Foundational Biology
1/154
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
155 Terms
compartmentalisation
The division of cellular functions into separate membrane-bound compartments, allowing for specialised environments and increased efficiency within a eukaryotic cell.
nucleus
The membrane-bound compartment that contains the cell's genetic material and regulates gene expression and cell division.
nucleus structure
surrounded by a double membrane/nuclear envelope. Presence of nuclear poles that control movement of molecules across envelope. Contains the most DNA in the cell. Continuous/attached to the rough ER.
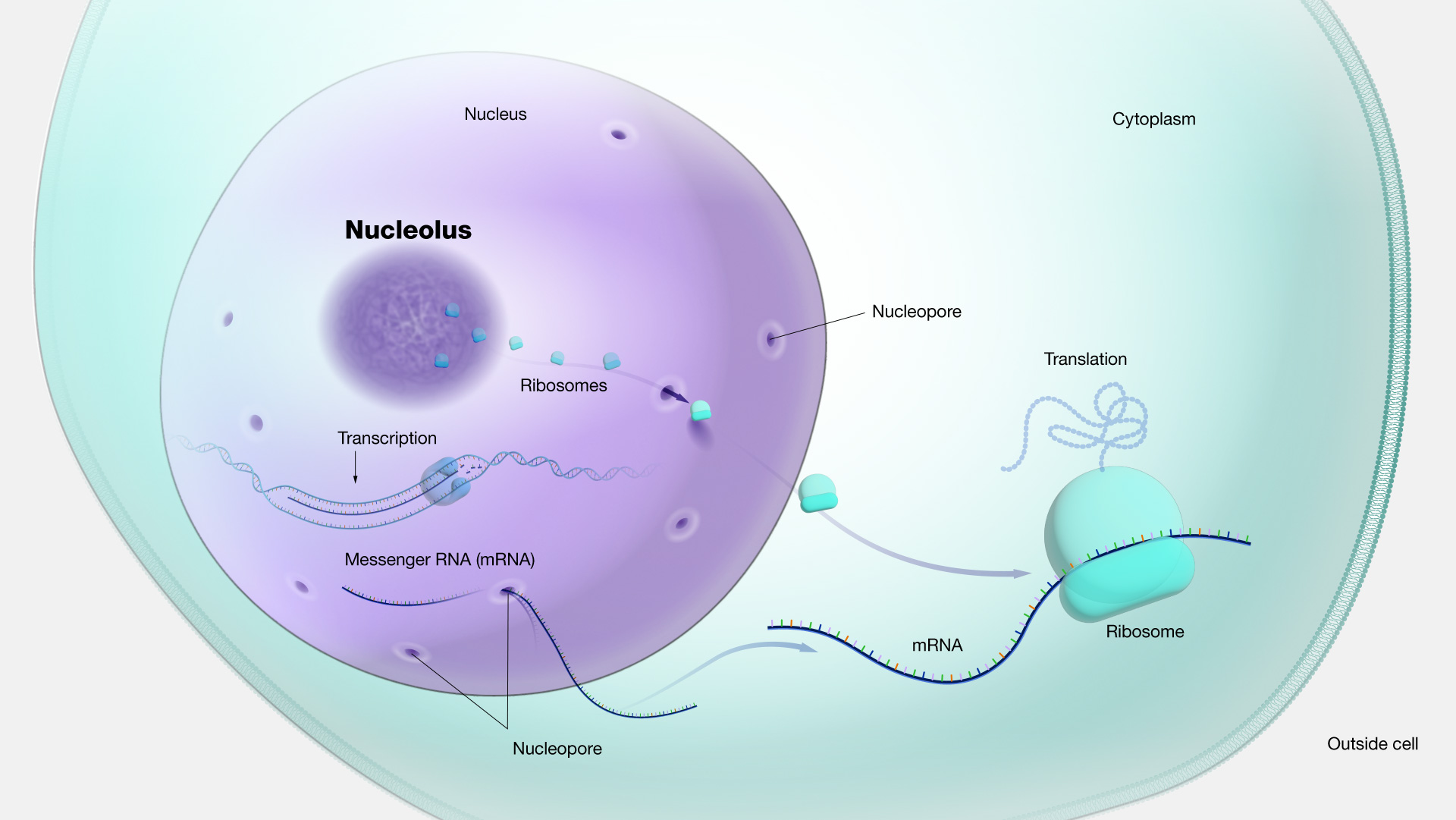
nucleolus
small dense spherical subregion of nucleus with transcribing/ribosomal genes
chromosome
long strands of DNA supercoiled and covered in histones. carries genetic information. found in nucleus, important for heredity, gene expression and cell division.
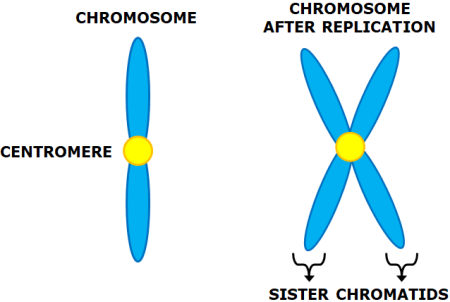
chromosome structure
made up of a chromatin, complex of histone and DNA which condenses to form chromosomes. held together by kinetochore, makes the centromere. 2 identical sister chromatids make a chromosome.
origin of nucleus
from invagination (inward fold) of plasma membrane around nucleotide of ancient prokaryote. looks millions of years. forming both nucleus and ER.
nucleoid
religion on prokaryotic cells, not surrounded by membrane. contains single circular DNA molecule, not formed into chromosomes.
mitochondrion.
a cell may have several or 1 large one. it is surrounded by 2 membranes, outer membrane and inner membrane, with the inner projections of it called the cristae. carry out aerobic respiration of all eukaryotes.
