AQA A level Chemistry 3.1.1 Atomic structure
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Draw a labelled diagram of the structure of an atom. (3)
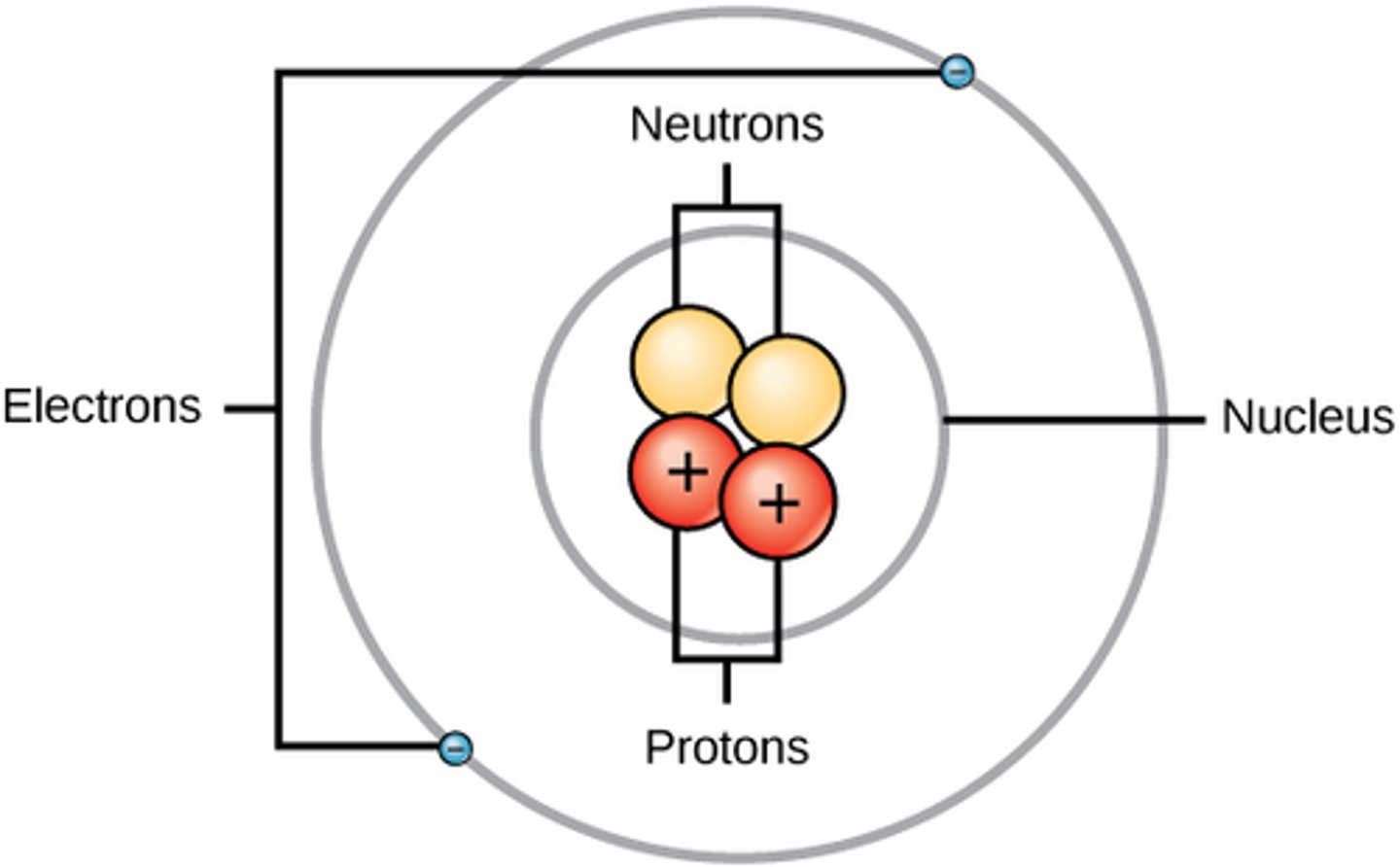
What is the relative mass of a proton? (1)
1
What is the relative charge of a proton? (1)
1
What is the relative mass of a neutron? (1)
1
What is the relative charge of a neutron? (1)
0
What is the relative mass of an electron? (1)
The relative mass of an electron is 1/1840.
