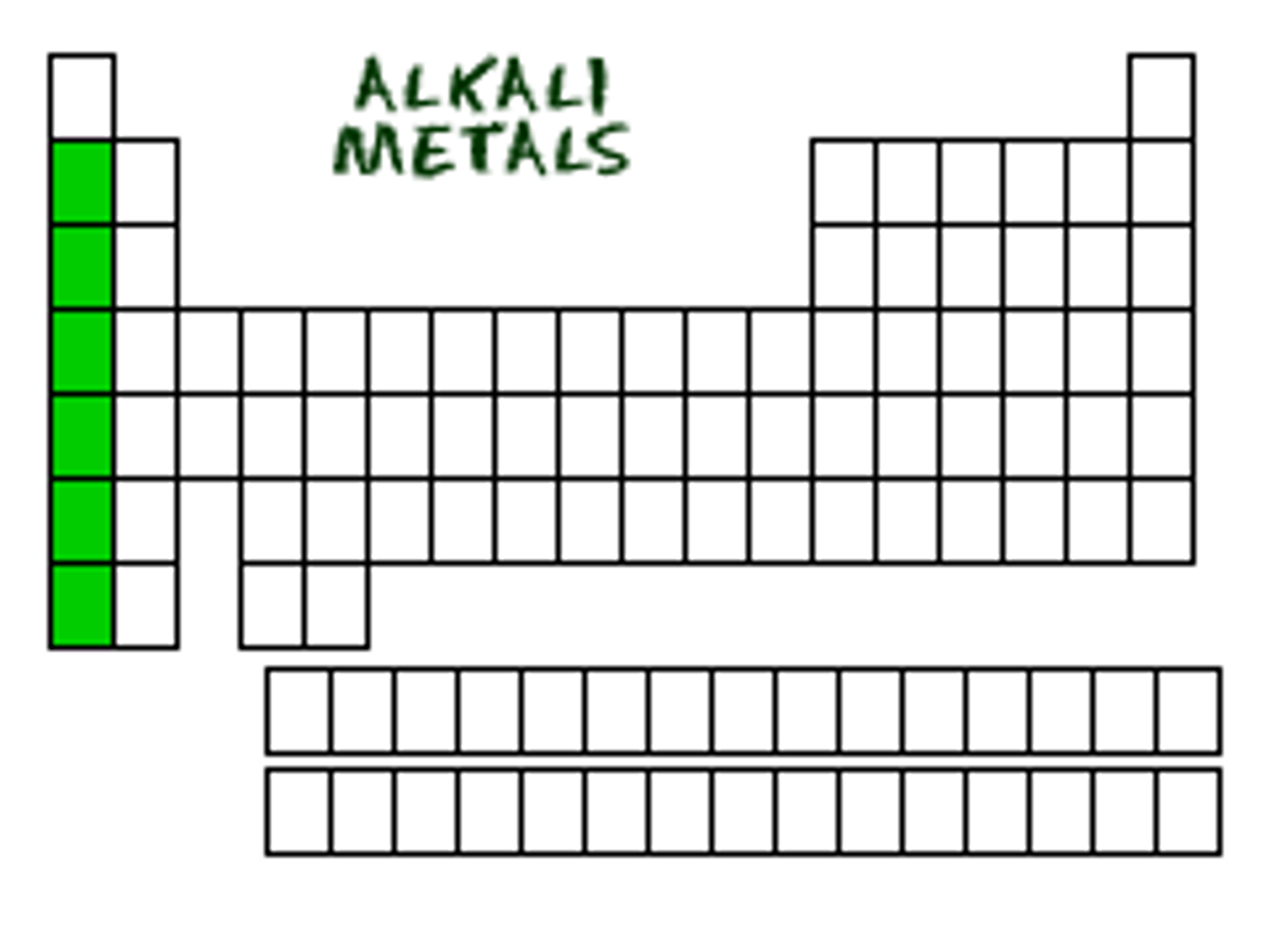
The elements in Group 1A of the periodic table (except Hydrogen).
VIOLENTLY REACT with water, and produce alkaline (basic) substance.
