Lecture 7 (Fibrous Proteins, Gibbs Free Energy & ATP Energy Coupling)
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms

What are globular proteins vs. fibrous proteins?
Globular:
Compact proteins with several secondary structures
Often enzymes, transporters, signaling proteins, and receptors
Fibrous:
Long strands/sheets
Usually one secondary structure
Provide support, shape, protection, and force transmission
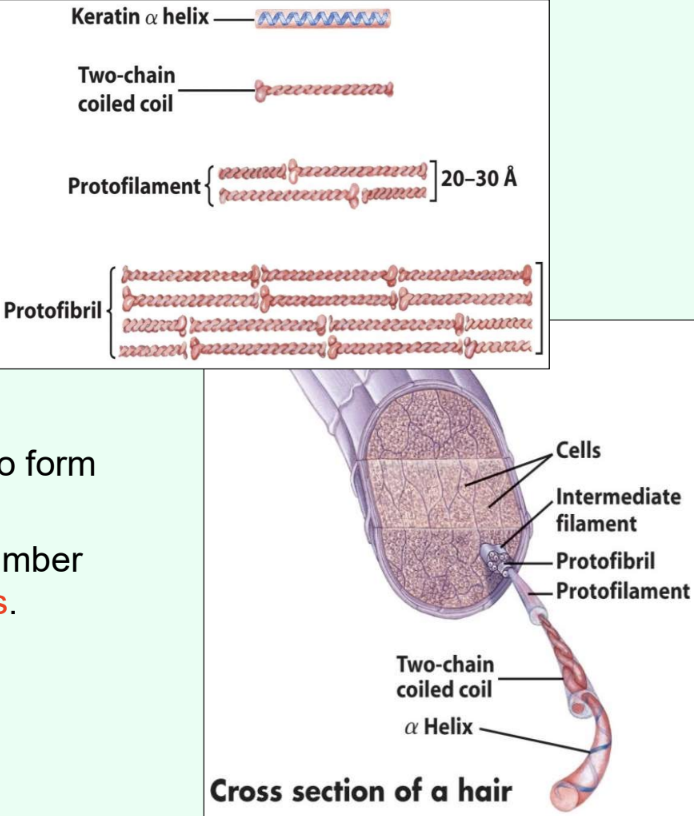
What is α-keratin and its structure?
High-tensile-strength fibrous protein in hair, nails, hooves, and horns
Two α-helices wound into a left-handed coiled coil → protofilaments → protofibrils
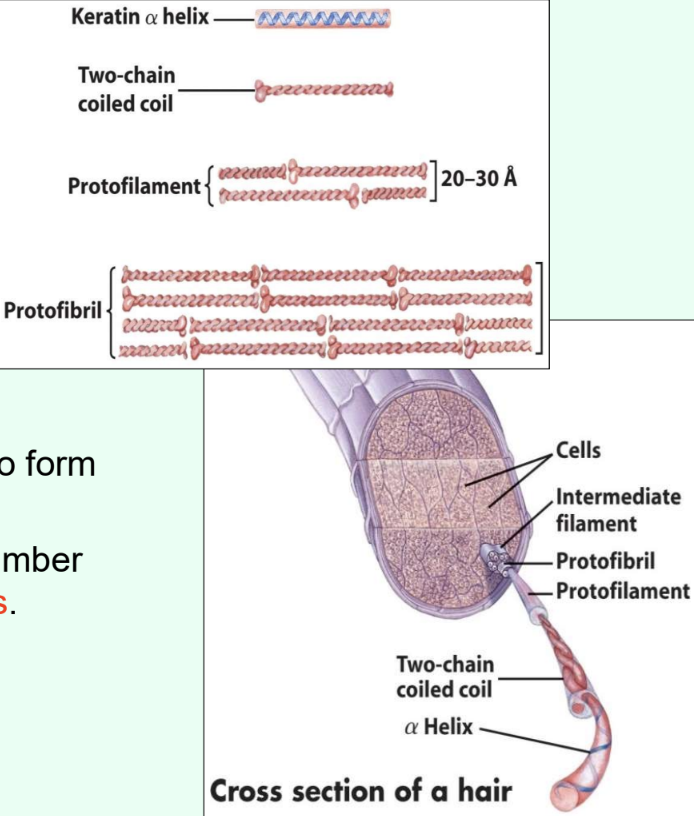
What determines α-keratin strength?
Number of intermolecular disulfide bonds
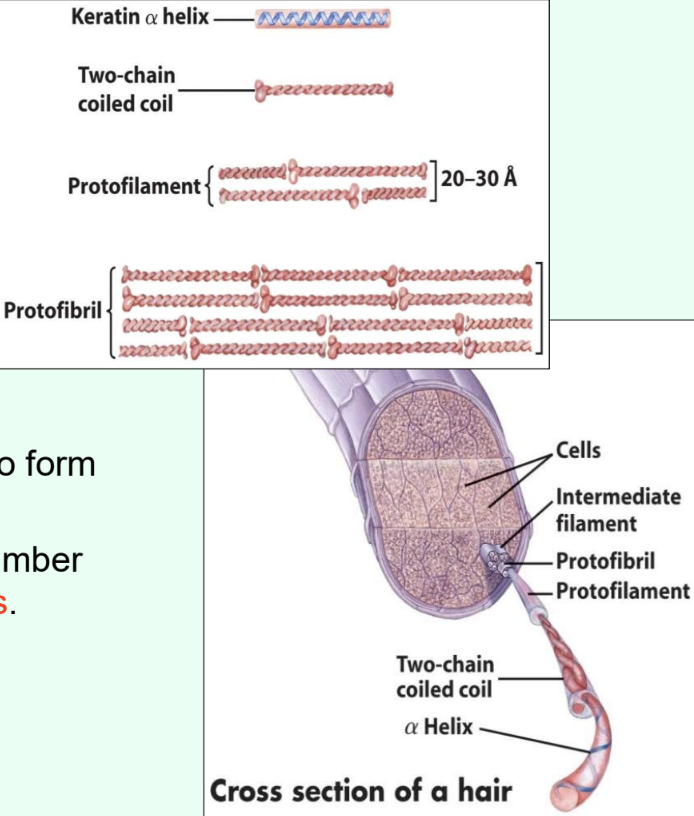
What protein family does α-keratin belong to?
Intermediate filaments
What is collagen found in? And what is collagen’s repeating sequence?
Cartilage, bone, and tendons
G-X-X
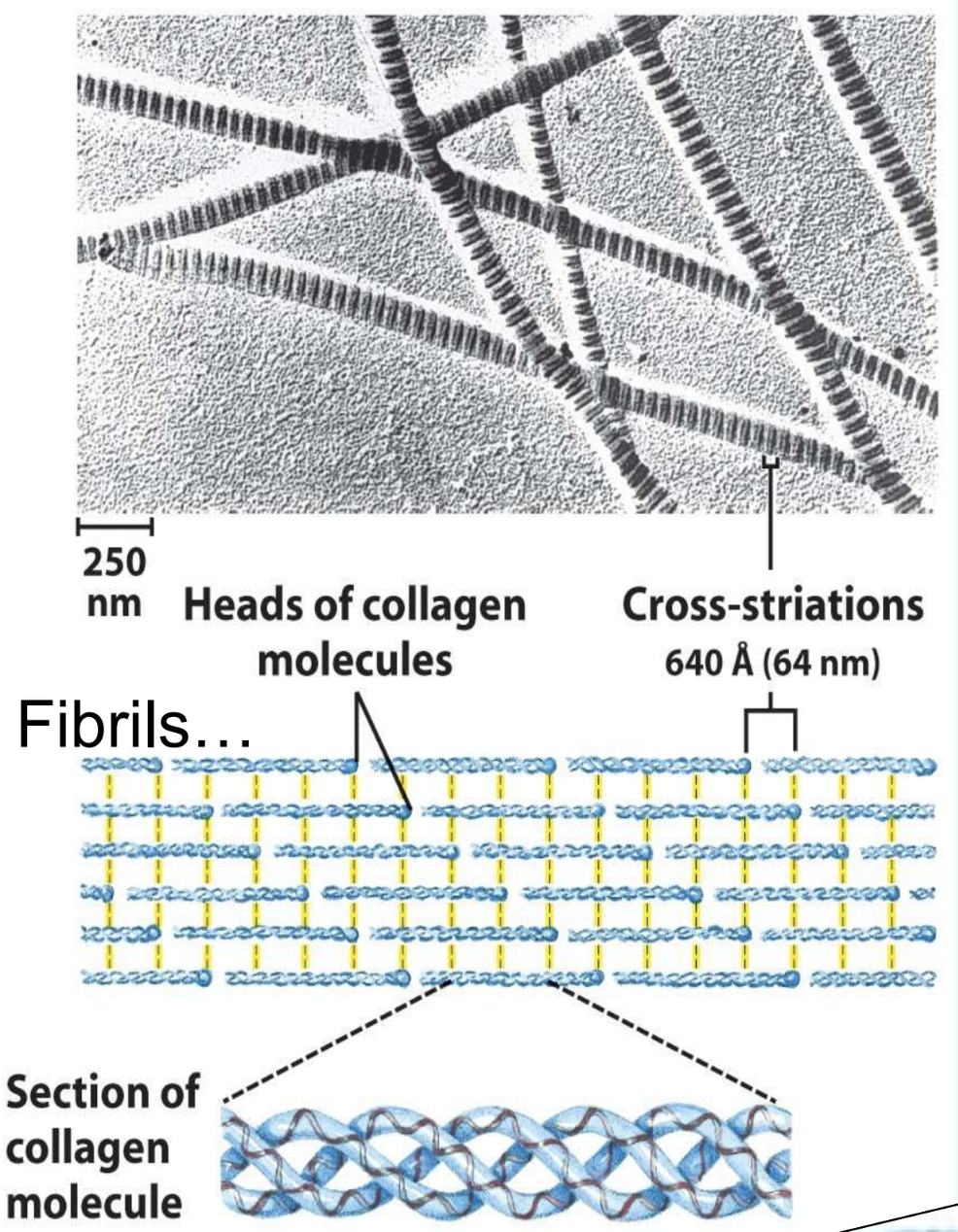
What is collagen’s helix structure?