What is the relative charge of an electron? (1)
-1
Where are protons and neutrons positioned within an atom? (1)
Within the nucleus of the atom.
Where are electrons positioned within an atom? (1)
Electrons are located in orbitals within subshells around the nucleus of an atom.
What are isotopes? (2)
- Isotopes are atoms of the same element with the same number of protons.
- They have different mass numbers due to varying numbers of neutrons.
What are the physical and chemical properties of isotopes? (2)
- Isotopes have slightly different physical properties because of their different masses.
- They exhibit similar chemical properties since they have the same electronic configuration.
What was John Dalton's early model of the atom? (2)
- In the early 19th century, John Dalton described atoms as solid spheres.
- He suggested that different spheres represented different elements.
What was J. J. Thomson's contribution to atomic structure? (2)
- In 1897, J. J. Thomson discovered the electron.
- This showed that atoms were not solid and indivisible, leading to the 'plum pudding model' of a positively charged sphere containing electrons.
What was Rutherford's gold foil experiment? (4)
1. In 1909, Rutherford and his students Geiger and Marsden fired positively charged alpha particles at a very thin sheet of gold.
2. Most alpha particles passed straight through, showing that most of the atom is empty space.
3. A few particles were deflected strongly, suggesting a small, dense, positively charged nucleus.
4. The plum pudding model was disproved and replaced by the nuclear model of the atom.
What was the key idea of Rutherford's nuclear model? (2 marks)
- The atom contains a tiny, dense, positively charged nucleus.
- This nucleus is surrounded by a cloud of negative electrons, with most of the atom being empty space.
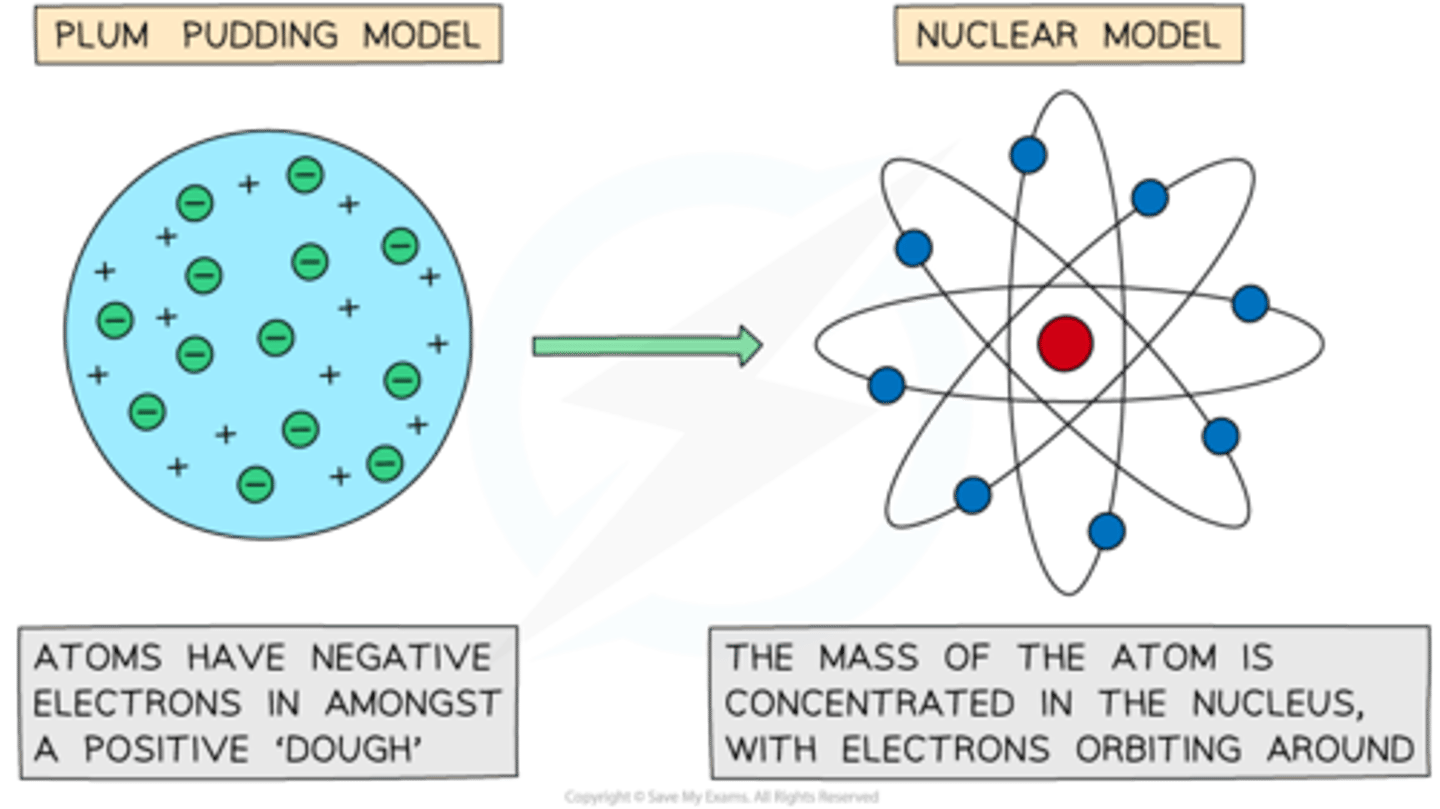
What was the problem with Rutherford's model and how did Bohr solved it? (3 marks)
- In Rutherford's model, electrons in a 'cloud' would spiral into the nucleus, making the atom collapse.
