Rational Drug Design Part 1
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What are lipinski rules state (4). Exceptions?
Poor adsoprtion or permation are more likely when?
>5 H-bond doners (sum of all OHs and NHs)
MW > 500
Log P > 5 (or MlogP > 4.15)
> 10 H-bond acceptors (sum of all Ns and Os
Exception: substrates for biological transporters
What is Log P? Log D?
Log P = log (compiund orgnanic/compond aqueous). at neutral pH (7.4)
Log D (is at a specific pH). (u have to include the ionized and unionized portions)
How is lipophilicity of a compound commonly estimated
Log P from otactonol/water partitionitong
Lipophilicyt is a major determinant of many ADME/Tox peroetpeis
Why octanol
lenght of the cell memrbnae. thicknes sof the bilayer mickers the thicknes sof hte cell membrnae
What factors affect logP
Molercular volume
Dipolarity
Hydrogen bond acidity
Hydrogen bond basicity
What is the impact of LogD7.4 on drug-like properties
When Log <1, log = 1 to 3, 3 to 5, > 5?
Solubility
Permeability
Metabolism
Volume of distribution
Oral adsorption and BBB penetration
Renal clearance

How many HBD does NH2 have
2
How many hydrsogen bond acceptors does COO have
2
What happens when u get higher potency
You get worse oral adsorption
What does Vebers rules define
Rules for good bioavilability
<= 10 rotatable bonds
<= 140 A PSA or <= 12 total hydrogen bonds (acceptors + donors)
PSA
Polar surface area
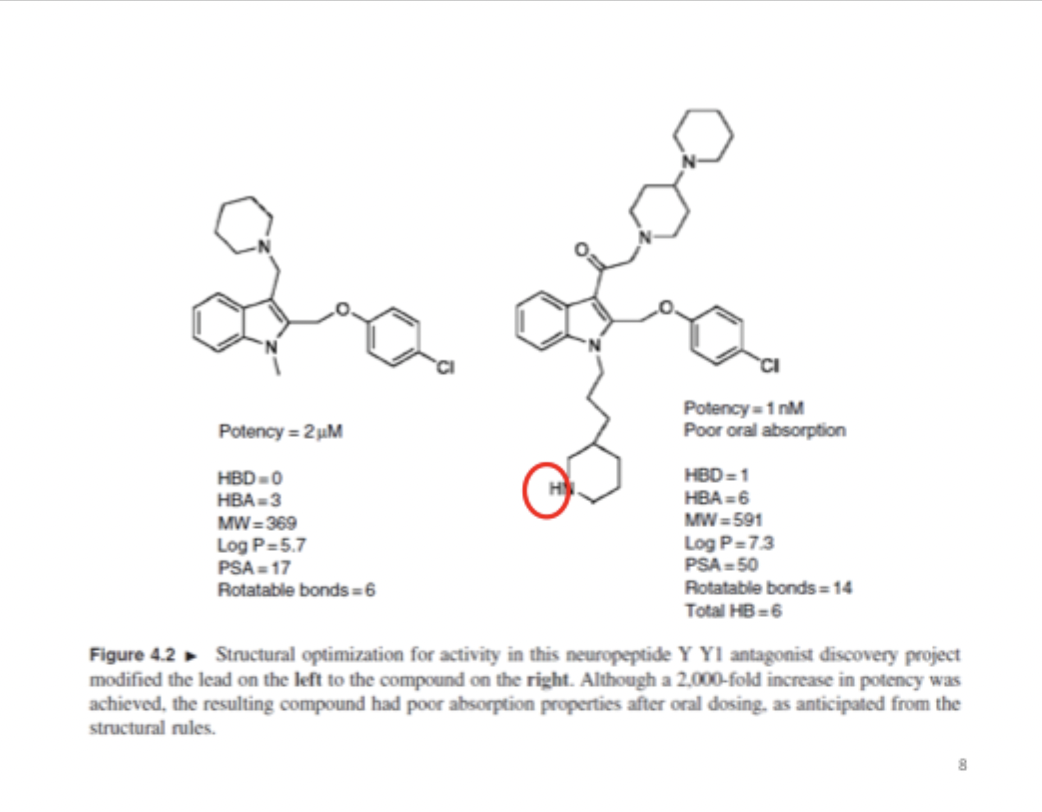
does this compound have good bioavailability according to vebers rules
What are the properties of lead-like compounds

whats template conservation
in order to maintain lead-like compound properties, the researchers apply template conservation. The lead template is conserved during lead optimization
What is the rule of 3
Set of rules for lead like compounds. Guidelines for hte propertie sof molecules used in fragment based screening techniques.