Each chain is left-handed
3 chains coil into a right-handed triple helix
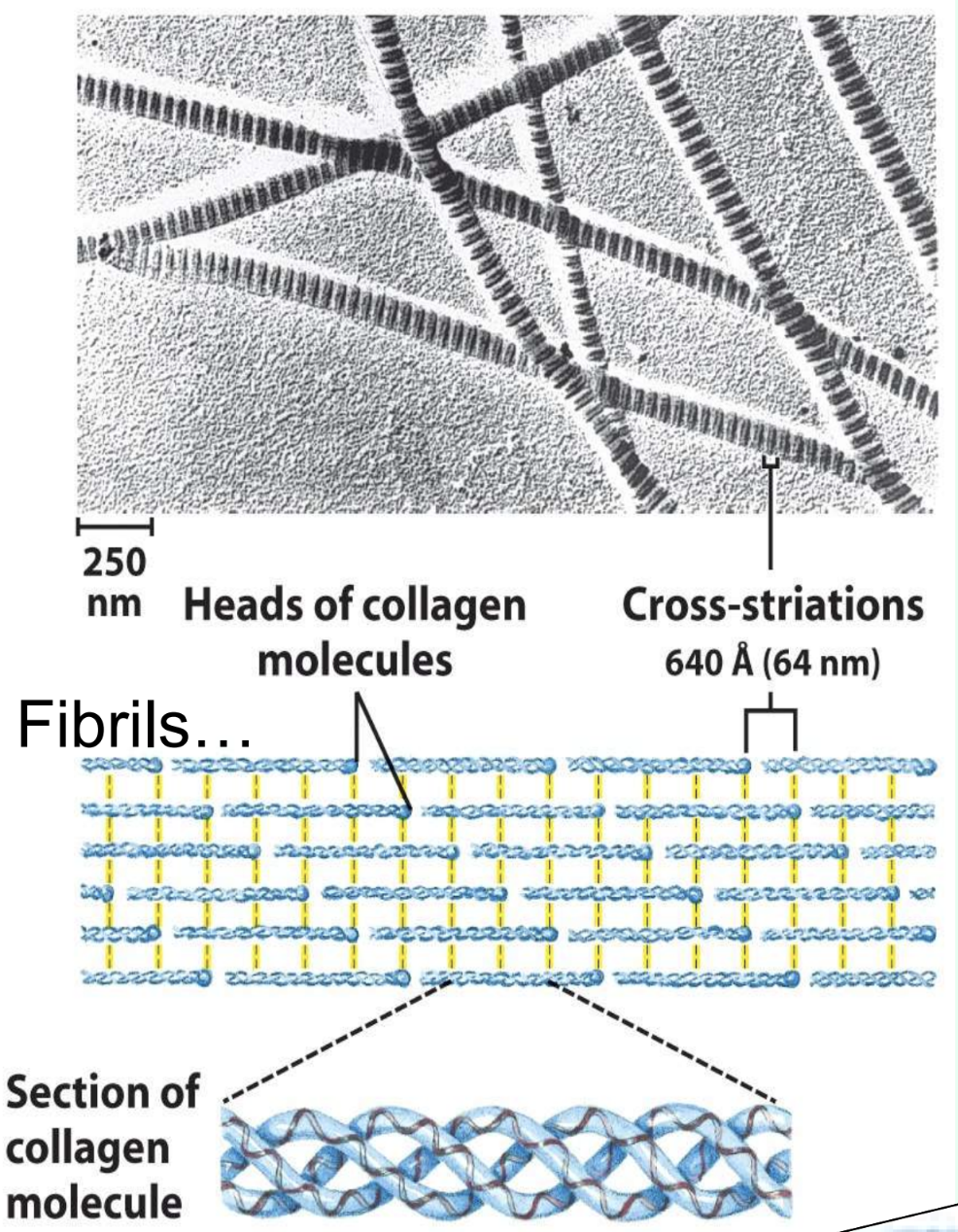
What stabilizes collagen fibrils?
Unusual interchain cross-links between Lys, 5-hydroxylysine, and His
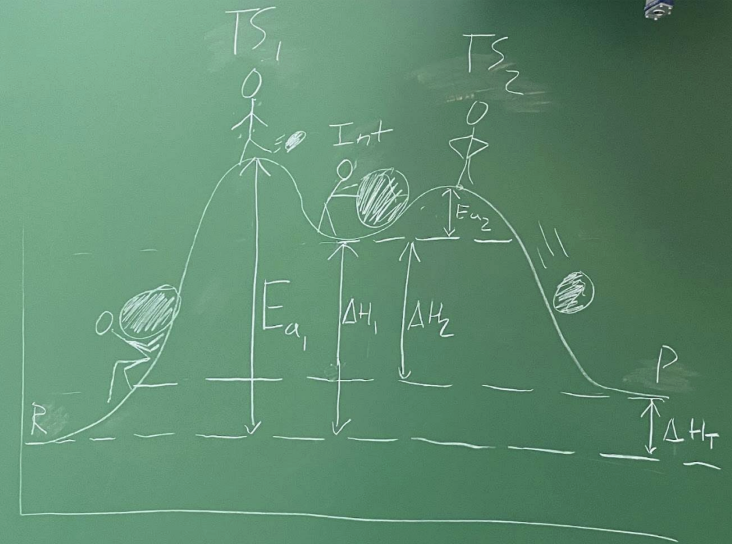
What is Gibbs free energy (G)?
Thermodynamic potential reflecting internal energy + disorder
Related to stability/reactivity
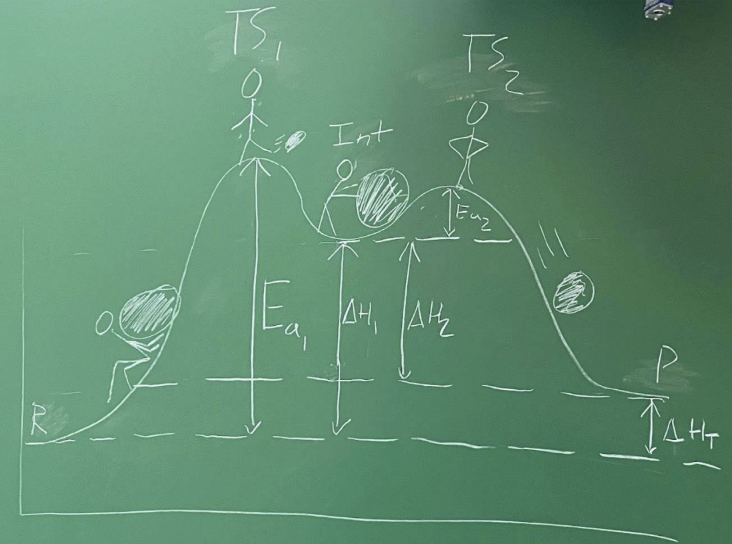
What does ΔG determine? And what are the units?