- Niels Bohr proposed that electrons exist in fixed orbits (shells) at specific energies.
- Movement between shells involves the emission or absorption of electromagnetic radiation of fixed frequency.
Why was Bohr's model was accepted? (2)
- Bohr's model matched experimental observations of radiation absorbed and emitted by atoms.
- This provided evidence that electrons occupy fixed energy levels.
How was the Bohr model was refined? (2)
- It was later discovered that not all electrons in a shell have the same energy.
- The model was refined to include sub-shells.
What are the stages of a TOF mass spectrometer? (4)
- The first stage is ionisation.
- The second stage is acceleration.
- The third stage is ion drift.
- The final stage is detection.
What is a mass spectrometer used for? (2)
- A mass spectrometer is used to determine all the isotopes present in a sample.
- It is also used to determine the elements or compounds within the sample.
Why is a mass spectrometer operated under a vacuum? (1)
To prevent air particles from becoming ionised and registering on the detector.
What are the two ways of ionisation in TOF mass spectrometry? (1)
- Electron impact.
- Electrospray ionisation.
What are the steps in electron impact ionisation? (3)
1. Firstly, a vaporised sample is injected at low pressure.
2. Then, an electron gun fires high-energy electrons at the sample, knocking out an outer electron.
3. This forms positive ions with different charges (e.g., Ti -> Ti⁺ + e⁻).
What are the steps in electrospray ionisation? (3)
- First, the sample is dissolved in a volatile, polar solvent.
- It is then injected through a fine needle at a high voltage.
- This causes the molecule (M) to gain a proton (H⁺), forming MH⁺.
Why must the electrospray solvent be volatile and polar? (2)
- The electrospray solvent must be volatile so that it can evaporate easily, leaving ions to move towards the negative plate.
- It must also be polar so that it can supply molecules with hydrogen ions.
What types of molecules are usually ionised by electron impact and electrospray ionisation, and why? (3)
- Electron impact is generally used for elements and compounds with a low relative molecular mass, as larger organic molecules tend to fragment under the high-energy conditions.
- Electrospray is suitable for larger organic molecules, such as proteins, because it is a gentler method that minimises fragmentation.
- The choice of ionisation technique depends on the size and stability of the molecule under ionising conditions.
What is the second step in TOF mass spectrometry? (3)
- Acceleration is the second stage of TOF mass spectrometry.
- This is when all positive ions are accelerated by an electric field generated by a negatively charged plate.
- Each ion is given the same kinetic energy.
What determines the velocity of each positive ion in TOF mass spectrometry? (3)
- The mass of the ions determines their velocity.
- Lighter ions achieve a higher velocity.
- While heavier ions move more slowly, as shown by the equation KE = 1/2mv².
How are ions separated in the third stage of TOF mass spectrometry? (3)
- In the third stage, ions are separated based on their m/z values, even though they have the same kinetic energy.
- Ions with higher m/z values take longer to travel through the drift area.
- As a result, different ions have different flight times, which are recorded once they reach the detector.
What happens during the fourth and fifth stages of TOF mass spectrometry? (4)
- In the fourth stage, ions reach the detector and each ion gains an electron from it.
- This electron gain generates a small current.