Rank the bonds
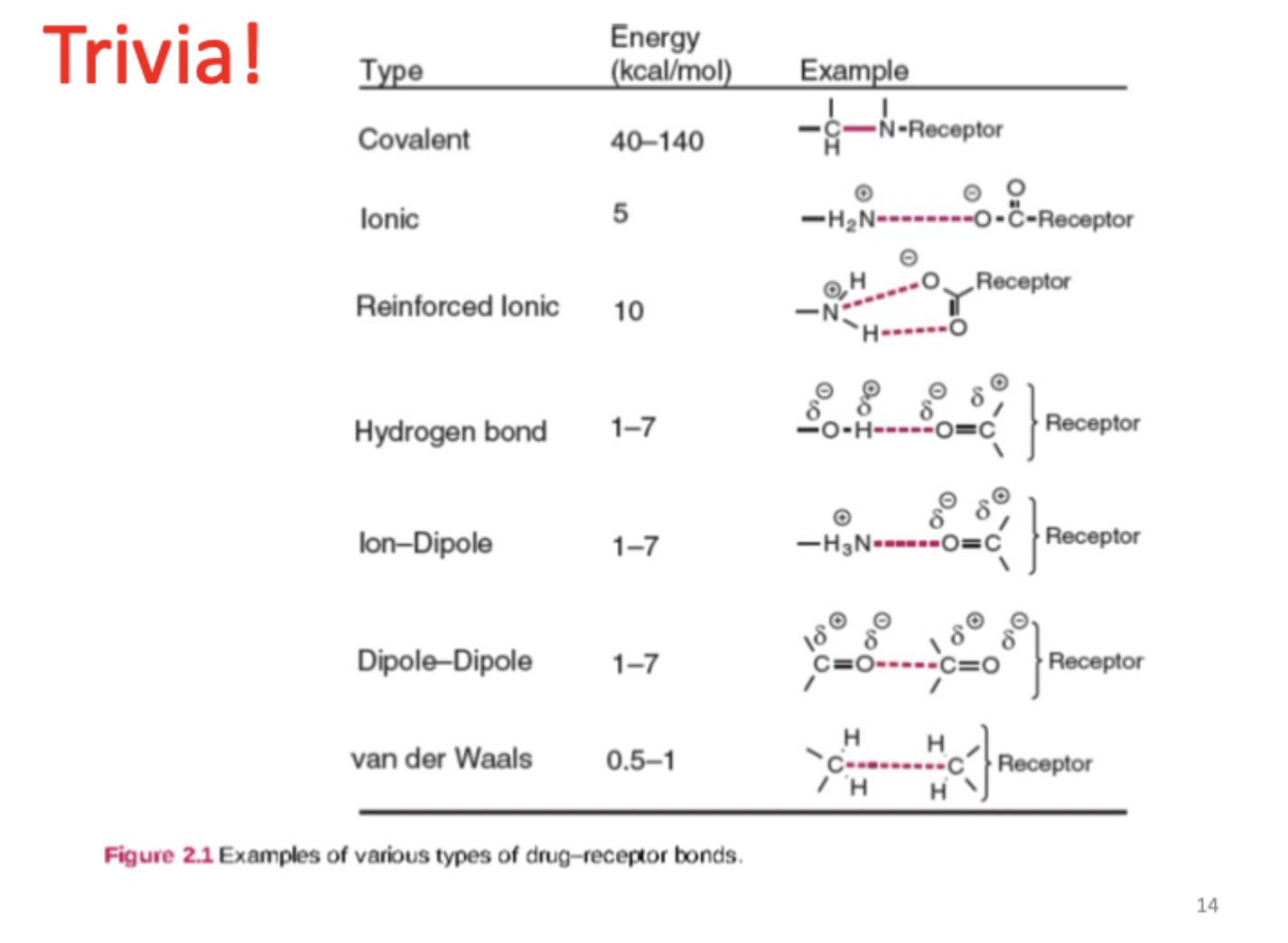
What is imatinib mesylate?