Reaction direction
Equilibrium position
Useful energy available for work
J/mol or cal/mol
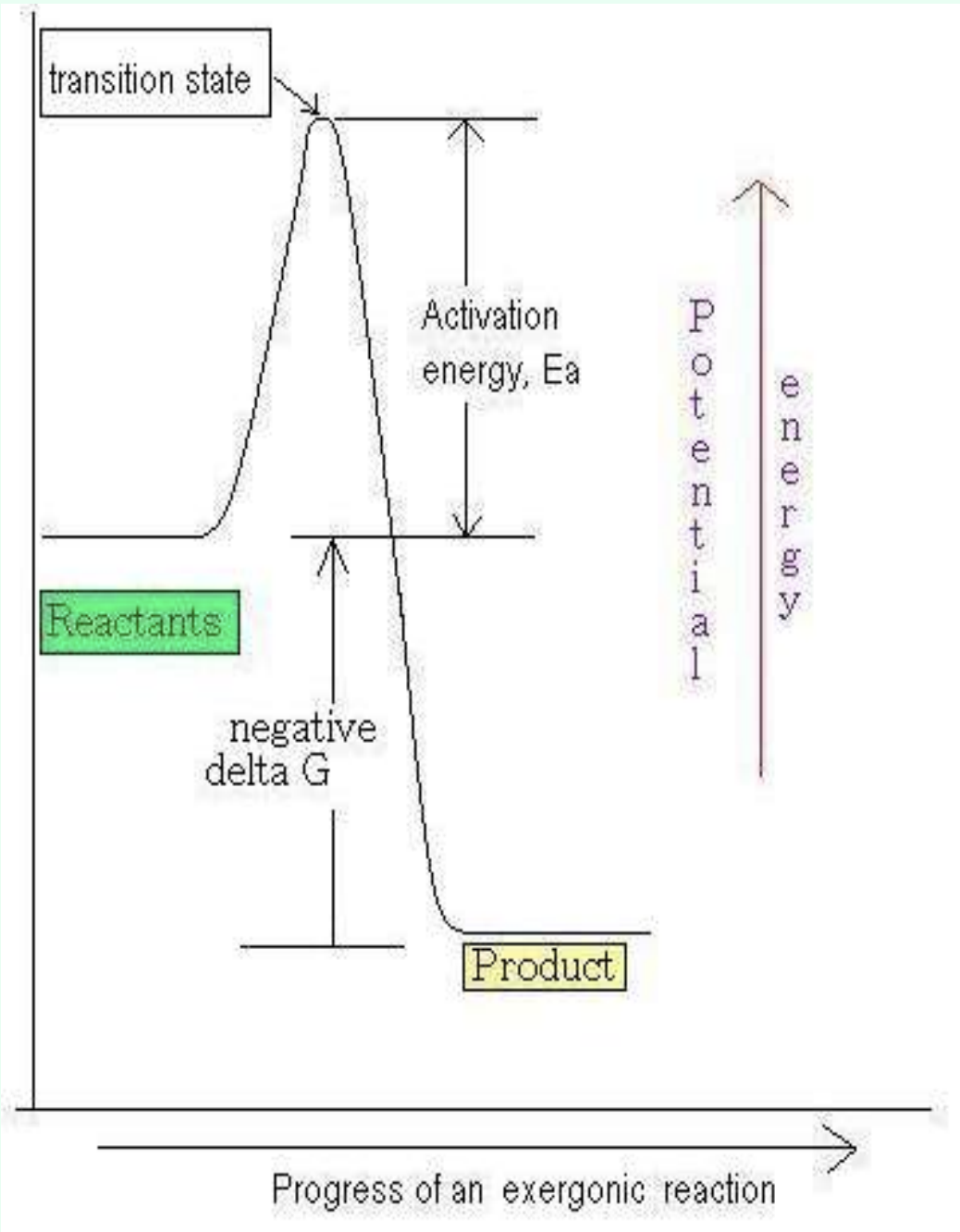
What does negative ΔG mean?
Exergonic reaction
Releases energy
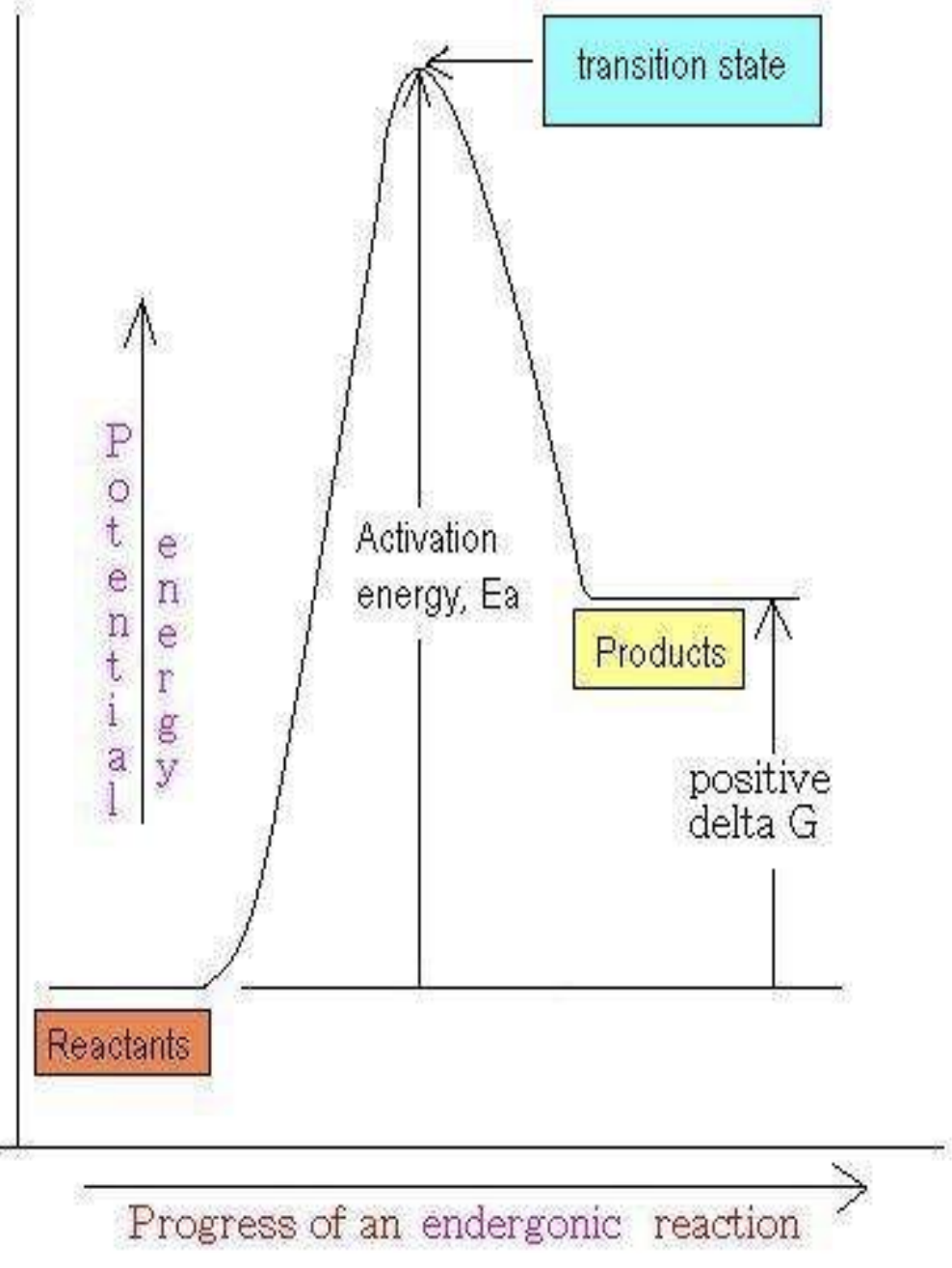
What does positive ΔG mean?
Endergonic reaction
Requires energy input
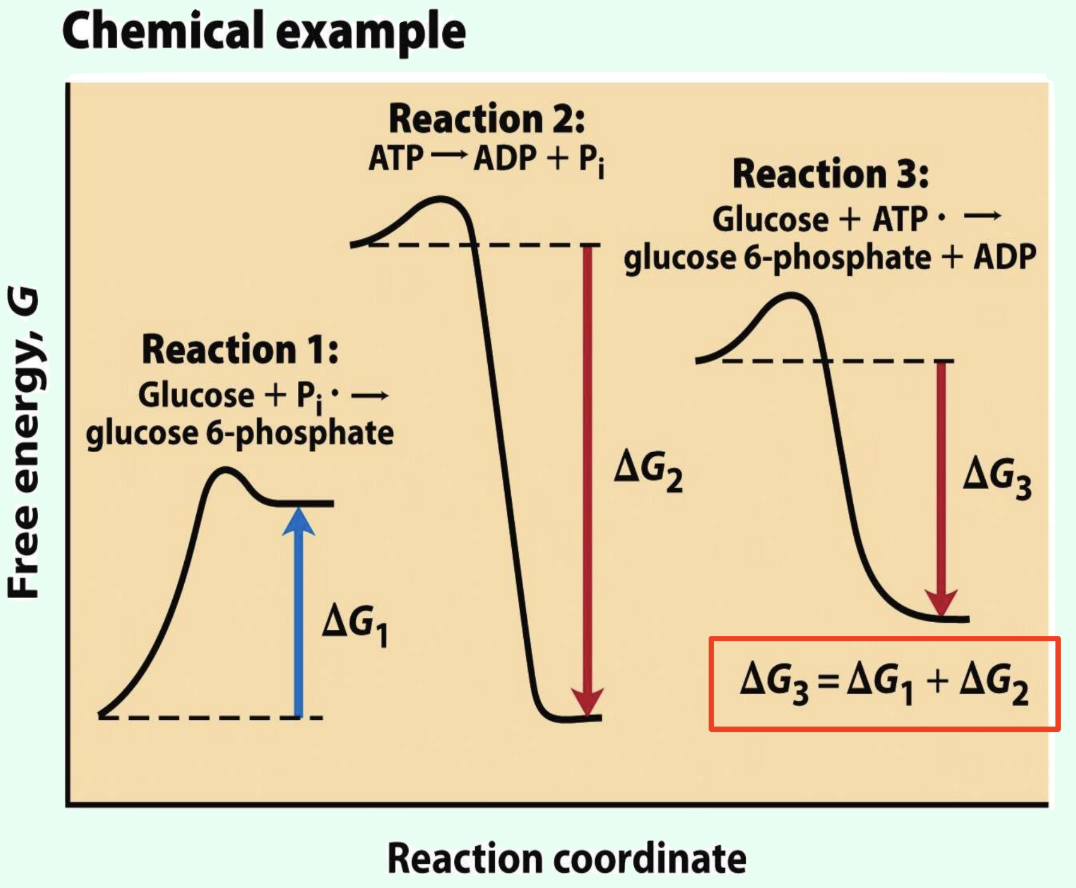
What does it mean that ΔG values are additive?
Coupled reaction ΔG values sum together
What is ΔG°′?