DEFINITION
Alkali Metals
TERM
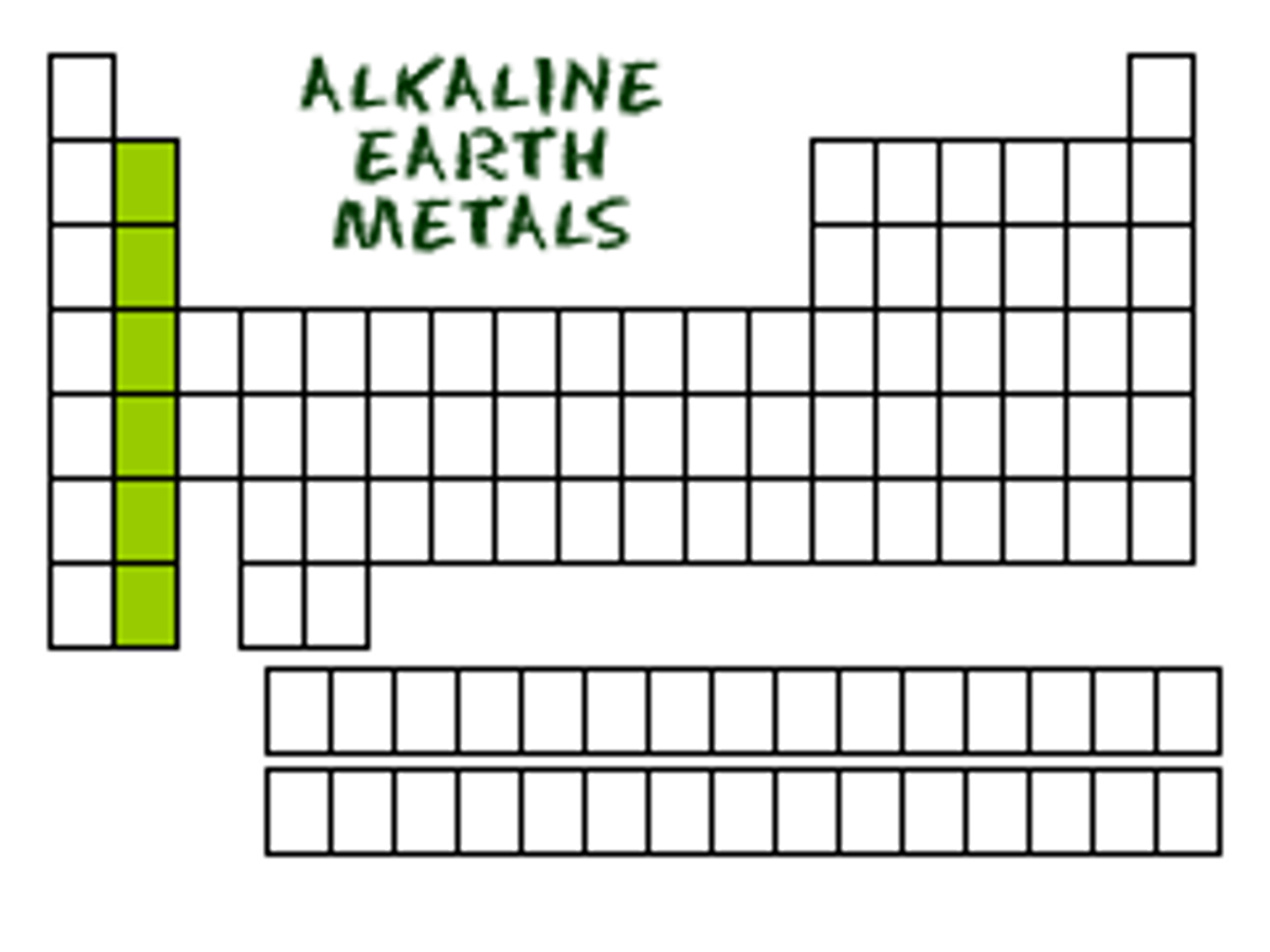
The elements in Group 2A of the periodic table.