Gleevec
Wht are the steps for De novo desing
`1. Interaction sites
2. Fragment fitting
3. Bridging
Anchor and grow algorithm
Identify rotatable bonds
Identify and dock a rigid anchor
Add molecular fragments in layers
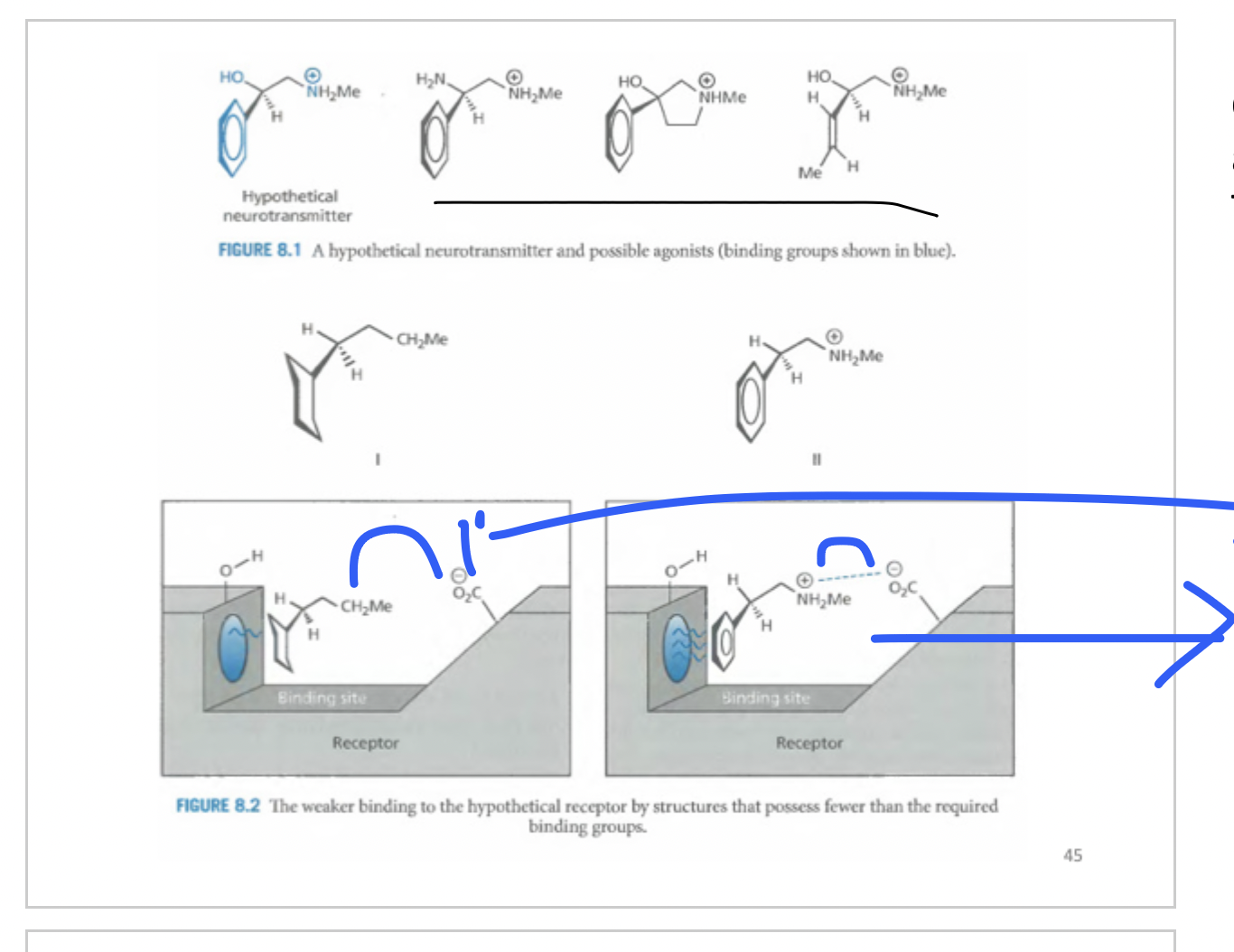
Whats happening here?