Free energy change under biochemical standard conditions
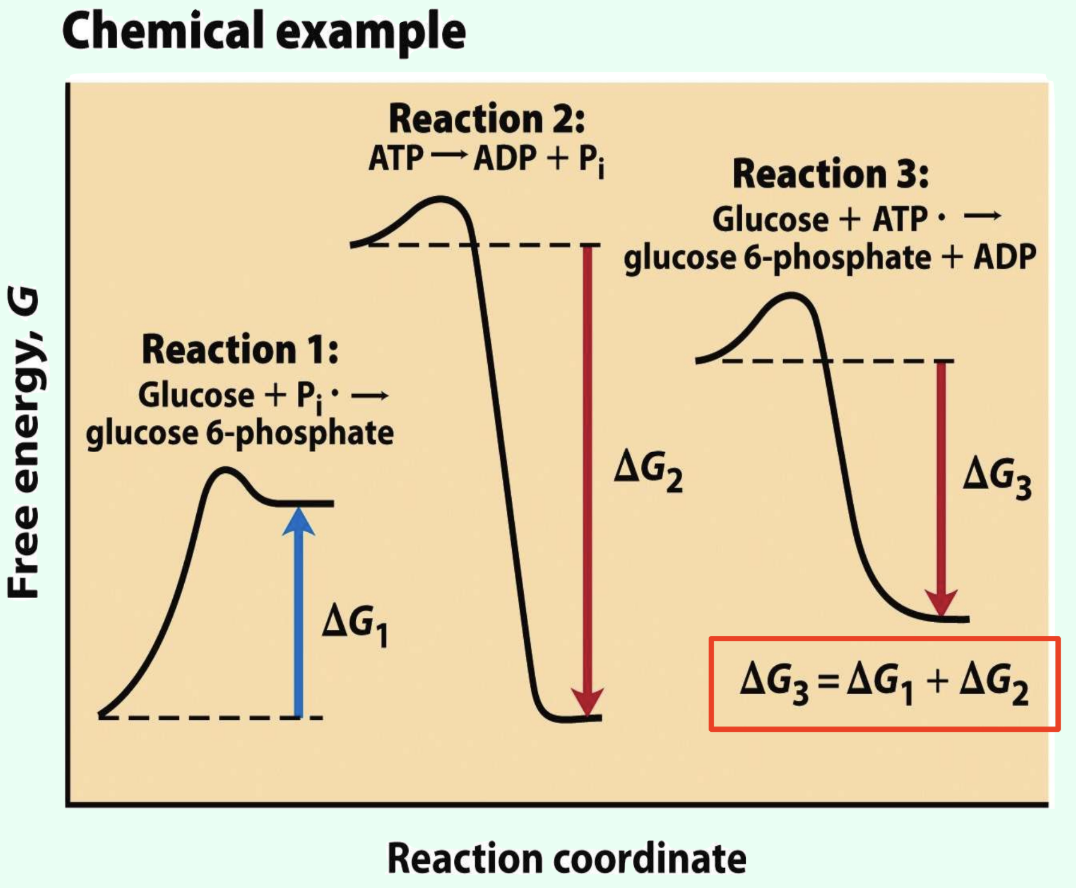
How do cells drive unfavorable reactions?
Coupling positive-ΔG reactions to highly exergonic reactions
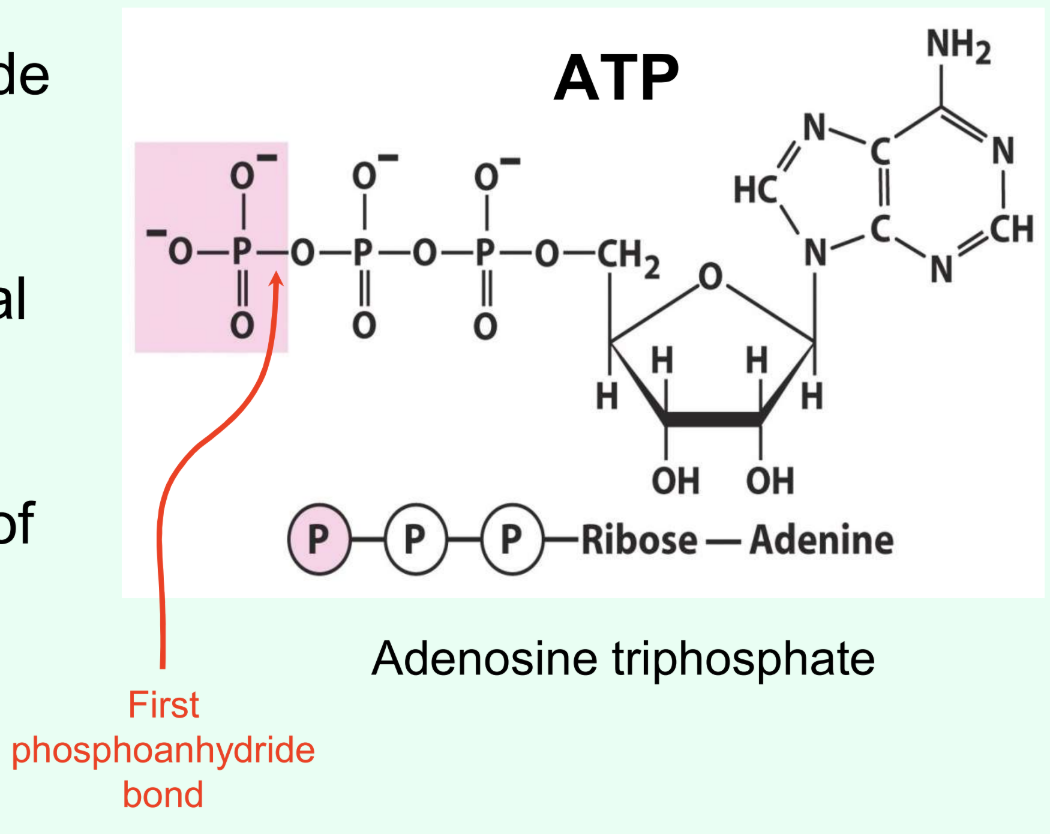
What ATP bond hydrolysis is highly exergonic?
Terminal phosphoanhydride bond
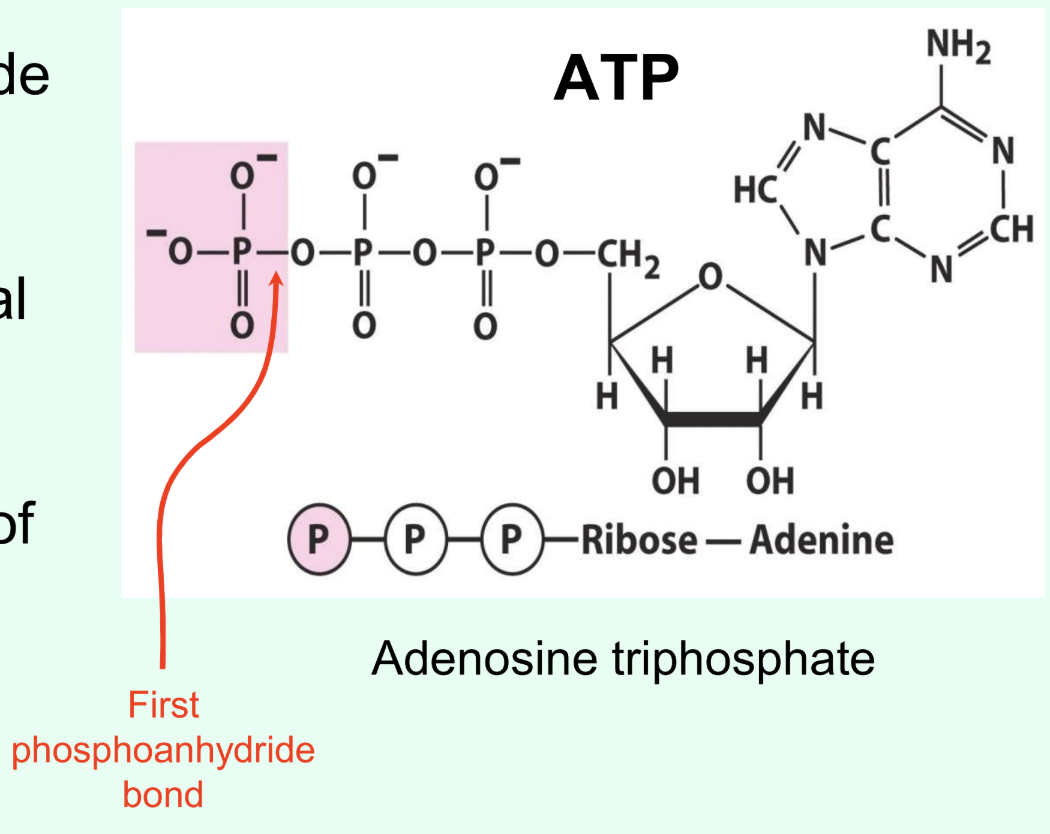
Why is ATP used as cellular energy currency?