Shiny, Silvery-white
Very reactive
DEFINITION
Alkali Earth Metals
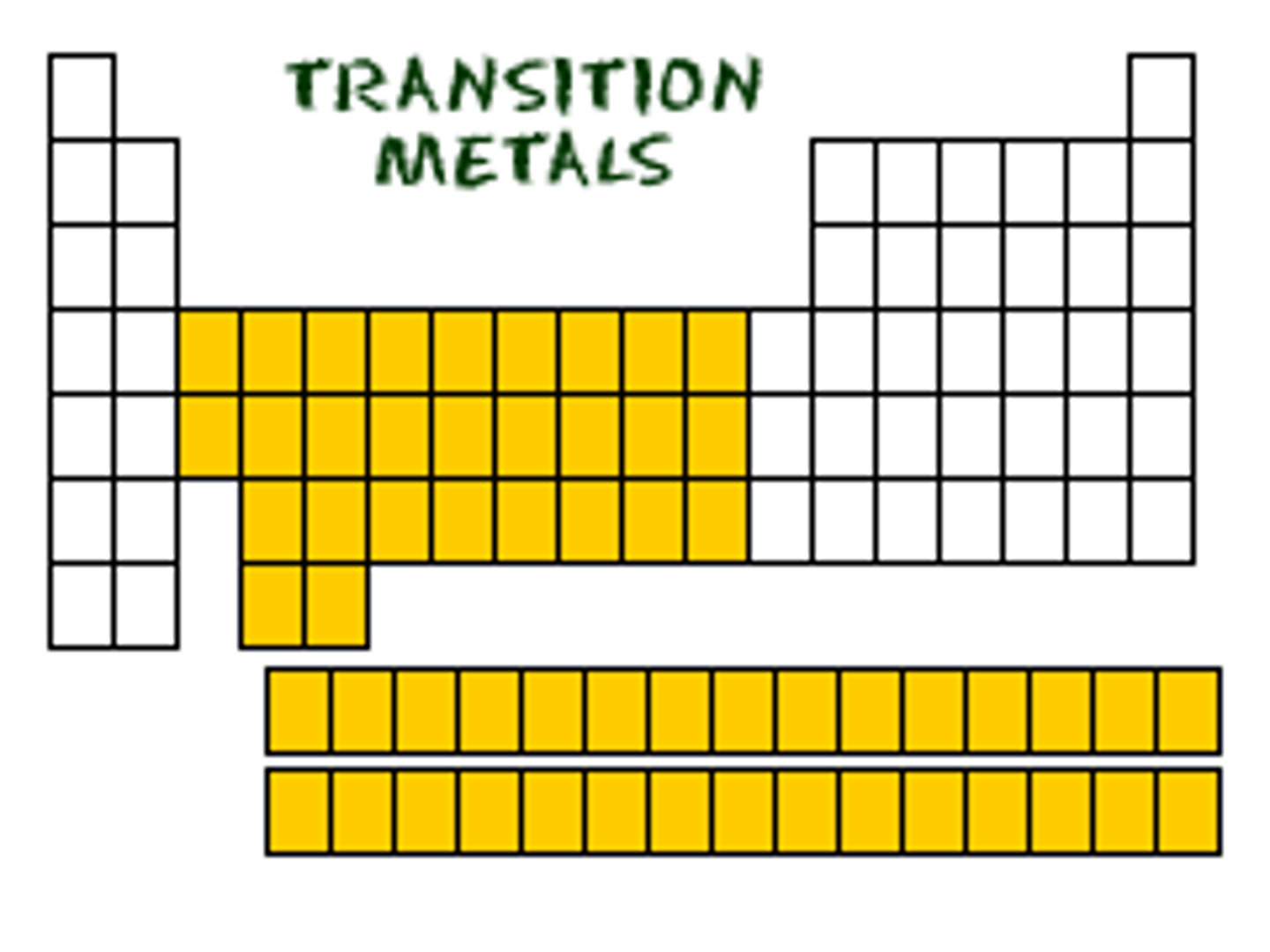
TERM
Group B elements
Form a bridge between the elements of the left and right sides of the Periodic Table (middle of the table).
-Non-reactive metals.
-Less predictable than other elements.