Ch2Me has no electrostatic interactions but far away, with NH2Me there is electrostatic interaction
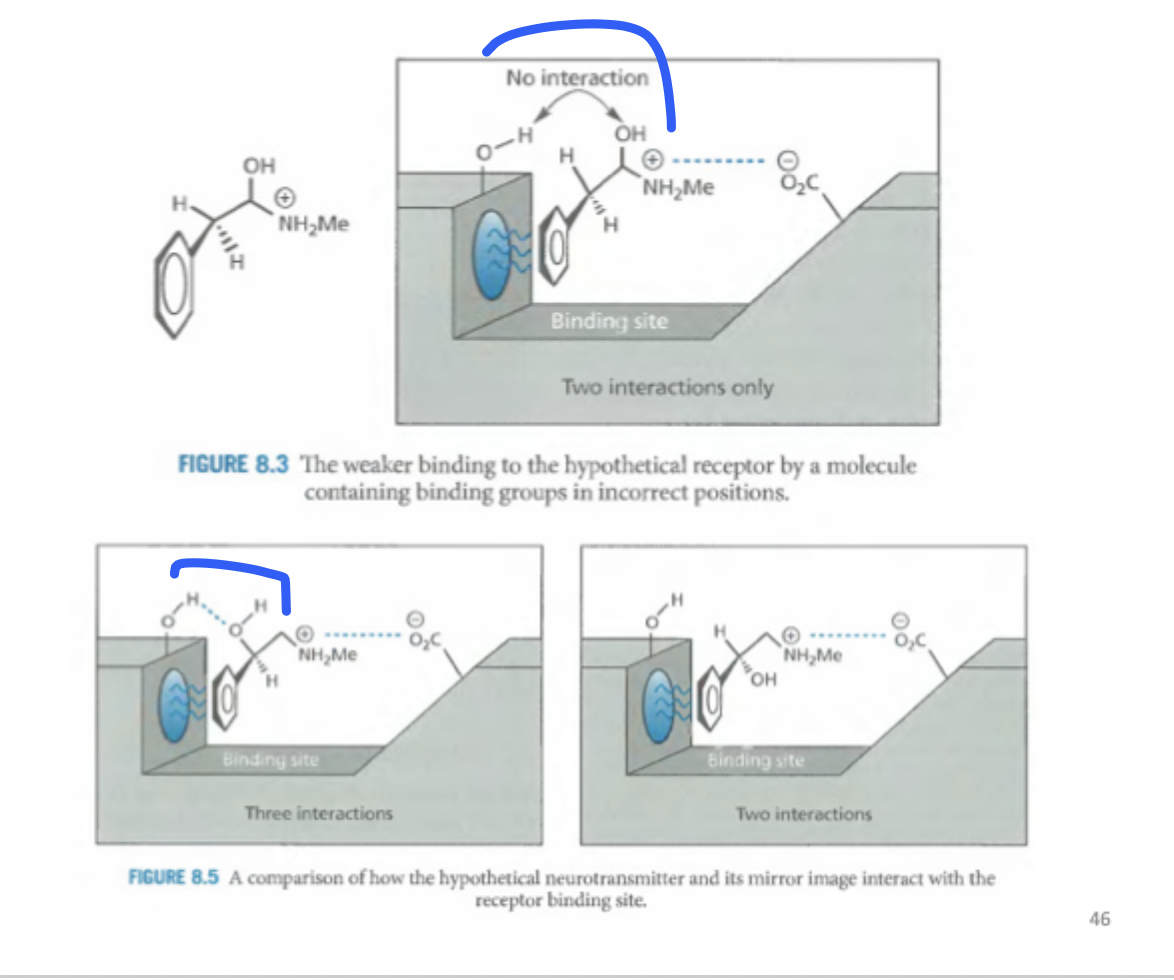
Whats happening here