ATP hydrolysis provides free energy to drive many metabolic reactions, like protein synthesis
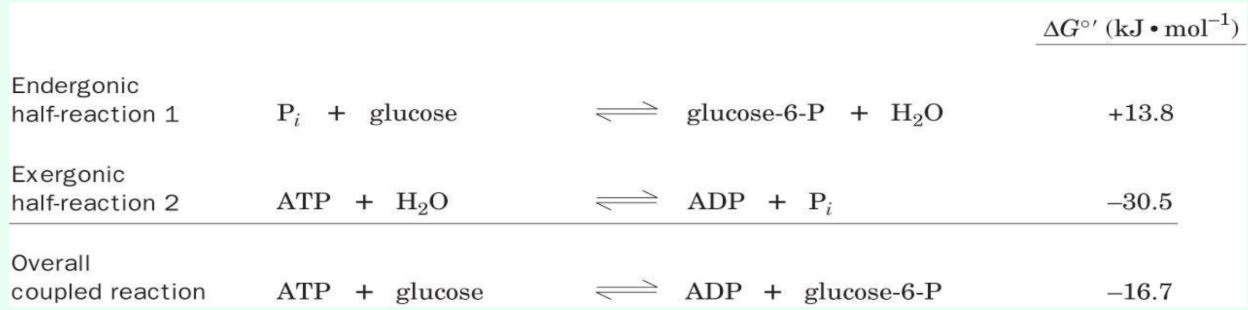
What is required for reaction coupling?
A shared intermediate
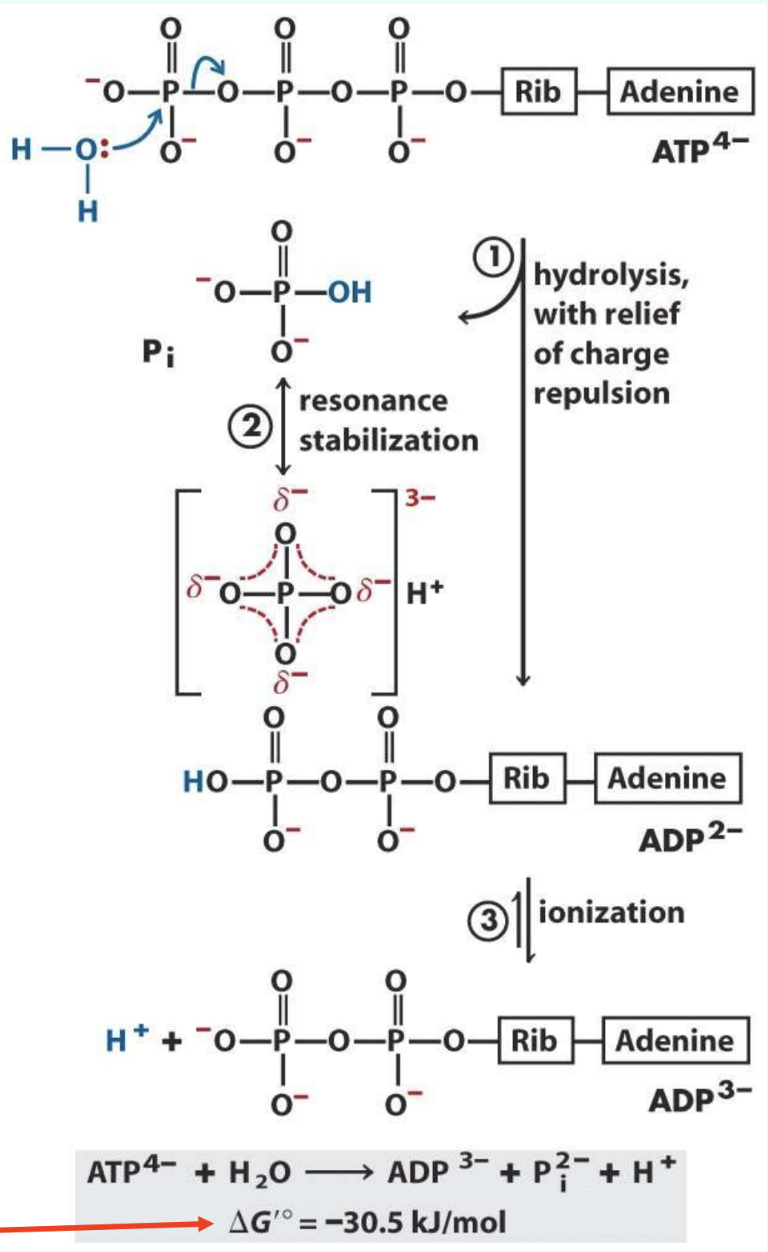
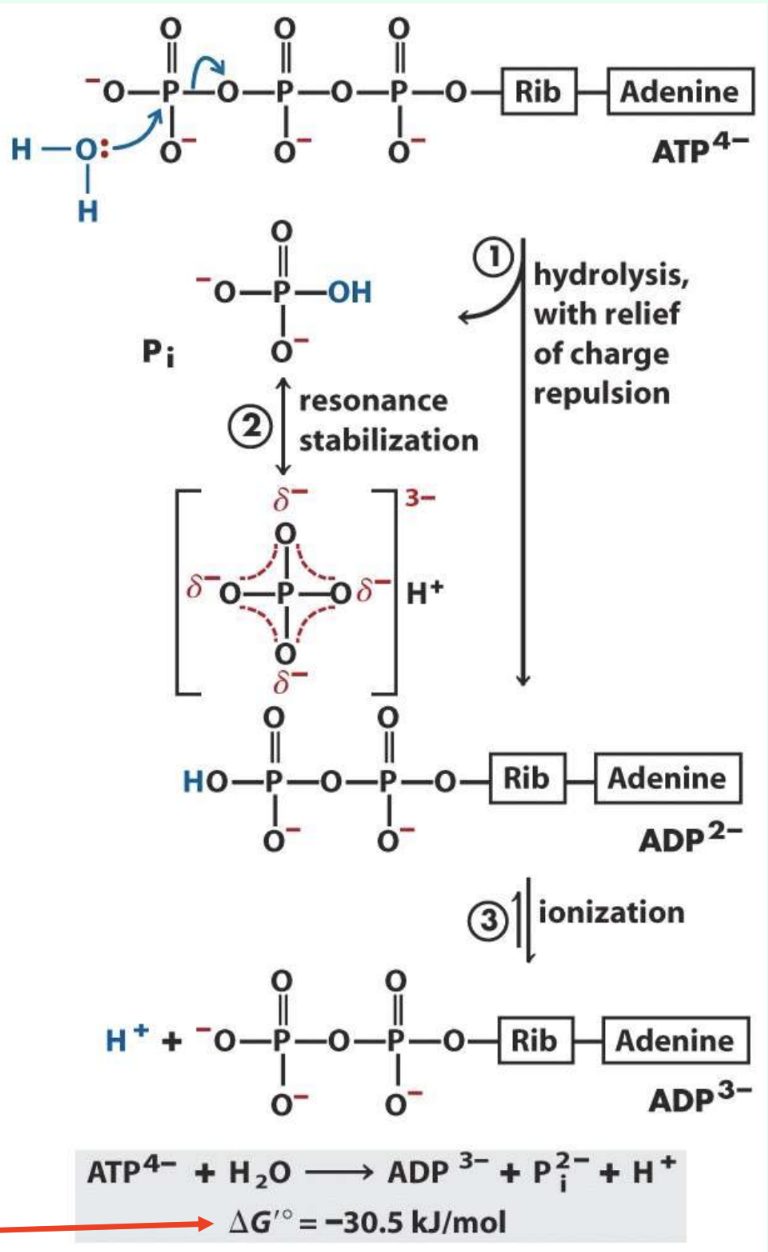
Why is ATP hydrolysis strongly negative?
Relieves charge repulsion
Pi resonance stabilization
ADP ionization
Better product solvation
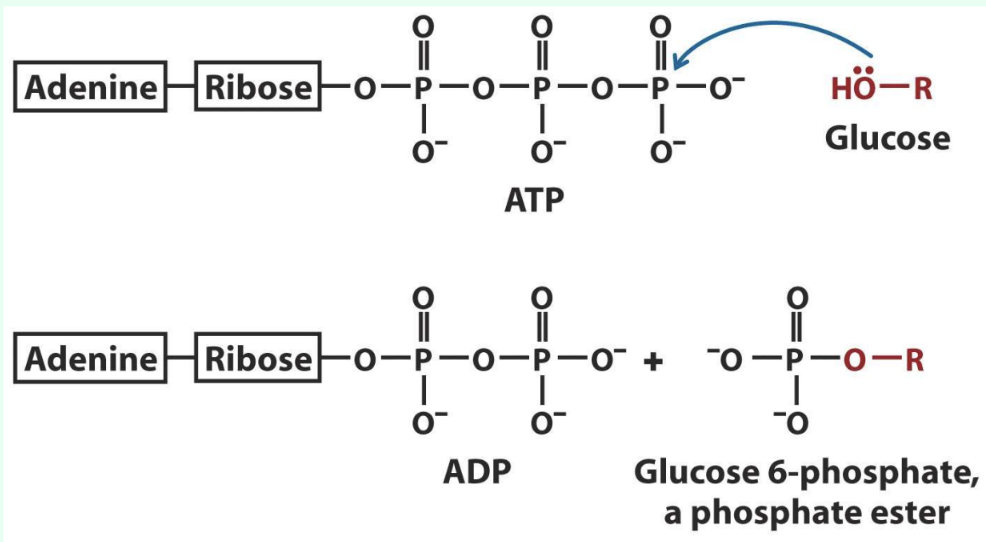
What is phosphoryl transfer?