-Paramagnetic properties
DEFINITION
Transition Metals
TERM
Elements that have properties of both metals and nonmetals.
DEFINITION
Metalloids
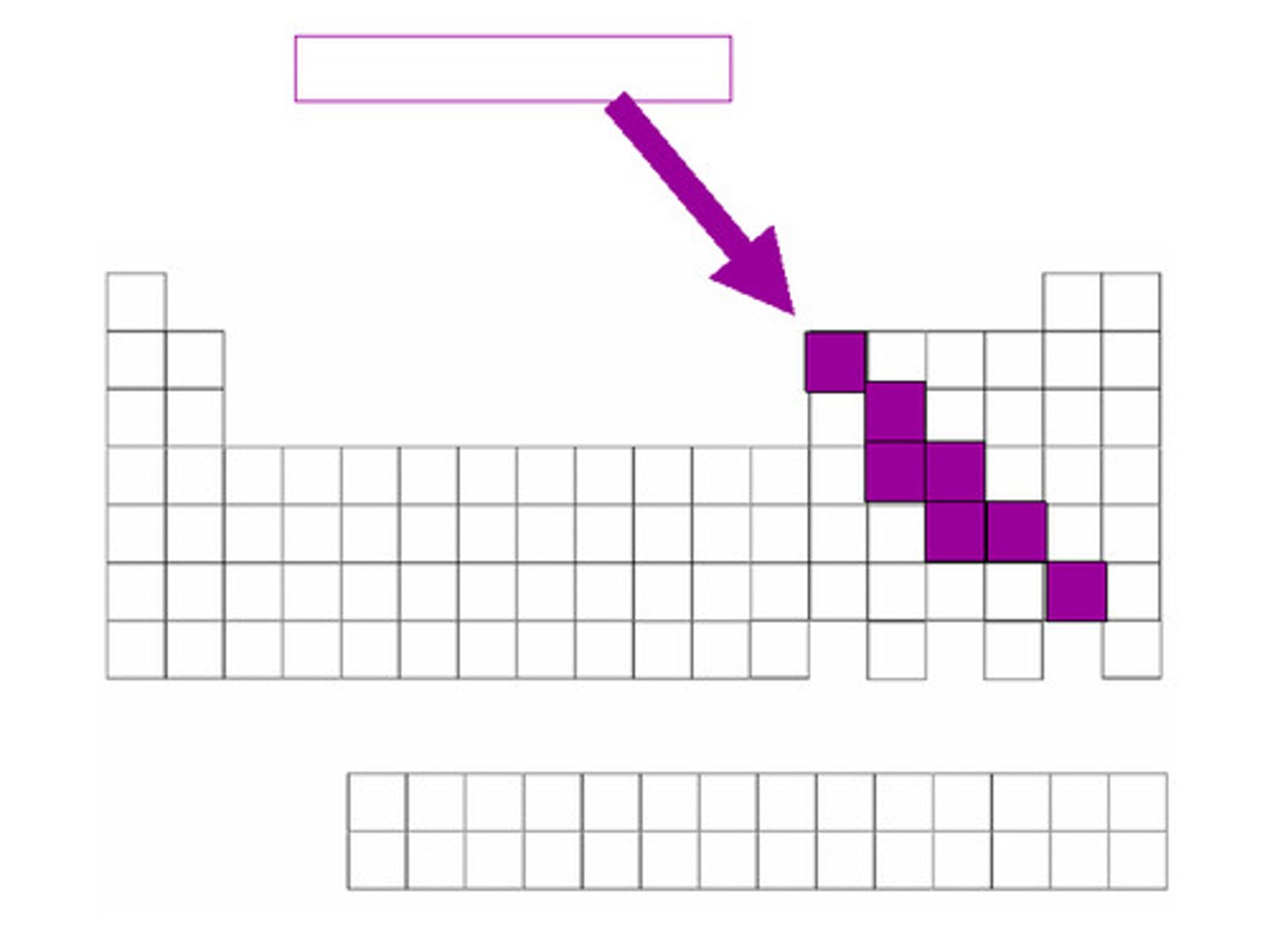
TERM