plastid
A plastid is a broad category of double-membrane organelles in plant and algae cells. eg. a chloroplast is a specific type of plastid specialised for photosynthesis
what is life
Living things are made of common elements and organised into cells. They contain genetic information, grow and change, respond to their environment, and carry out chemical reactions to build and use molecules. They obtain and use energy to survive, and exist in populations that can evolve over time.
cell theory
A fundamental unifying theory of biology stating that all living organisms are made of cells, cells are the basic unit of life, all cells arise from pre-existing cells, and thus modern cells evolved from a common ancestor.
evolution
The process where variation among individuals leads to differential reproduction, causing populations to change over time leading to evolution.
where did life come from
Theory 1: arisen spontaneously on Earth, environmental conditions enabled the formation of organic molecules from inorganic substances (e.g. DNA bases), supported by experiments like the Miller–Urey experiment (simulated early Earth’s conditions)
Theory 2: originated extraterrestrially arriving via meteorites such as the Murchison meteorite, which contains organic compounds associated with living systems.
formation of life
Hadean period: Earth formed, when the first oceans developed, little to no oxygen
Archean period: Life began, water present ~3.8 billion years ago, early prokaryotes (e.g. cyanobacteria) appearing around 3.5 billion years ago. 1.4 billion years later, unicellular eukaryotes evolved
Cambrian period: ~600 million years ago, more complex life began to diversify, terrestrial animals.
stromatolites
Layered structures formed by cyanobacteria, often called “living fossils,” that provide evidence of early life on Earth.
large time gap between bacteria, unicellular eukaryotes and terrestrial animals
no ozone layer
temperature conditions not optimial
poor oxygen atmosphere
photosynthesis
6CO₂ + 6H₂O ⇌ C₆H₁₂O₆ + 6O₂
Carbon dioxide and water are converted into glucose and oxygen using energy (light)
UV radiation types
UV-A, UV-B, UV-C
UV-C = most harmful to organisms
UV-A, UV-B = least harmful
can cause DNA damage and mutations
Early life protection from UV
water absorbed and reduced radiation, protecting organisms which is why they remained aquatic
How did oxygen first build up in the atmosphere
cyanobacteria undergo photosynthesis, overtime CO2 in the atmosphere is being cycled out and O2 replacing it (for cellular respiration
forming the ozone layer
UV radiation splits oxygen molecules: O2 → O + O
oxygen radicals then collide with O2: O + O2 = O3
ozone molecules accumulate in the stratosphere forming a protective ozone layer
took 1.5 billion years
function of ozone layer
absorb mostly UV-C
partially absorbs UV-A and UV-B
massively deceasing harmful radiation onto earth’s surface and life
Cambrian Explosion