The hydroxyl group is not close to the receptor. but if you take the miror image, we can establish 3 interactions
1) electrostatic
2) hydrogen bonding
3) aromatic hydrophobic grou
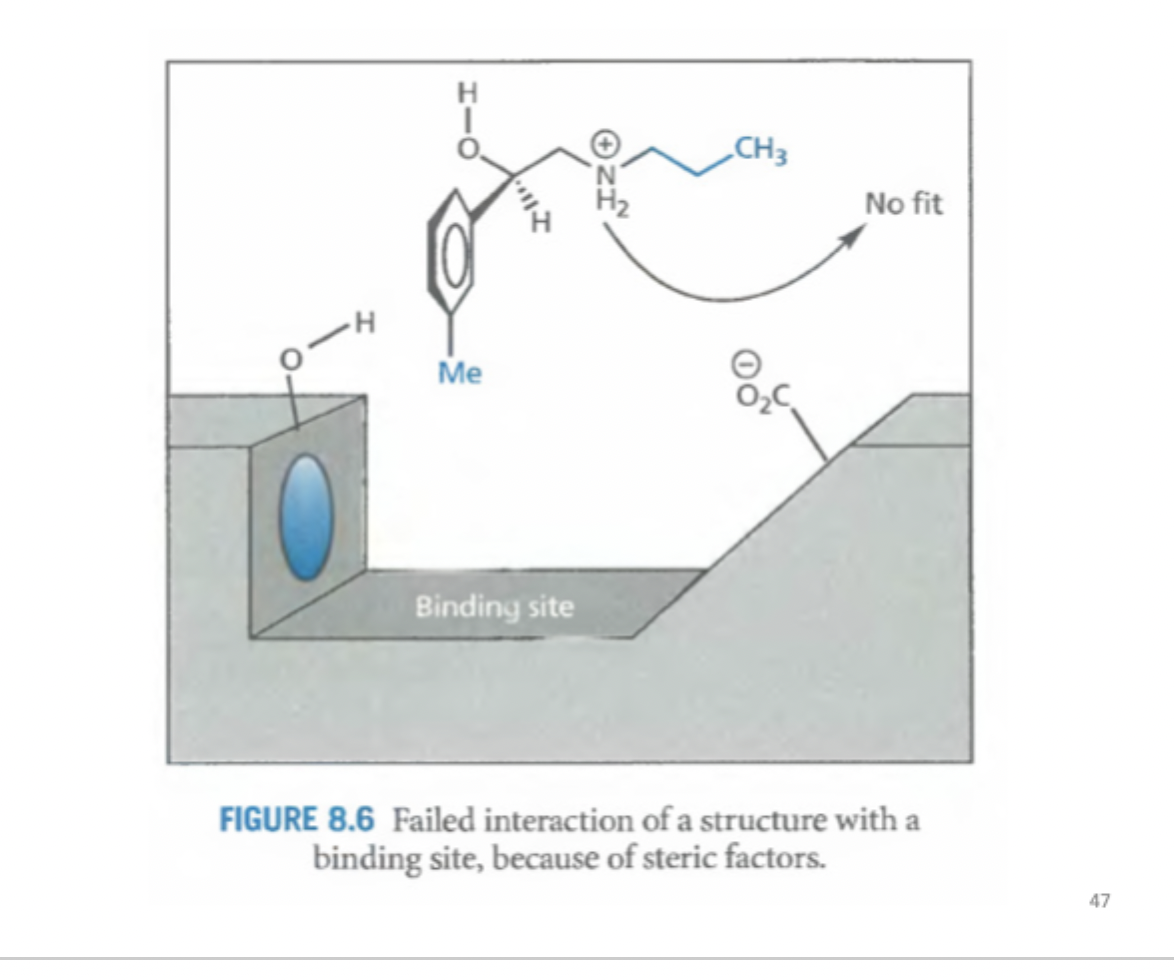