-Brittle when solid
-Dull
-Poor conductors
-Lower melting and boiling points than metals
DEFINITION
Non-metals
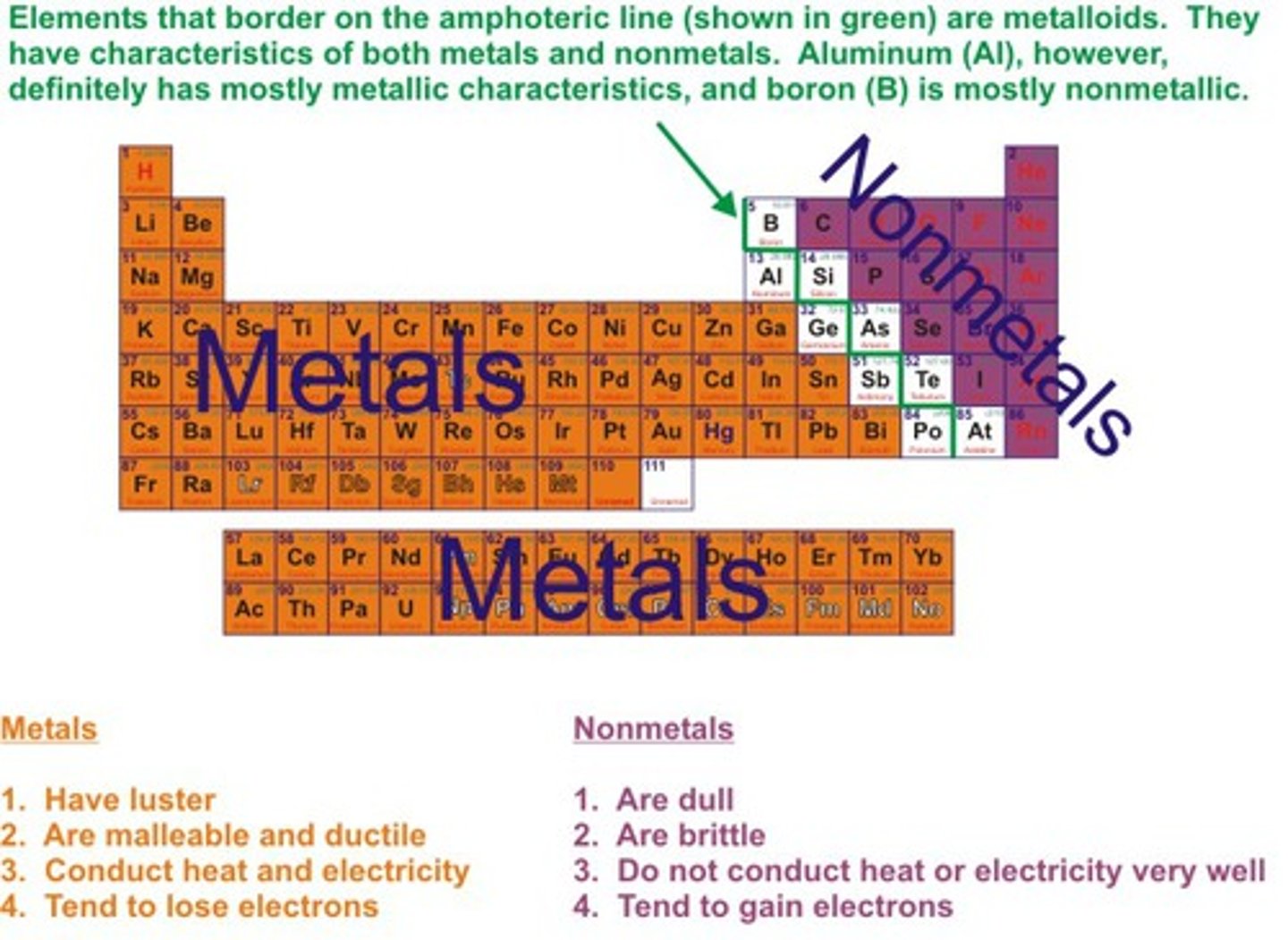
TERM
The elements in Group 7A of the periodic table.