- The current is then fed into a computer for analysis.
- The magnitude of the current is proportional to the abundance of the ion species detected.
What is relative atomic mass? (2)
- Relative atomic mass is the weighted average mass of the isotopes of an element.
- It is measured relative to 1/12th of the mass of a carbon-12 atom.
What is relative isotopic mass? (1)
Relative isotopic mass is the mass of a particular isotope of an element compared with carbon-12.
What is relative molecular mass (Mᵣ)? (1)
Relative molecular mass is the average mass of a molecule compared with the mass of a carbon-12 atom, which is taken as 12.
Example: Calculate the relative molecular mass of ethanol (C₂H₆O). (2 marks)
- Mᵣ = (2 × 12.0) + (6 × 1.0) + (1 × 16.0).
- Mᵣ = 46.0.
What is the formula for RAM? (1)
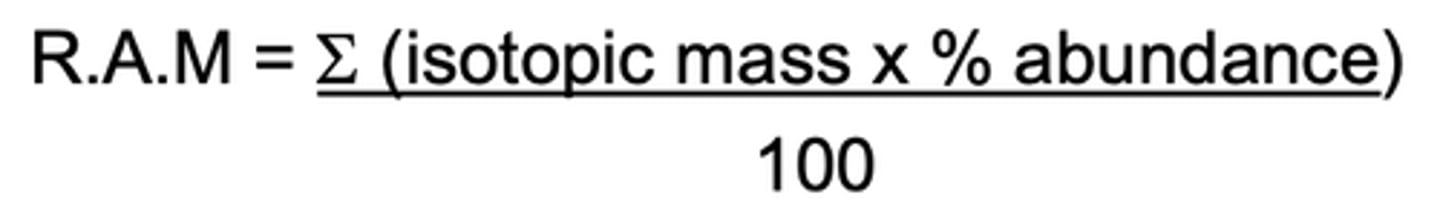
How might they trick you with relative atomic mass questions? (1)
They may provide relative abundances that do not total 100 but are based on the sample's total relative abundance.
What is the isotopic composition for chlorine and bromine? (2)
- The isotopic composition for Chlorine is 35Cl (75%), 37Cl (25%).
- The isotopic composition for Bromine is 79Br (50%), 81Br (50%).
What is the ratio of diatomic ions for chlorine and bromine? (2)
- The ratio of diatomic ions for Chlorine is 9:6:1.
- The ratio of diatomic ions for Bromine is 1:2:1.
What is a molecular ion? (1)
A molecular ion is the highest m/z peak on the mass spectrum.
What must you remember when identifying the molecular ion with electrospray ionisation? (1)
You must subtract 1 from the m/z value to account for the H⁺ ion.
What are the principal energy levels divided into? (2)
- The principal energy levels are divided into sub-energy levels.
- These are labelled as s,p,d and f.
How many electrons does each sub-energy level hold? (4)
- Sub-energy level s holds 2 electrons.
- Sub-energy level p holds 6 electrons.
- Sub-energy level d holds 10 electrons.
- Sub-energy level f holds 14 electrons.
What are sub-energy levels further divided into? (1)
Sub-energy levels are further divided into orbitals, which each hold up to 2 electrons with opposite spins.
What do orbitals represent? (1)
Orbitals represent the mathematical probabilities of finding an electron at any point within a certain distance from the nucleus.