Transfer of a phosphoryl group to an acceptor
Main way chemical energy is transferred in metabolism
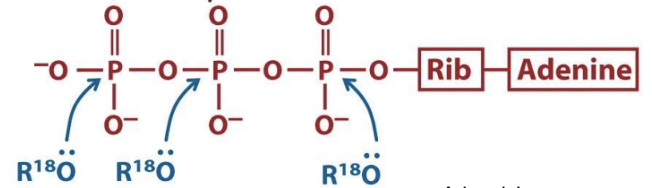
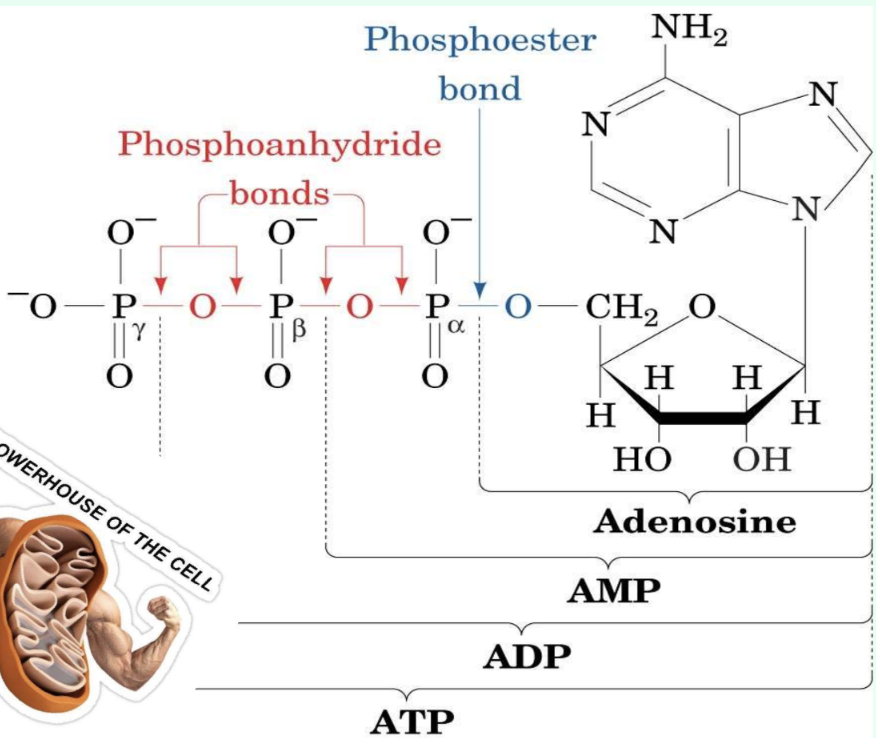
What are the three ATP attack positions?
γ = phosphoryl transfer
β = pyrophosphoryl transfer
α = adenylyl transfer
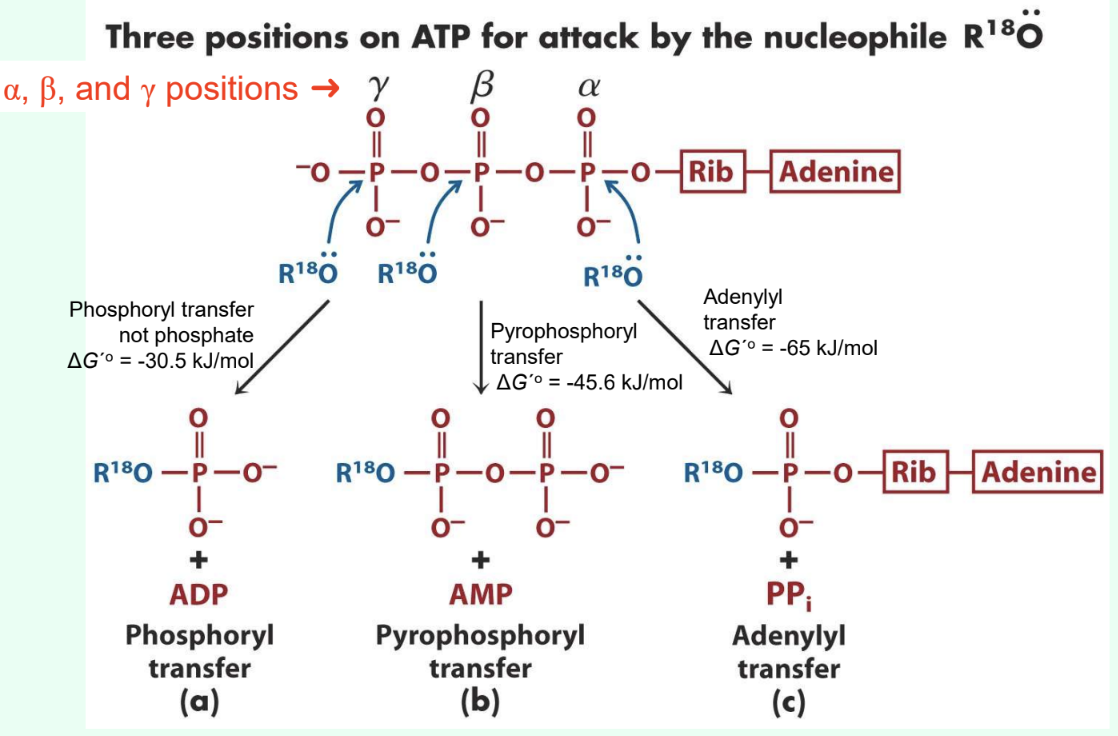
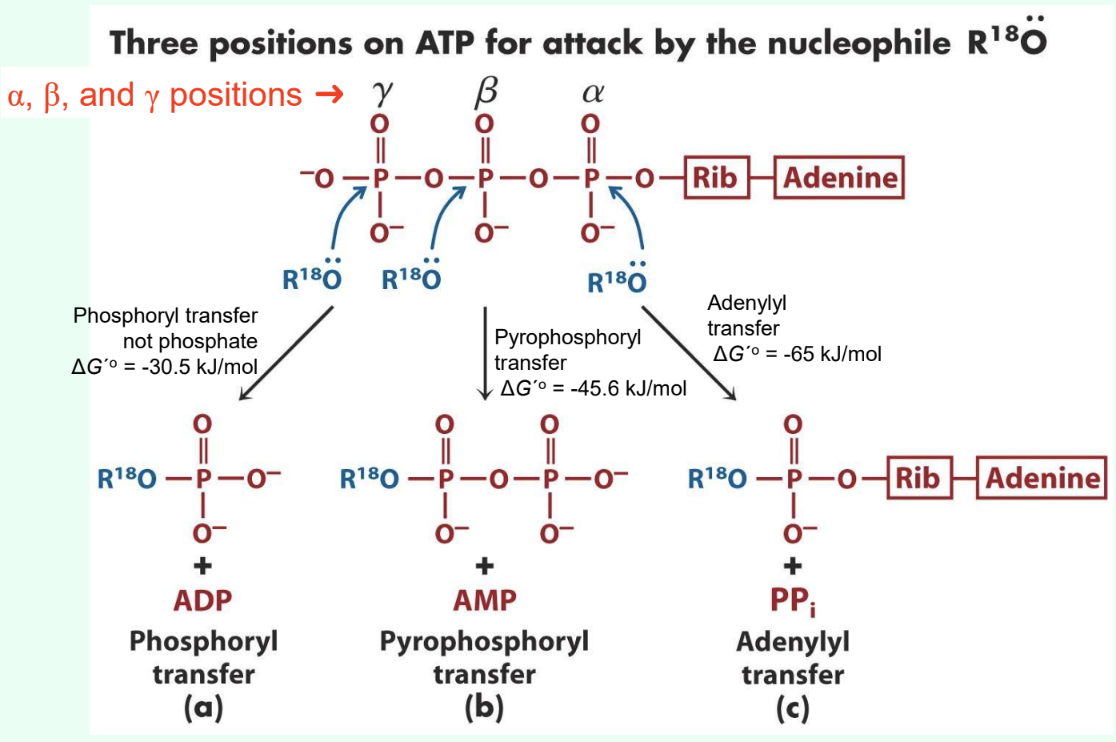
What does γ-phosphate attack transfer?
Phosphoryl group
Produces ADP
ΔG°′ ≈ −30.5 kJ/mol
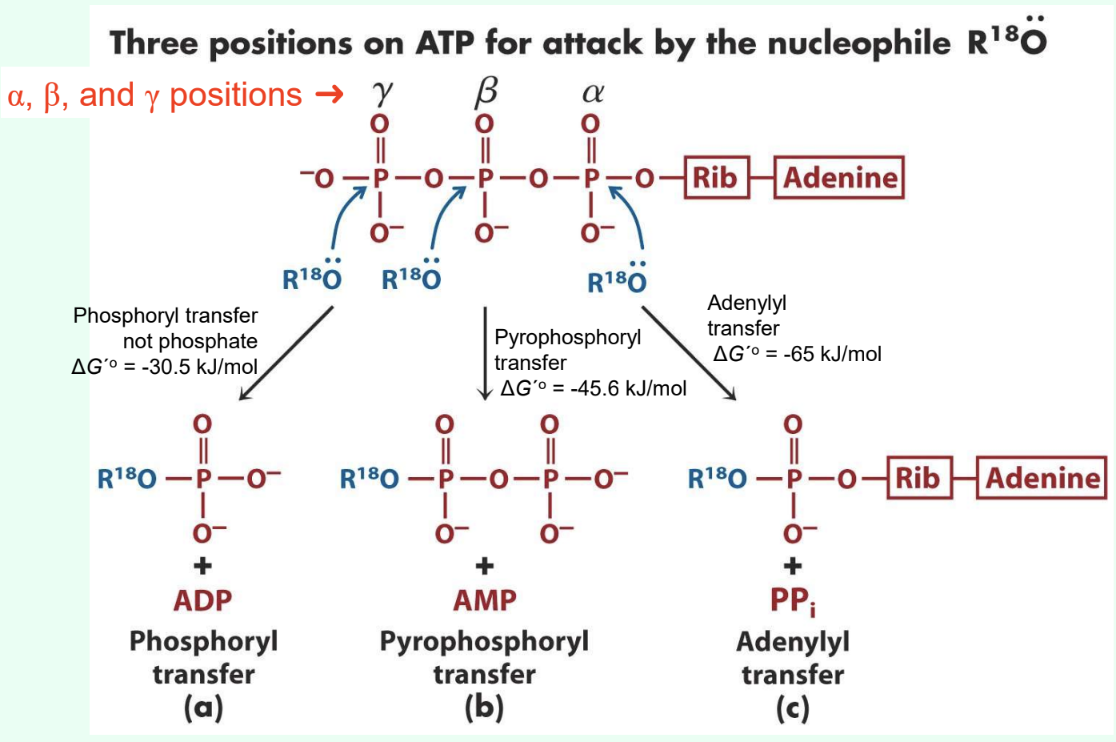
What does β-phosphate attack transfer?