-Very reactive
-Poisonous
-Bleaches and disinfectants
DEFINITION
Halogens
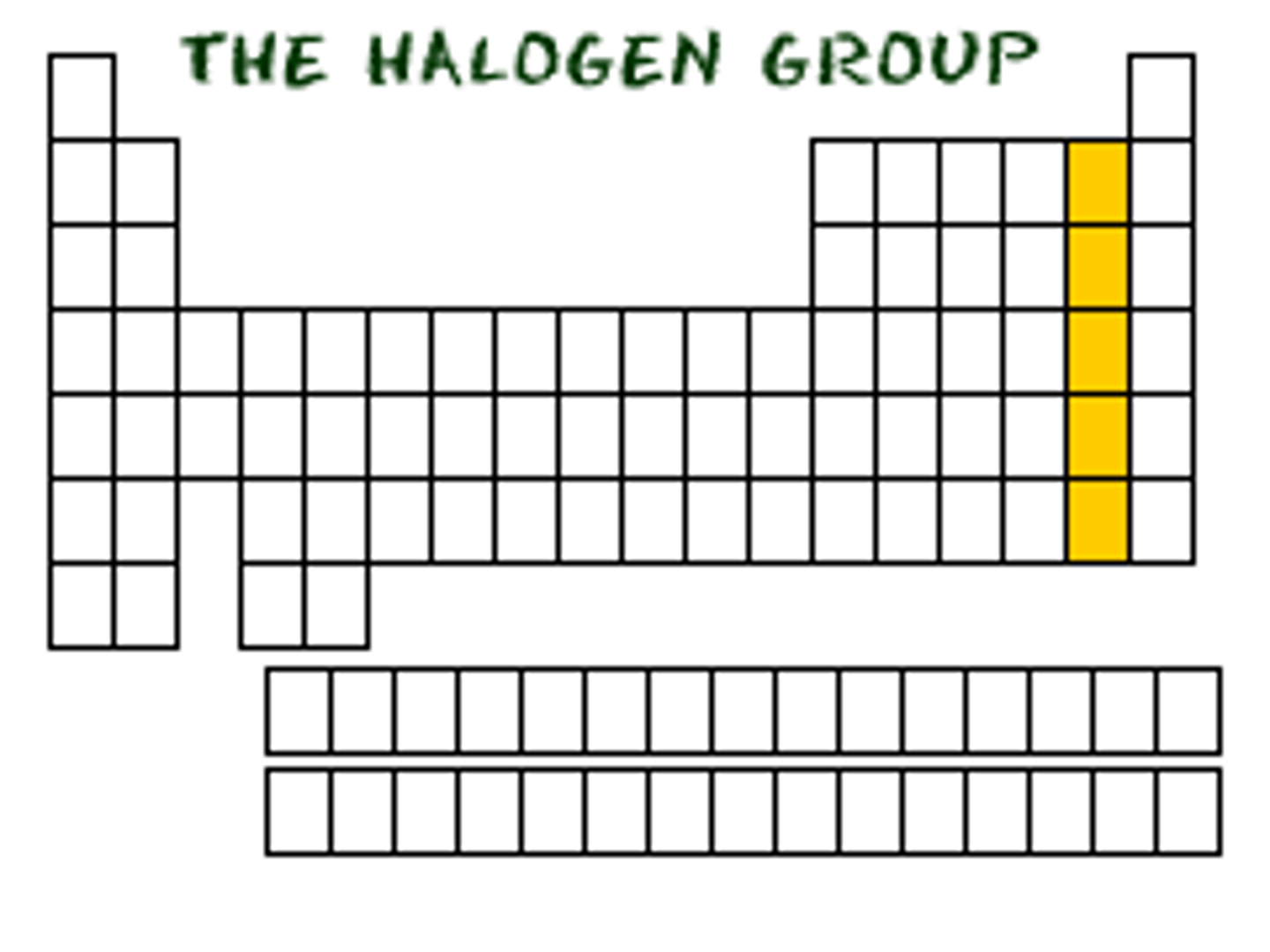