What is the order in which atoms fill up with electrons? (1)
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶
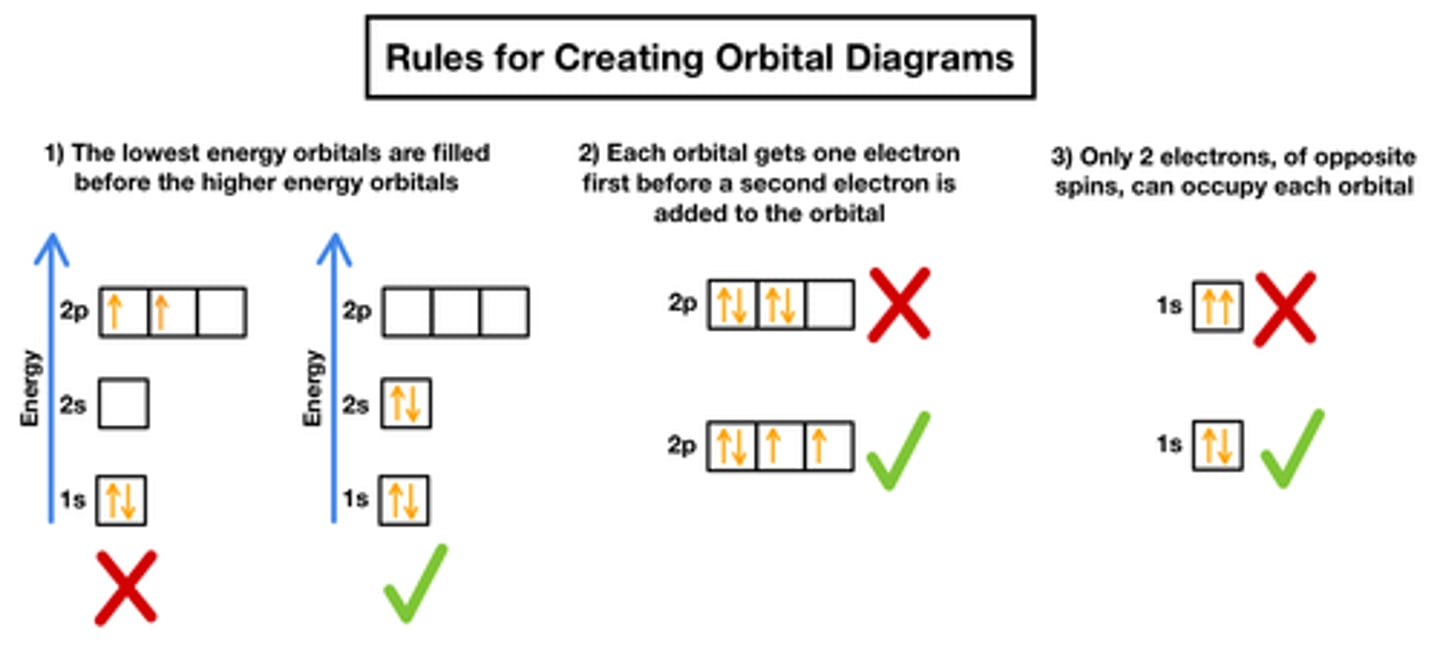
What is the rule for spin diagrams? (3)
Electrons fill orbitals singly before pairing up.
What is the relationship between the position of the outer electron and the placement of the element on the periodic table? (2)
- The sub-energy level of the outermost electron determines the block in which an element is placed on the periodic table.
- For example, sodium's outer electron is in the s sub-level, so sodium is located in the s-block.
Draw a diagram to show where all the blocks are on the periodic table. (3)
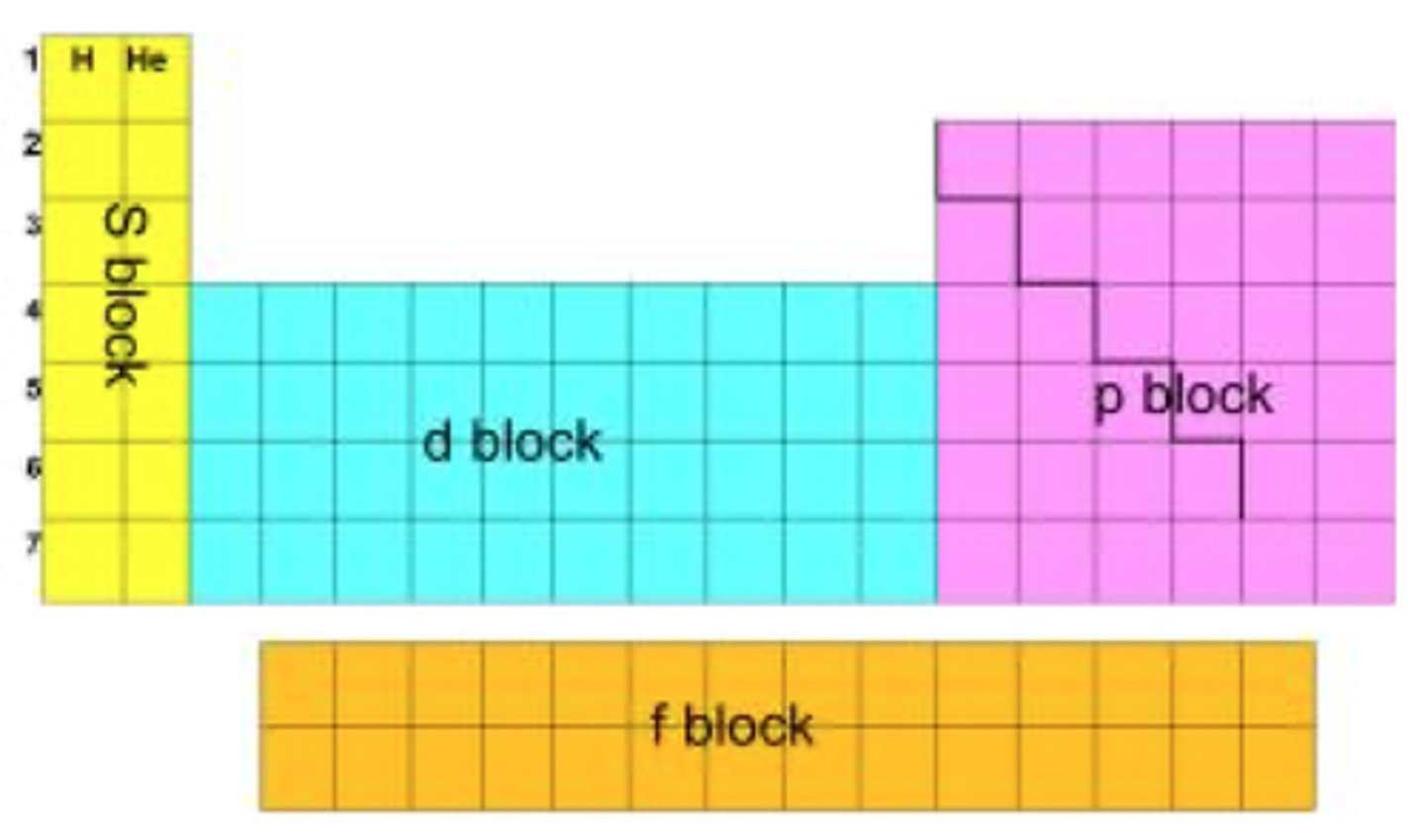
How do d-block elements lose electrons? (1)
D-block elements lose electrons from the 4s orbital before the 3d orbital.
What are the exceptions to conventional electronic configuration? (2)
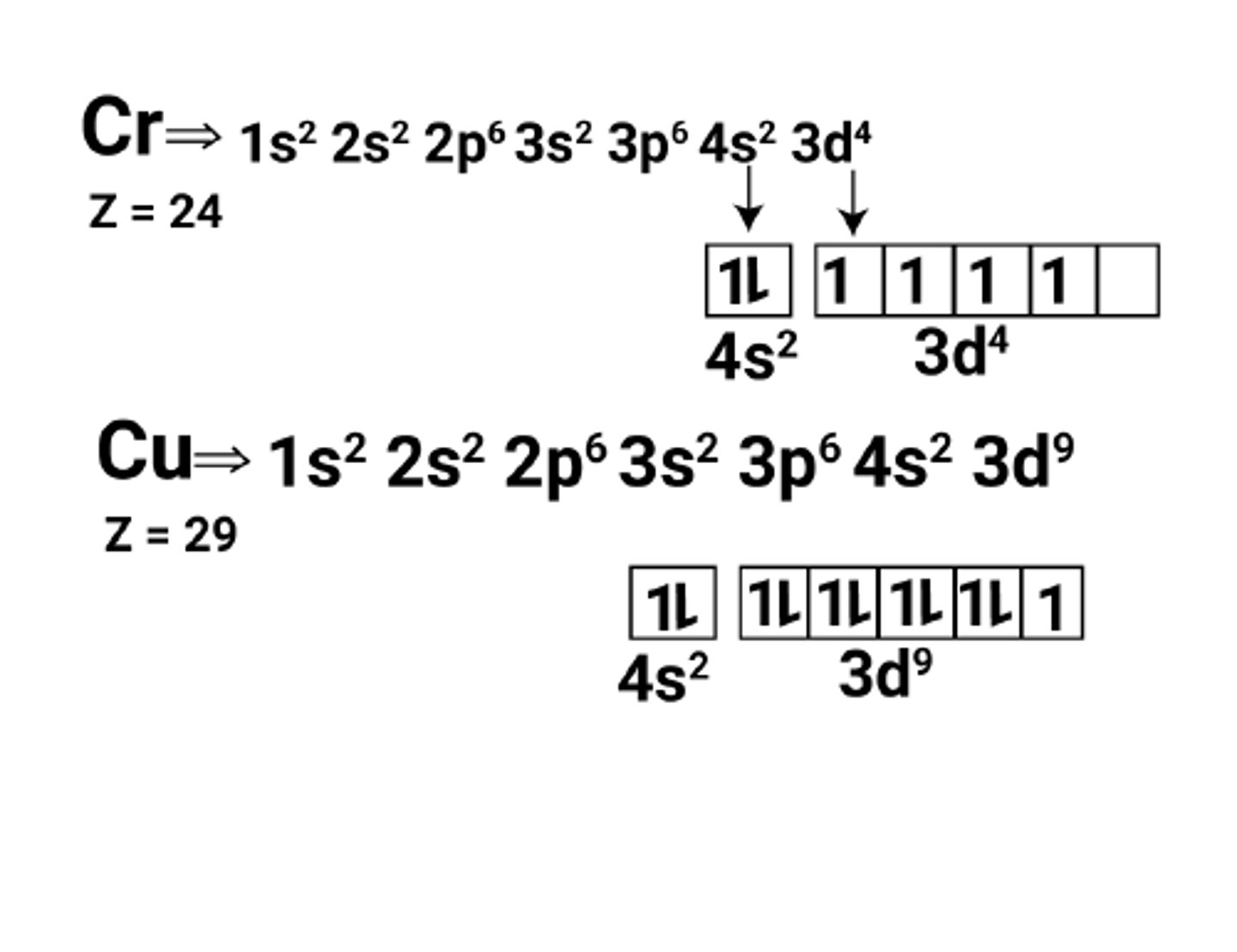
- Copper (Cu) and chromium (Cr) are exceptions to the conventional electronic configuration.
- They promote an electron from the 4s orbital to the 3d orbital, resulting in a half-filled or fully filled 3d subshell.
What is the electron configuration for copper and chromium? (1)
What is first ionisation energy? (1)