rapid diversification of life, 541 mya
major animal groups and major body structures first appeared
triggered by ozone layer formation
mostly fossils from this period due to the beginning of hard shell and exoskeletons (previous were soft-bodies and less likely to fossilise)
taxonomy
the system used to classify and organise living organisms based on shared characteristics
highest level of classification → 3 domains of life (Eukarya, Archaea, Bacteria
kingdoms of life
used to group organisms in the Eukarya domain
6 kingdoms overall (Animalia, Plantae, Fungi, Protista, Bacteria, Archaea
Animalia
multicellular
heterotrophic (must acquire food)
no cell walls
most can move
Plantae
multicellular
autotrophic (make own food, photosynthesis)
cell walls (made of cellulose)
mostly non-motile
Fungi
heterotrophic (absorb nutrients)
cell walls (made of chitin)
mostly decomposers
Protists
mostly single celled eukaryotes
can be plant like, animal like or fungus like
Prokaryotes
simple, unicellular organisms (bacteria and archaea) that do not have a nucleus
what structures do all prokaryotes have
cell membrane
cytoplasm
ribosomes
DNA in a nucleoid region ( not a nucleus)
what do most bacteria have outside the cell membrane
a cell wall outside the plasma membrane made of peptidoglycan for support and protection
what is a bacterial capsule
a sticky outer layer made of polysaccharides, helps with protection and attachment
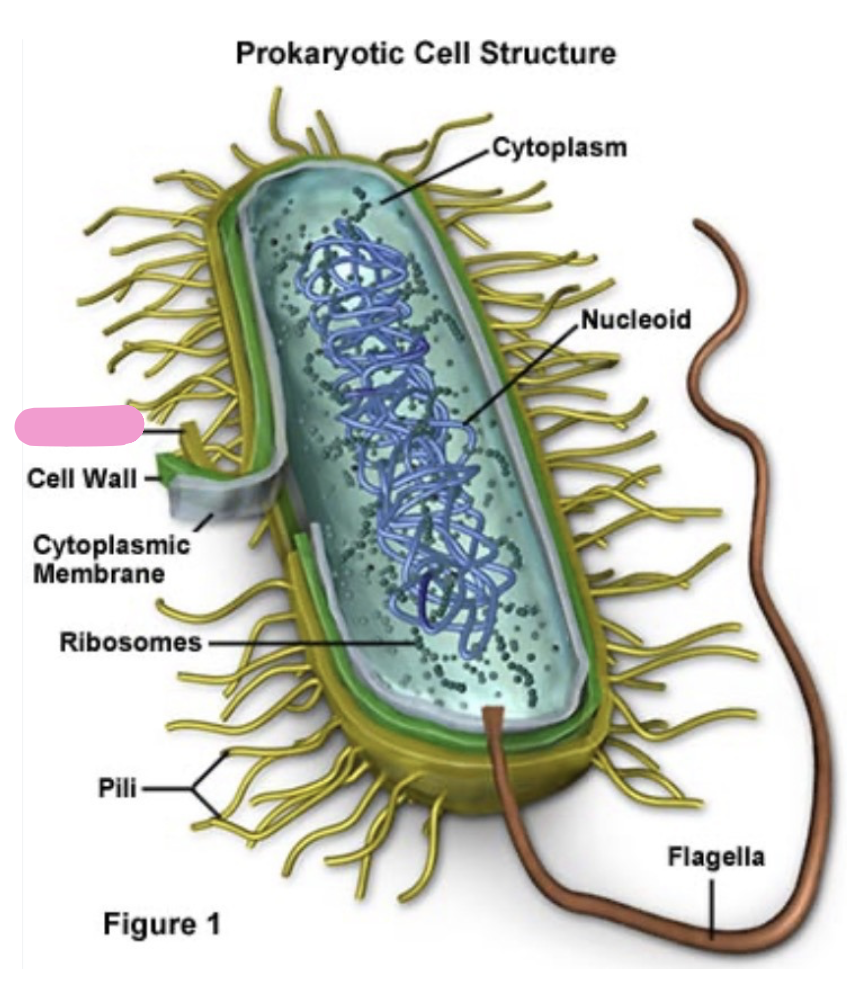
what is a flagellum
movement (helps bacteria swim or move towards nutrients)
more complex in eukaryotes
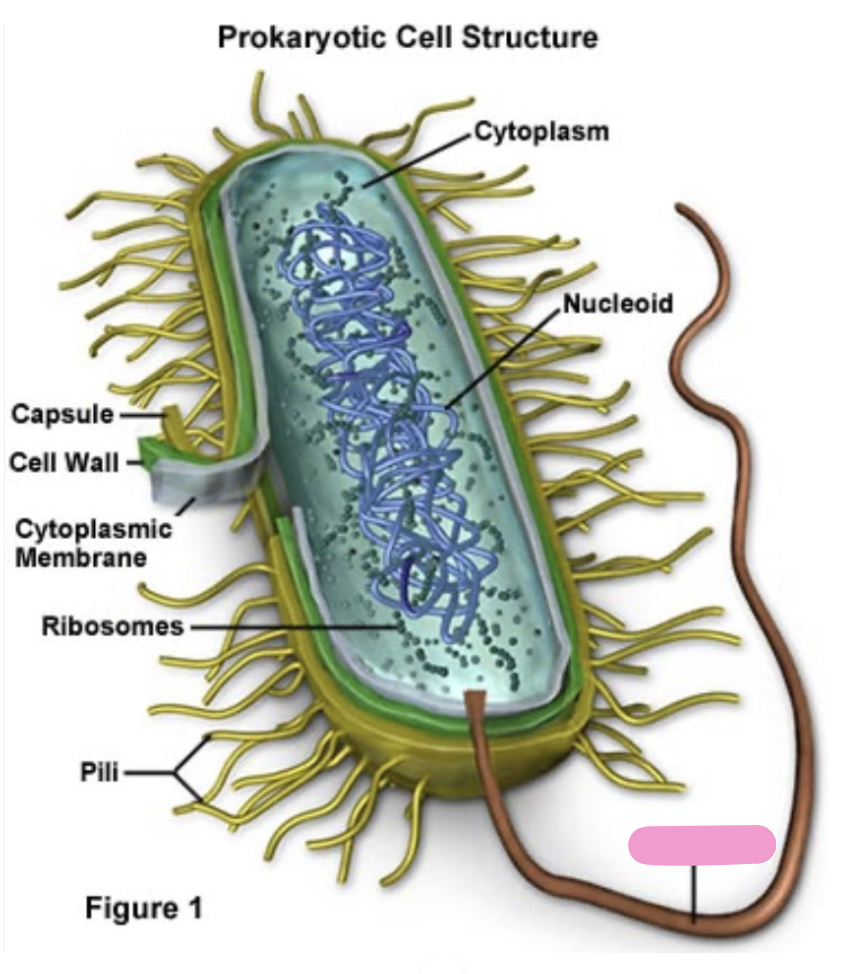
what are pili
help with attachment to surfaces
transfer of DNA between bacteria
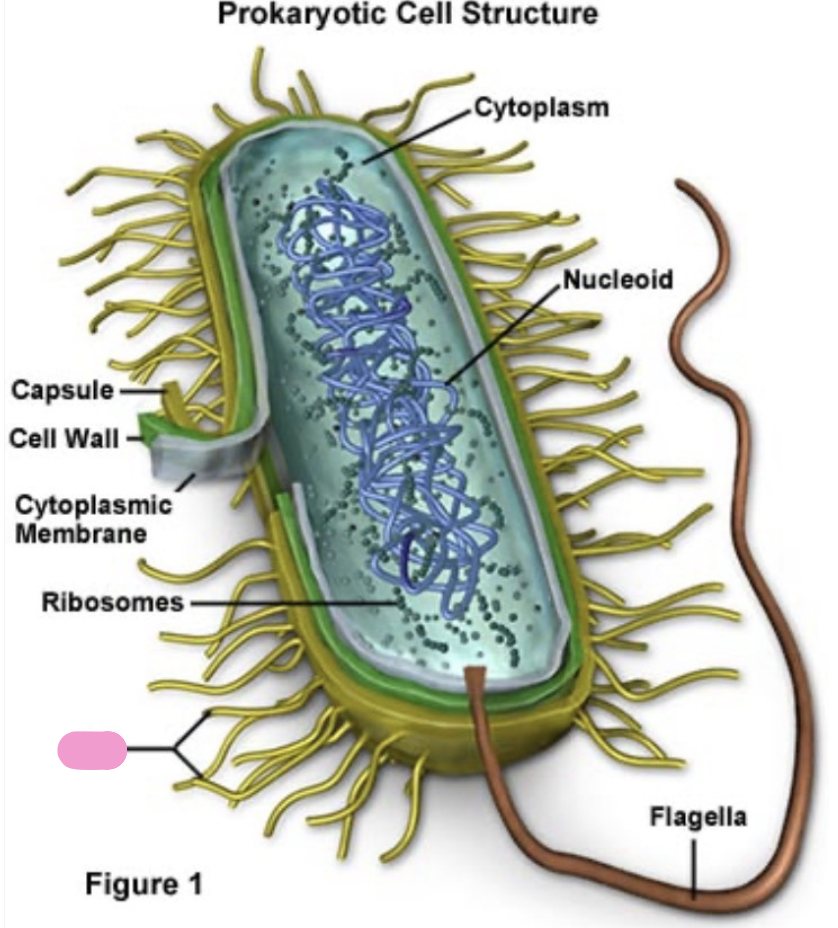
what are ribosomes
small cellular structure made of ribosomal RNA and proteins that carry out protein synthesis
what do ribosomes do
site of protein synthesis, where amino acids are joined to form proteins
prokaryotic ribosomes vs eukaryotic ribosomes
prokaryotic:
smaller and simpler
less complex structure
eukaryotic:
larger and more complex
have extra structural components (extensions) thus can make more proteins
atomic structure
the whole universe is made of 2 catergories
matter
energy
matter
anything that has mass and takes up space/volume
the fundamental unit of matter = the atom
Atoms structure
have a central nucleus made of protons (positive charge) and neutrons (no charge)
surrounded by a cloud of electrons (negative charge)
atoms are mostly empty space (vacuum) between the nucleus and electrons
what is electronegativity
a measure of an atom’s ability to attract its bonding (outer) electrons (-) towards its nucleus (+)
how does number of protons effect electronegativity
more protons → more positive nucleus → higher electronegativity
how does electronegativity change across the period
it increases from left to right because nuclear charges increases while shielding stays similar.