If you add an alkyl group → the long chain will not allow the. molecule to dock on the binding site
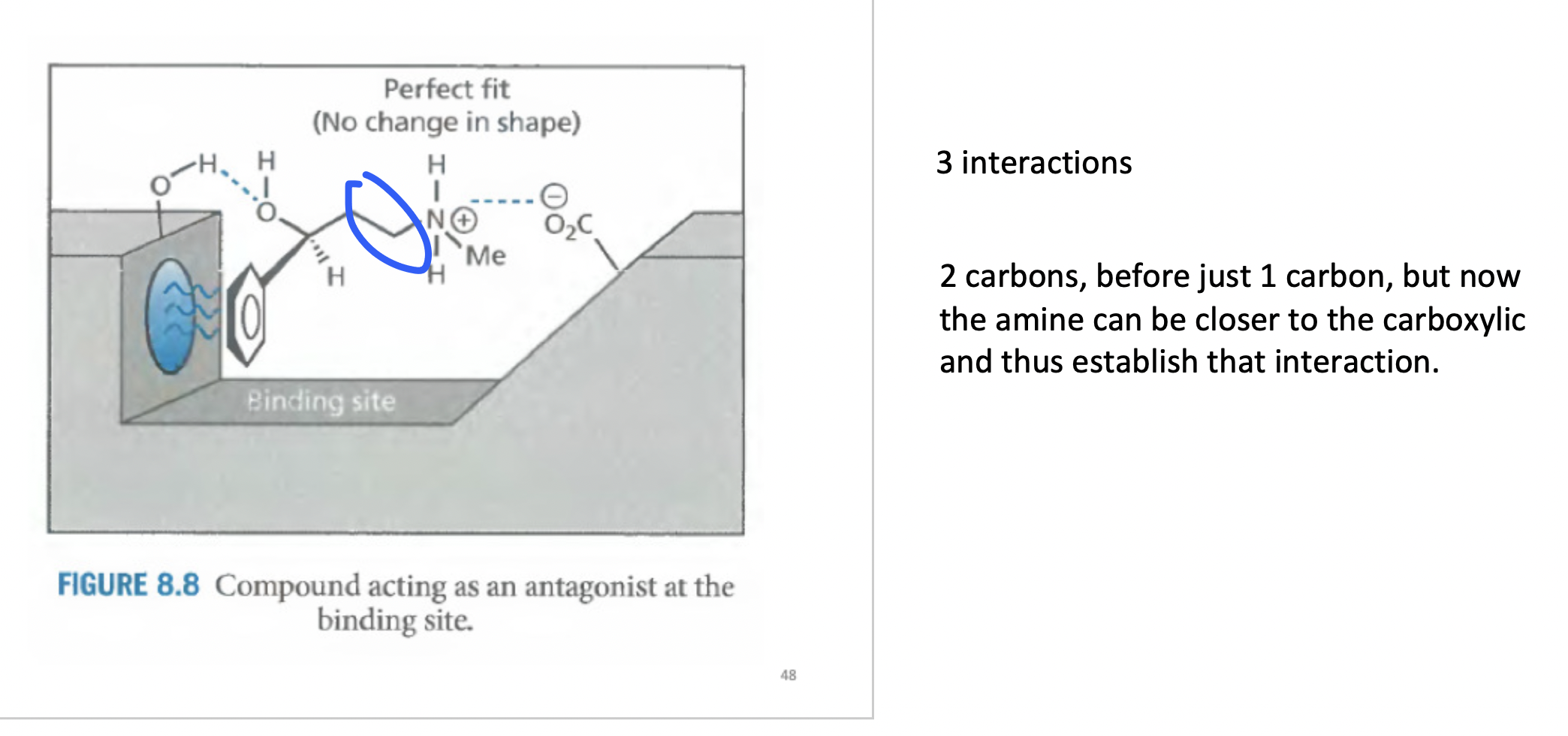
Perfect fit, the distance is just right