Pyrophosphoryl group
Produces AMP
ΔG°′ ≈ −45.6 kJ/mol
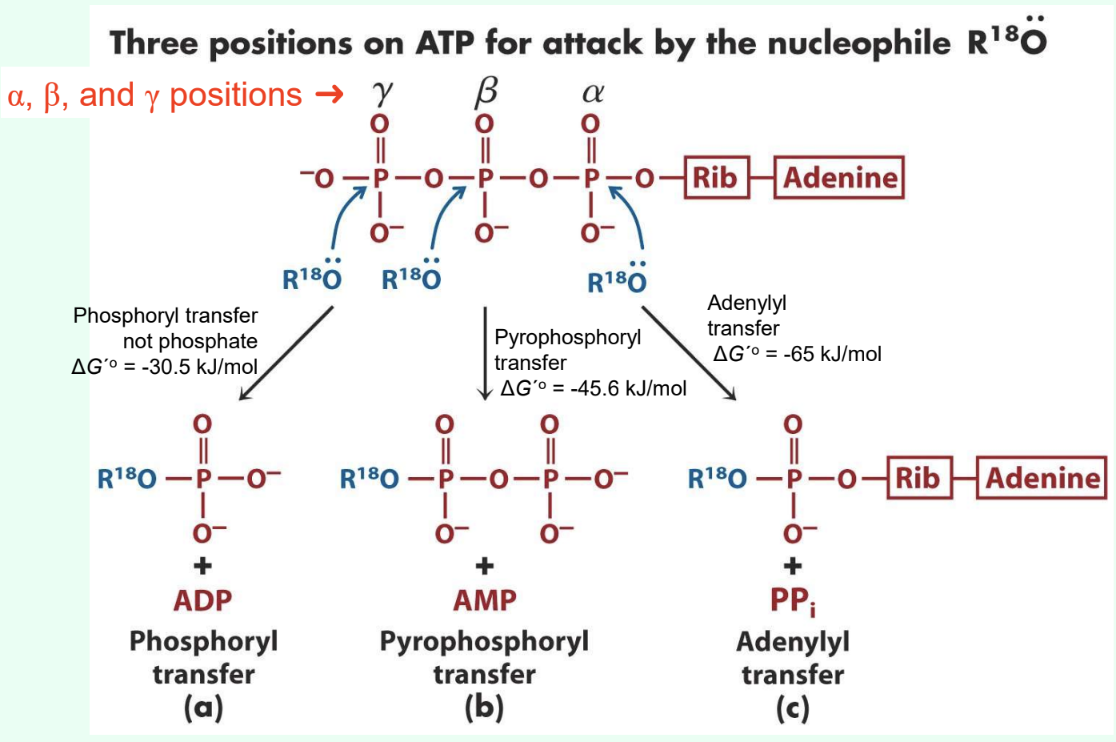
What does α-phosphate attack transfer?
Adenylyl group/5′-AMP
Produces PPi
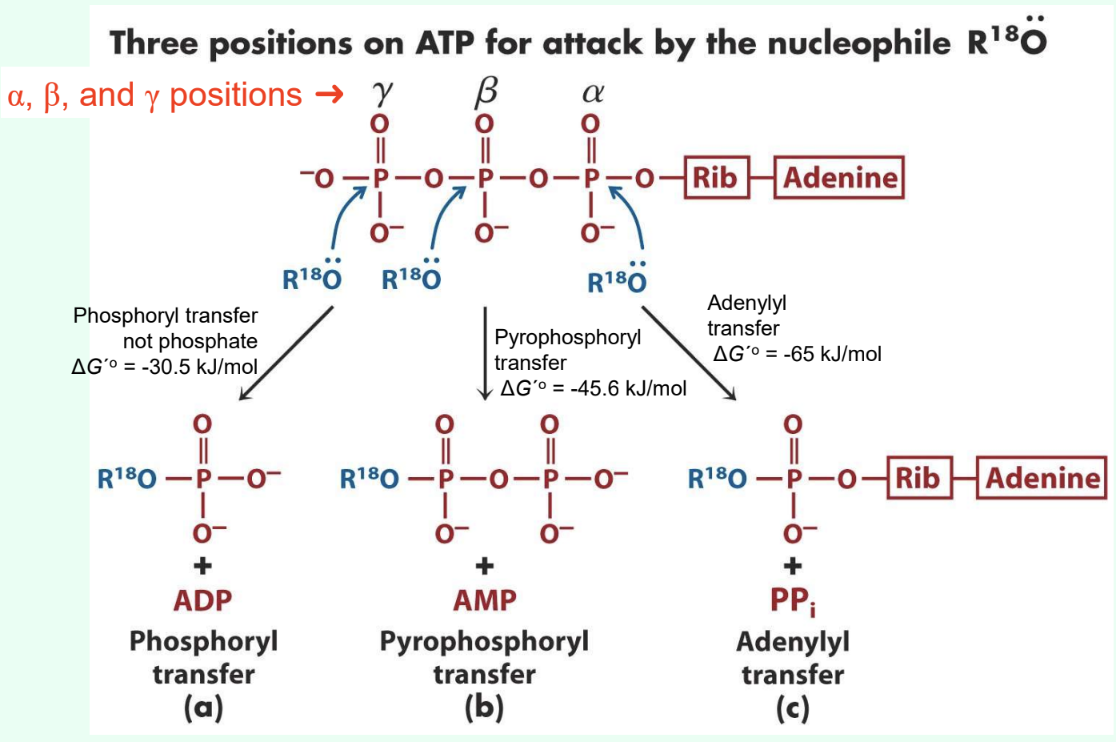
Why are adenylylation reactions highly favorable?
PPi hydrolysis drives reaction
Overall ΔG°′ ≈ −65 kJ/mol
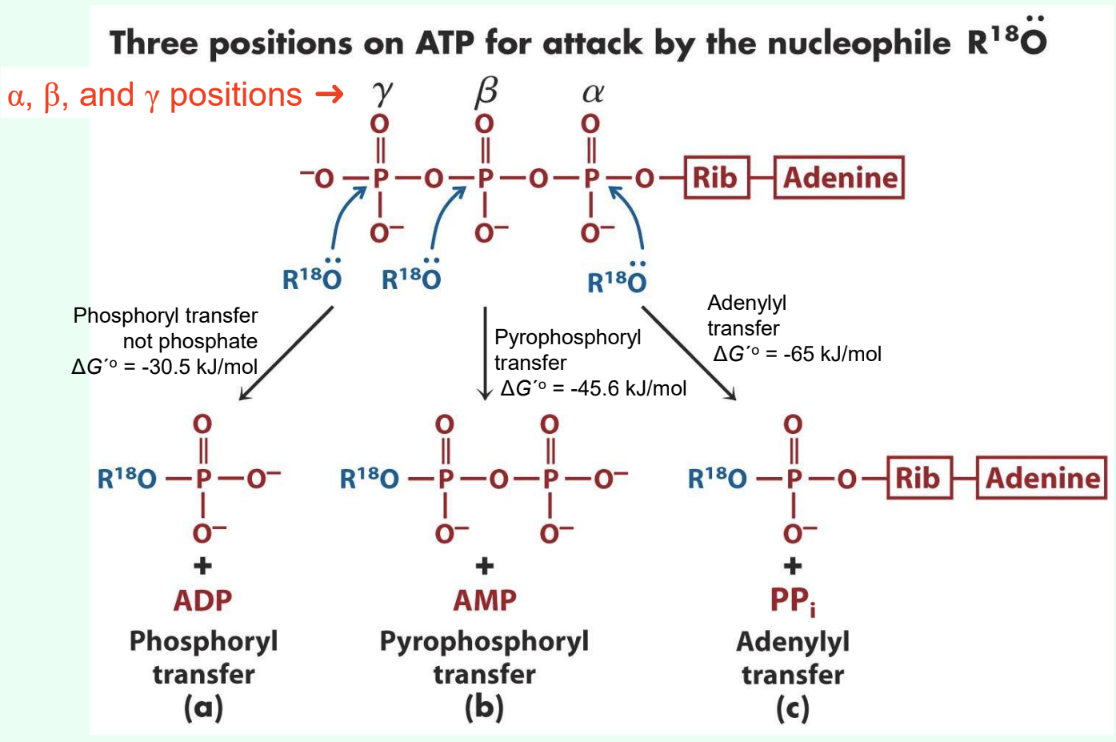
Why are adenylylation reactions useful?