how does electronegativity change down a group?
it decreases down a group because electrons are further from the nucleus and more shielded
nuclear charge
the total positive charge of an atom’s nucleus, determined by the number of protons. (Z)
how does number of electron shells nucleus affect electronegativity
more electron shells → greater distance → less attraction → less electronegativity
two main factors that control electronegativity
nuclear charge (no. of protons)
distance/shielding (number of electron shells)
what are bonds
when 2 atoms together they can share outer electrons, forming a bond
intramolecular bond types
ionic, polar covalent, non-polar covalent
ionic bonds
electronegativity difference > 1.9
more electronegative atom steals electrons from another atom
creates permanently charged cation (+) and anion (-)
polar covalent
electronegativity difference → 0.4-1.9
two atoms unevenly share a pair of electrons
the electrons are closer to more electronegative atom
non-polar covalent
electronegativity difference < 0.4
two atoms evenly share a pair of electrons
electrons are equidistant between nuclei
carbon
the primary component of all known life
has 4 electrons in its outshell and can form up to 4 covalent bonds
what are macromolecules
large molecules (polymers) container thousands or more atoms
made of smaller repeating subunits called monomers
carbohydrates, proteins, nucleic acids, lipids
but lipids are not polymers → subunits are fatty acids and glycerol
intermolecular forces
electrostatic forces between atoms, molecules or ions
ion-dipole, dipole-dipole, London disperson force, hydrogen bonding
London dispersion forces
weakest intermolecular attraction, caused by temporary fluctuations in electron distribution, creating instantaneous dipoles
exist in all molecules, but is the only force present in non polar molecules
the larger the atom, the more electrons ad greater surface area = stronger London dispersion force
dipole-dipole forces
intermolecular attraction between the positive end of one polar molecule and the negative end of another
the molecule must have an asymmetrical distribution of charges
meaning molecules have 1 positive side and 1 negative side
also called hydrophilic interactions
hydrogen bonding
strong type of dipole-dipole interaction, involves hydrogen bonded to highly electro negative atoms (N, O, F)