TERM
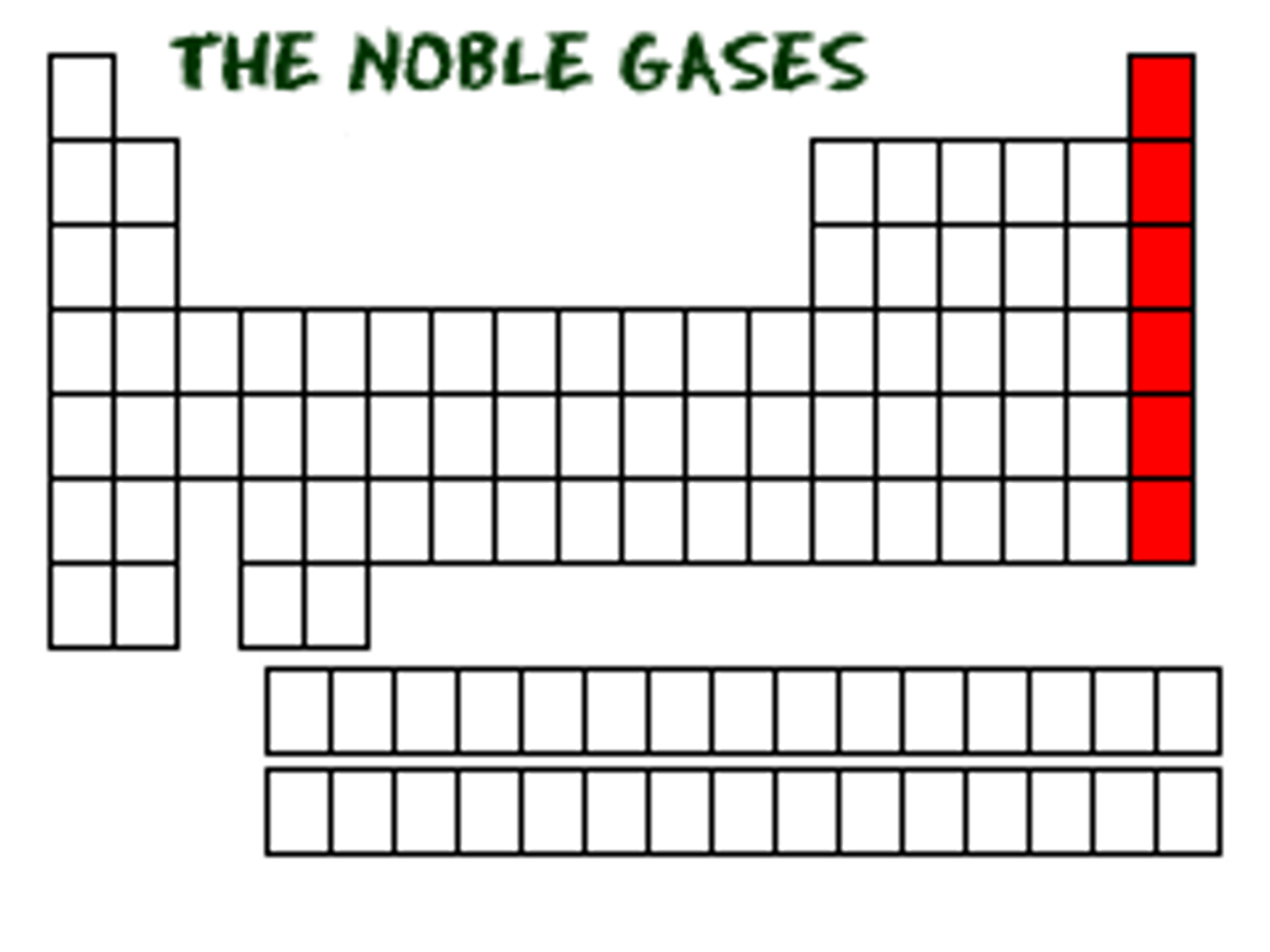
The elements in group 8A of the periodic table.