Often coupled to highly unfavorable metabolic reactions
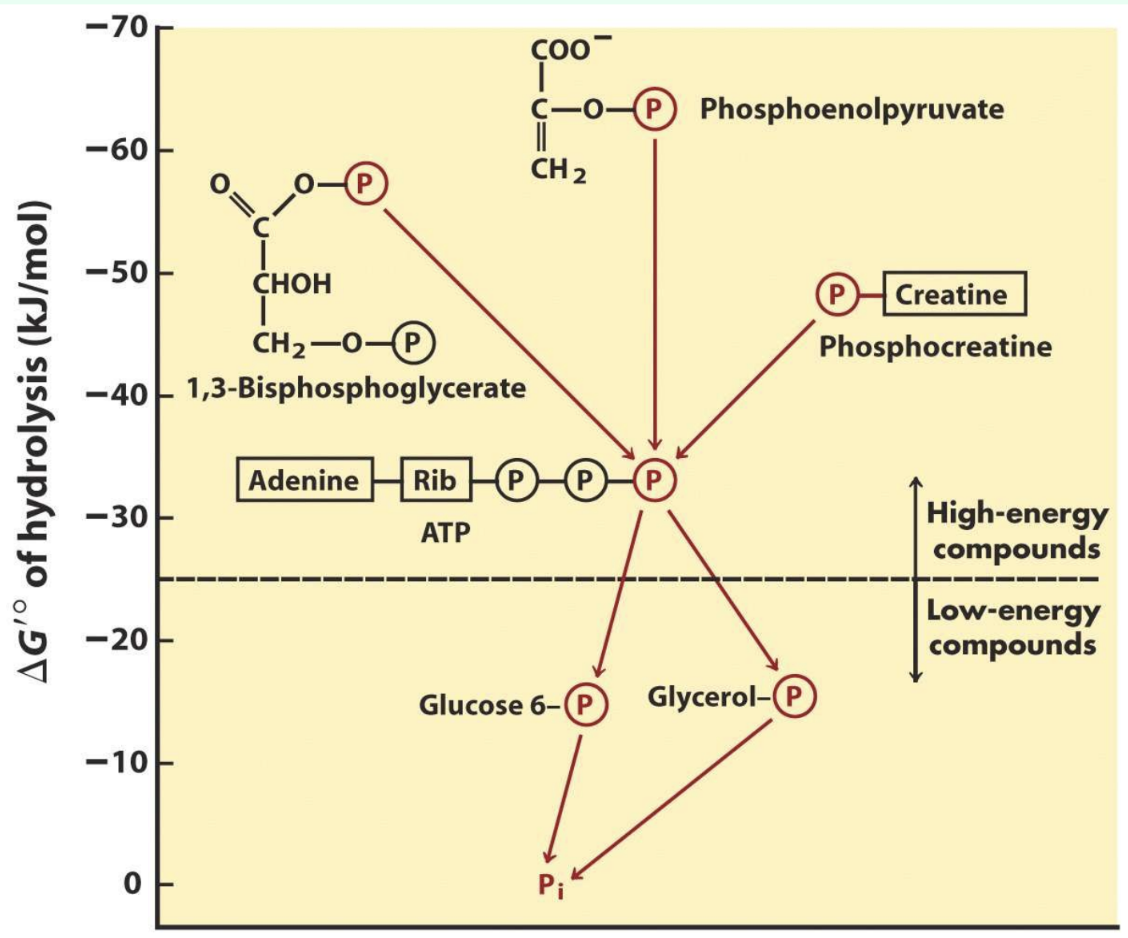
Why is ATP special among phosphorylated compounds?
Intermediate phosphorylation potential allows energy transfer from high-energy compounds to lower-energy compounds
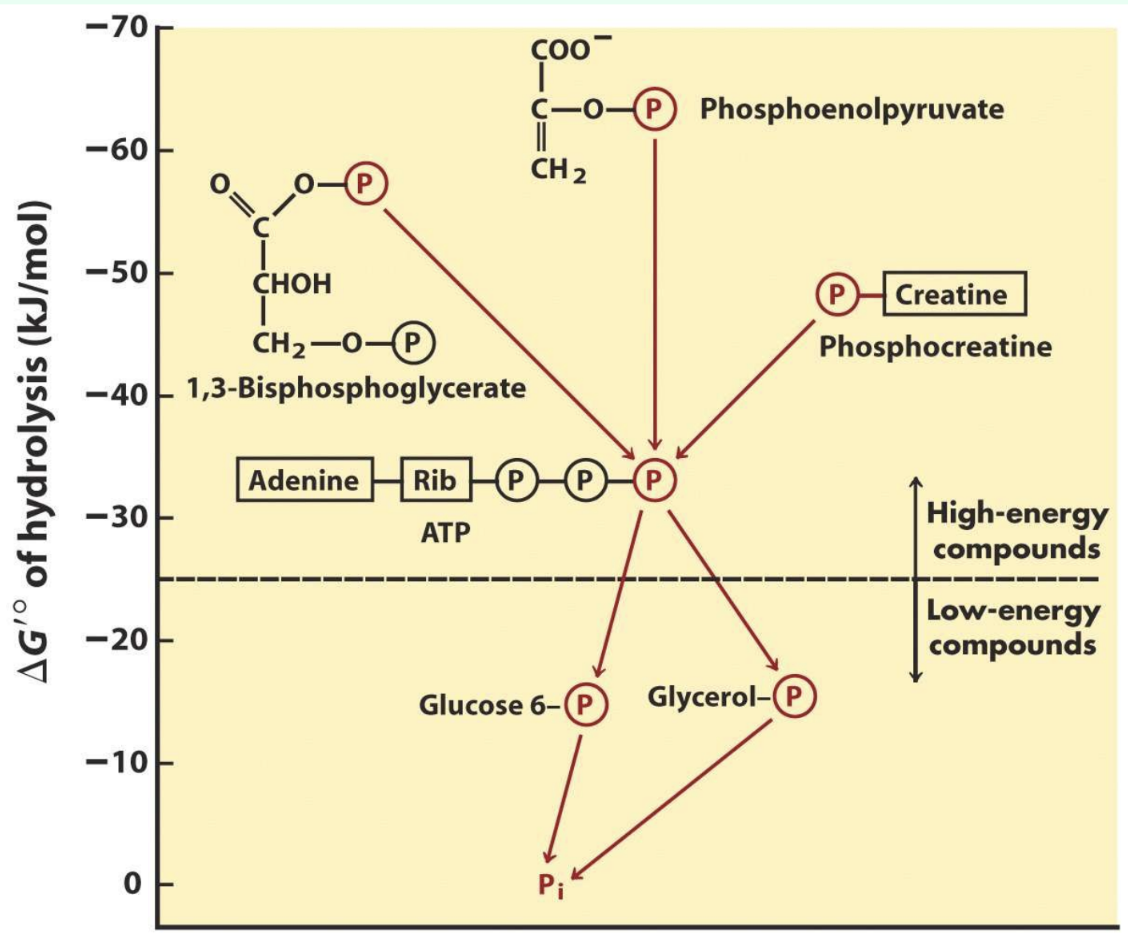
Why doesn’t ATP spontaneously phosphorylate molecules?
ATP is kinetically stable
Enzymes are needed
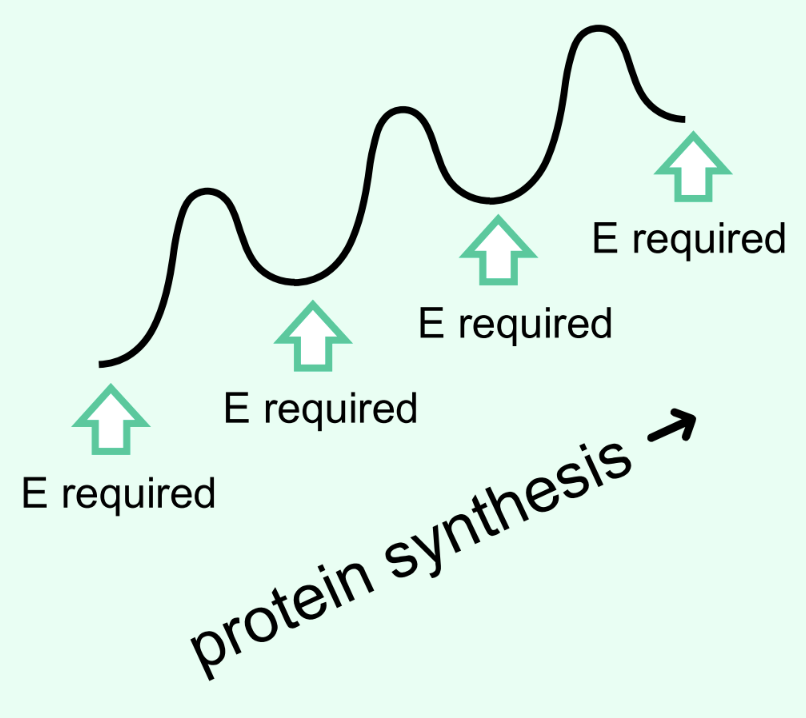
How is metabolism different from combustion?
Metabolism releases energy stepwise so cells can capture/use it;
Combustion releases energy all at once