the bond is extremely polar, causing the hydrogen electron to be pulled away and expose the hydrogen nucleus, leaving a naked positive charge (H+ proton)
the hydrogen forms a bond to other electron rich atoms given that it is a lone pair of electrons (generally another N, O, F)
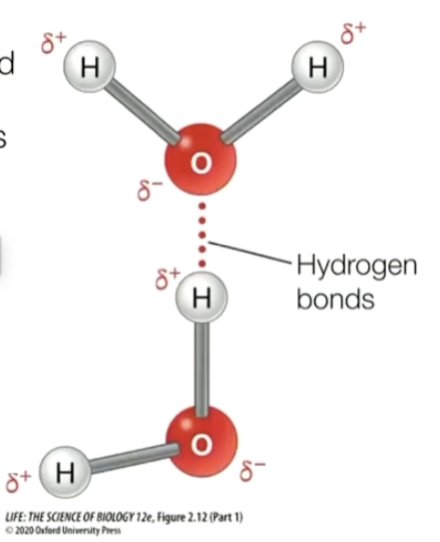
ion-dipole forces
strong intermolecular attractions between an ion and a polar molecule
occur when ionic compounds dissolve in polar solvents (like water)
between an ion (cation or anion) and a polar molecule (dipole)
order of strength for intermolecular forces
hydrogen < dipole-dipole < hydrogen < ion-dipole
how do water molecules interact with each other
properties explained through hydrogen bonding
water molecules form a dynamic network of hydrogen bonds that are constantly breaking and reforming
water properties
high melting point
high specific heat capacity
high heat of vaporisation
cohesion
adhesion
high specific heat capacity of water
water can absorb large amounts of energy before changing temperatures
because energy has to be used to break down the strong hydrogen bonds instead of just increasing kinetic energy and molecular movement
limits the rise in temperature, slows temperature change
stabilises environmental temperature, preventing rapid heating or cooling that can damage aquatic organisms
high melting point of water
water’s hydrogen bonding creates strong intermolecular forces that require significant energy to overcome before transitioning from solid ice to water
high heat of vaporisation
water requires large amount to completely break hydrogen bonds so water molecules can escape into gas phase, makes evaporation energetically costly
cohesion in water
attraction between water molecules, caused by hydrogen bonding
holds water molecules together and contributes to surface tension where water holds together at the surface
surface tension → resistance of the surface of water to being broken/stretched because water molecules have a strong cohesion to neighbouring water
adhesion in water
attraction between water molecules and other substances
water’s polarity allows it to form hydrogen bonds or electrostatic interactions with other polar or charged surfaces
seen in water sticking to glass or plant cell walls
what makes a molecule polar
when a molecule has one or more polar bonds, where the electronegativity differences are big enough to consider it a polar bond