-Colorless
-Odorless
-Extremely NONREACTIVE
DEFINITION
Noble Gases
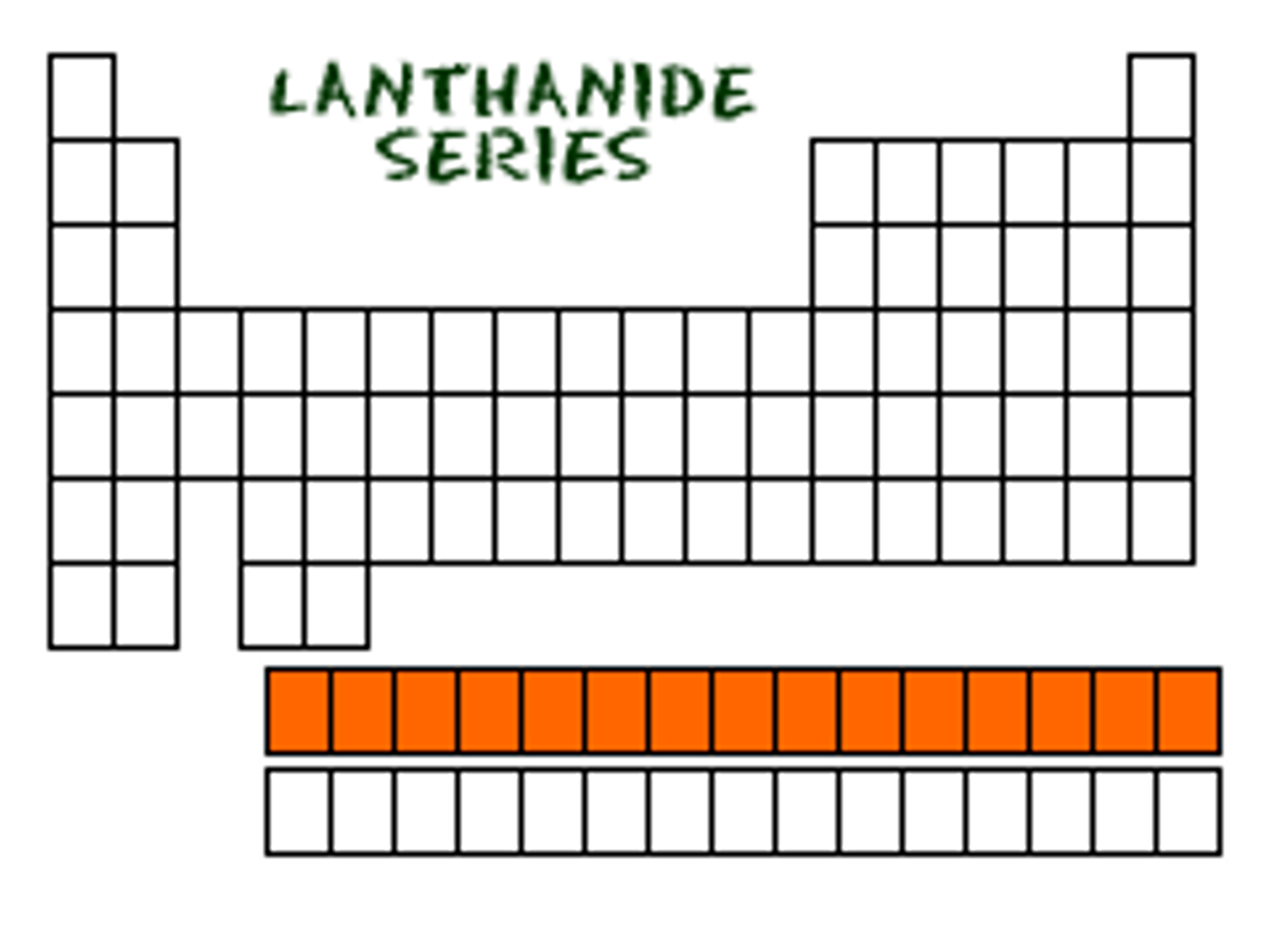
TERM
The elements with atomic numbers from 58 to 71
DEFINITION
Lanthanides
TERM
The last row of the periodic table made up of elements with atomic number 89 to 103
DEFINITION
Actinides