must be asymmetrical (one side is positive, one side negative)
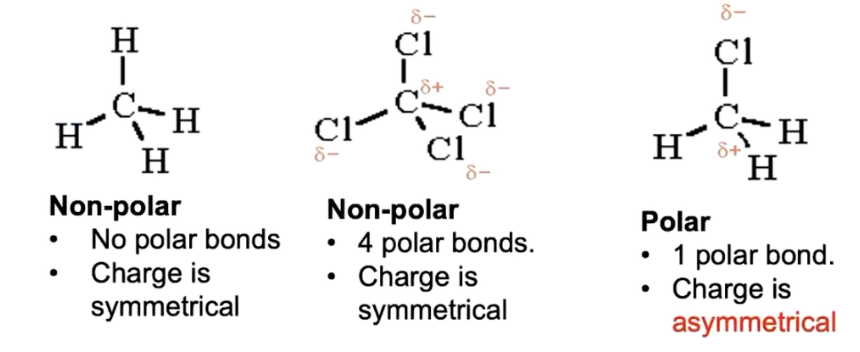
water’s polarity
water is a polar molecule because of an uneven distribution of electron density, creating bent asymmetrical shape
has a partial negative charge near the oxygen and a partial positive charge near the hydrogen atoms
arises from oxygen’s high electronegativity, which pulls shared electrons closer, allowing hydrogen bonds and acts as a universal solvent
hydrophilic
water loving, substances interact readily with water
polar or charged molecules
hydrophobic
water fearing, substances that do not interact well with water and tend to repel it
non-polar molecules
water as a medium for chemical reactions in living organisms
most biochemical reacts occur in aqueous solutions
because water dissolves many polar and ionic substances
molecules can move freely in it as hydrogen bonds in water are constantly breaking and reforming
water as an acid or base
amphiprotic behaviour
water can donate H+ (acid) = hydroxide OH-
water can accept H+ (base behaviour) = hydronium H3O+
water can react with itself to make hydroxide and hydronium
2H2O(l)⇌H3O+(aq)+OH−(aq)
water acting as an acid
H2O→OH-+H+
water donates H+ to become hydroxide
water acting as a base
H2O+H2→H3O+
water uses a lone pair on an oxygen and donates a proton H+
forms hydronium
hydrolysis
reaction where water is added and a covalent bond is broken
large molecules are split into smaller monomers
condensation
builds polymers and produces water, covalent bond is formed
reverse reaction of hydrolysis
carbohydrates
general formula = (CH2O)n → for polysaccharides
n = number of carbon atoms
basic unit → monosaccharides (basic sugars)
types of carbohydrates
1 subunit → monosaccharide (glucose, ribose, deoxyribose)
2 subunits → disaccharide (sucrose, maltose, lactose)
3-10 subunits oligosaccharide (often a receptor signal on end of proteins)
100s or 1000 subunits → polysaccharides (CH2O)n (starch, glycogen, cellulose)
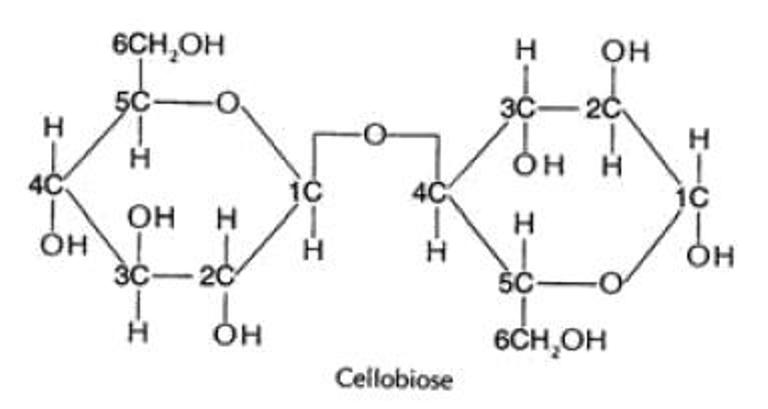
glycosidic bonds
a covalent bond formed when two monosaccharides (sugars) join via condensation making a disaccharide
water is released
also called ether linkage in carbohydrates
each sugar has multiple hydroxyl (-OH) groups
one sugar provides a H from an -OH
one sugar provides an -OH
combine to H2O
sugar-OH + HO-sugar → sugar-O-sugar + H₂O
sugar and O bridge is the glycosidic bond
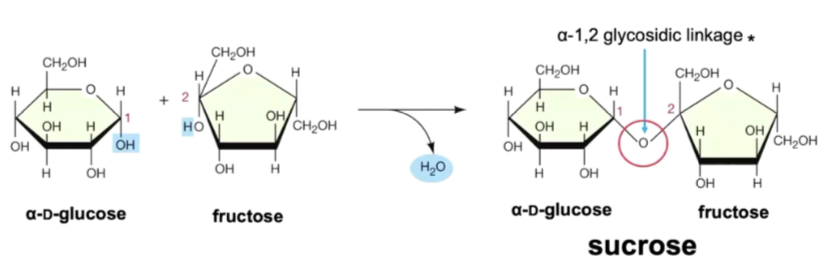
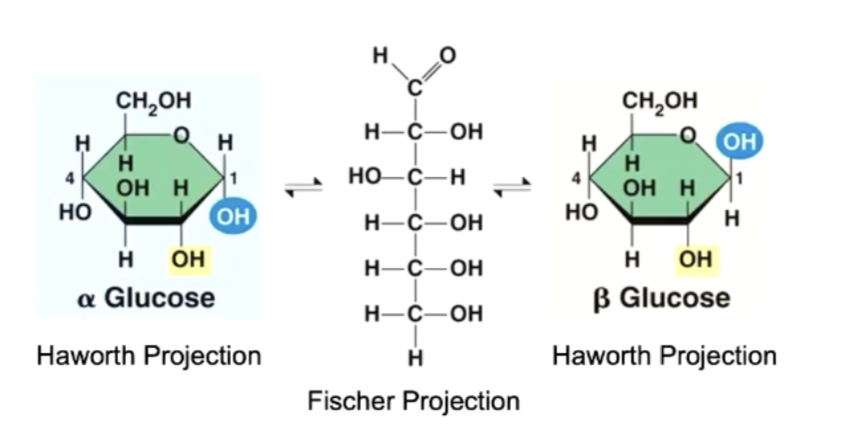
alpha and beta glucose
alpha glucose = OH below the ring at the carbon 1
beta glucose = OH above the ring at the carbon 1 (a glucose must flip upside down to form glycosidic bond)
why can humans digest starch but not cellulose
humans have enzymes for alpha 1,4 bonds (starch) but not beta 1,4 bonds (cellulose)
1,4 bond for carbon
carbon 1 of one glucose is joined to the carbon 4 of another glucose
linked by a glycosidic bond by a condensation reaction
glycogen
animal storage polysaccharide made of alpha-glucose, highly branched for rapid energy release (glucose does not need to flipped when forming glycosidic bond, branches easily and can form a coiled helix, more surface area)
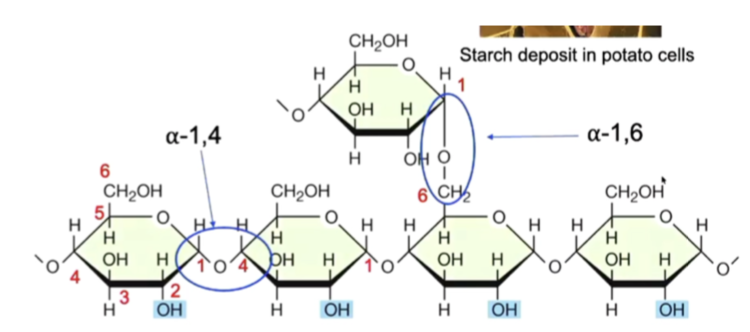
starch
plant energy storage polysaccharide made of alpha-glucose
easily degraded by enzymes (amylases)
primary energy storage compound in plants
alpha glucoses condense together, form an alpha 1,4 glycosidic link, a straight chain of glucoses
ends between branches can be digested and glucose can be taken off to access energy
can form coiled helix, similar to glycogen
cellulose
a structural polysaccharide in plant cell walls made of beta-glucose linked by beta-1,4 bonds
the chain forms hydrogen bonds, cellulose chains are grouped together as microfibrils
a straight rigid structure, cannot branch, more chemically stable than starch
humans cannot digest
cellulose chains into microfibrils
because of hydrogen bonding and London dispersion force (they’re large macromolecules)
cellulose fibrils maintain shape through hydrogen bonding between neighbouring hydroxyl group
condensation reactions form the chains, extensive hydrogen bonding links the chains together
Lipids
group of macromolecules mainly used for energy storage, membrane structure and signalling
non polar molecules (no charge) and cannot form hydrogen bonds, are insoluble
mainly held together by LDF, weak force individually but significant when many are together
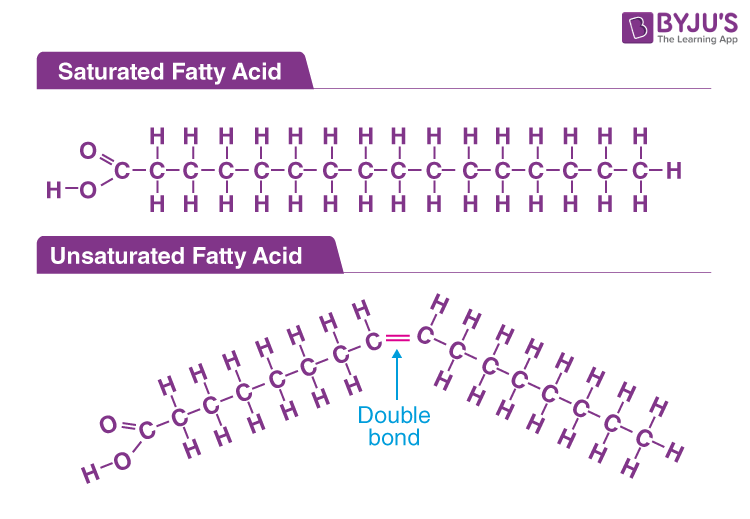
fatty acids
a long hydrocarbon chain with a carboxyl group (-COOH) at the end
saturated: only single bonds (C) between carbons, bonds can spin (flexible shape, and straight chains pack tightly together→ high LDF, decrease membrane fluidity and diffusion
unsaturated: one or more double bonds (C=C) in the carbon chain, bonds cannot spin and cause kinks, decreased packing, inflexible rigid structure → lower LDF, increase membrane fluidity and diffusion
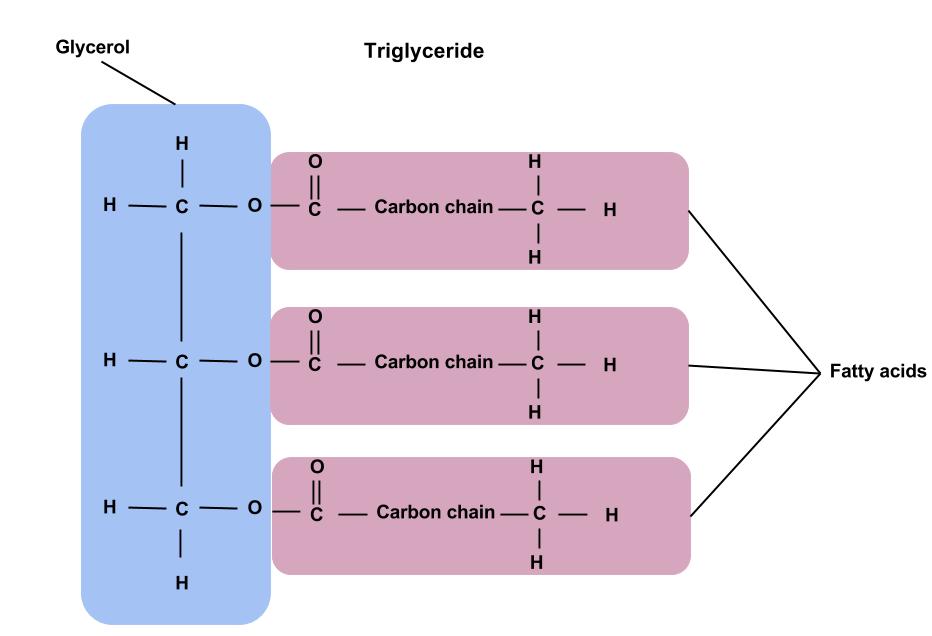
triglyceride
a type of lipid that is used for long-term energy storage, the oil and fat in our diet that gets stored within fat cells (adipose)
your body converts the extra calories into triglycerides, releasing them for energy during movement between meals
triglyceride synthesis: combine glycerol (a 3 carbon molecule with three hydroxyl groups) linked (via ester bond, condensation reaction) to 3 fatty acids (each has a carboxyl group at one end)
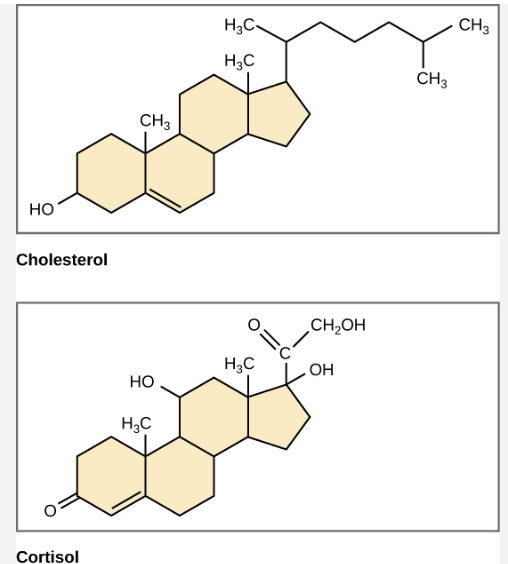
steroids
four fused carbon rings (the rings share carbons) from 17 carbons
three 6-membered rings (hexagon), one 5-membered rings (pentagon)
no fatty acid, a rigid ring structure
mostly non-polar but may have small polar groups attached
cell signalling
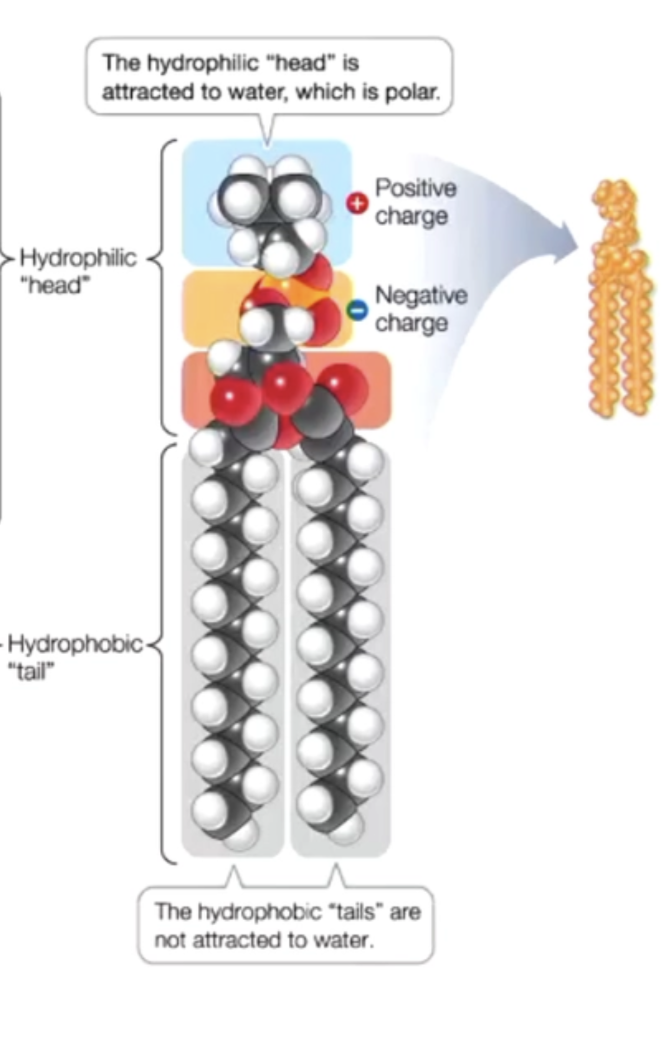
phospholipids
two fatty acid tails → form hydrophobic tails, very non polar
one phosphate group → hydrophilic head, very polar
allows both non polar and polar things to interact with it → called a surfactant
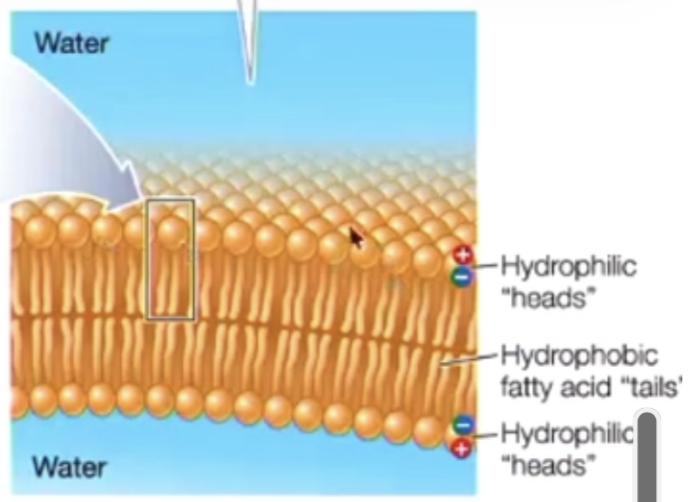
phospholipid bilayer
double later of phospholipid that forms basic structure of cell membranes
hydrophilic heads face outward towards water
hydrophobic tail faces inward cell away from water
inside the two layers of the bilayer are the fatty acid tails, forming a non polar interior
the heads can do ion bonding, dipole bonding, hydrogen bonding
tails can only do LDF (how the 2 layers are being held together)
this creates compartmentalisation, forming closed vesicles in water
nucleotides
the monomer of nucleic acids
consists of:
a nitrogenous base
pentose sugar (ribose or deoxyribose)
phospate group
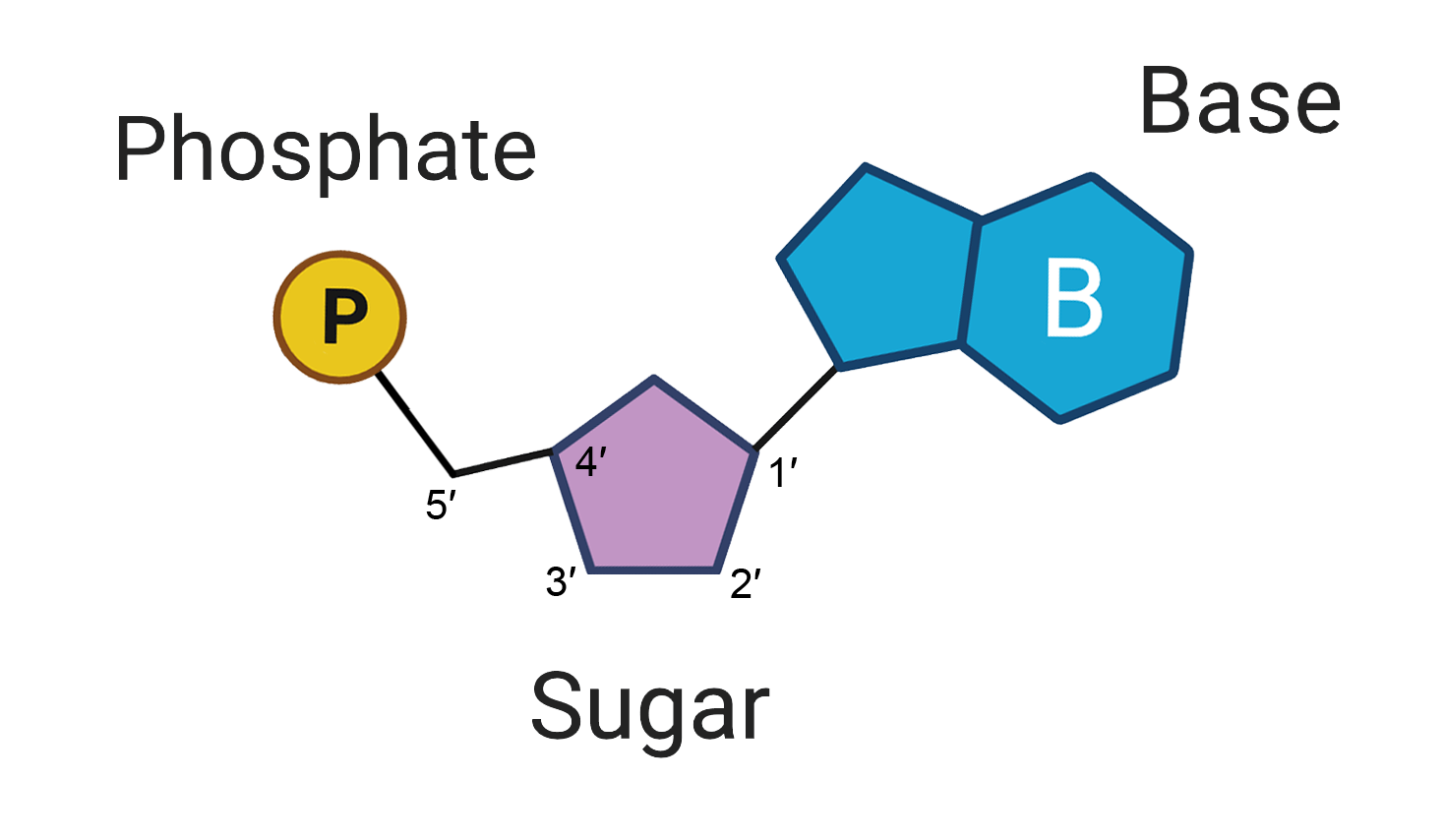
nucleoside
nitrogenous base + pentose sugar
nucleic acids
how we transfer and store info in living things
form long linear chains of receptive nucleotide units formed by phosphodiester bonds, making polynucleotides, that never branch
DNA: deoxyribonucleic acid
RNA: ribonucleic acid
DNA
deoxyribonucleic acid
4 nucleotides:
→ adenine
→ cytosine
→ guanine
→ thymine
has a double helix, 2 linear strands joined together by hydrogen bonds at the bases, antiparallel strands
stores genetic information in cells, is the template for RNA synthesis