The first ionisation energy is the minimum amount of energy required to remove an electron from a gaseous atom to form a gaseous ion.
What is the first ionisation equation for magnesium? (2)
The equation is Mg(g) → Mg⁺(g) + e⁻.
What is second ionisation energy? (1)
The enthalpy change when one mole of electrons is removed from one mole of gaseous 1+ ions, forming one mole of gaseous 2+ ions.
What is the equation for the second ionisation energy of titanium? (2)
Ti⁺(g) → Ti²⁺(g) + e⁻.
What is the equation for the third ionisation energy of titanium? (2)
Ti²⁺(g) → Ti³⁺(g) + e⁻
What are the factors affecting ionisation energy? (3)
- Nuclear charge.
- Atomic radius.
- Shielding
Why are successive ionisation energies always larger than the first? (3)
- After each electron is removed, the positive charge of the ion increases.
- This results in a stronger attraction between the nucleus and the remaining electrons.
- Therefore, more energy is required to remove each subsequent electron.
What does a large jump in successive ionisation energies indicate? (1)
A large jump in successive ionisation energies indicates the element's group in the periodic table.
Why does helium have the largest first ionisation energy? (3)
- Helium's first electron is in the shell closest to the nucleus.
- There is no shielding from inner electron shells.
- Helium has one more proton than hydrogen, resulting in a stronger electrostatic attraction between the nucleus and its electrons.
Why do first ionisation energies decrease down a group? (3)
- Outer electrons occupy shells that are progressively further from the nucleus.
- There is increased shielding from inner electron shells.
- As a result, the electrostatic attraction between the nucleus and the outer electron weakens.
Why is there a general increase in first ionisation energy across a period? (3)
- The number of electron shells remains constant across the period, so shielding is similar.
- The nuclear charge increases with each successive element.
- Therefore, there is a stronger attraction of the nucleus to the outer electron.
Why is there a small drop in first ionisation energy from magnesium to aluminium? (4)
- Aluminium begins filling the 3p orbital, whereas magnesium's outer electrons are in the 3s orbital.
- Electrons in the 3p orbital are slightly higher in energy and therefore easier to remove.
- The 3p electrons experience additional shielding from the 3s electrons.
- As a result, less energy is required to remove the outer electron in aluminium compared to magnesium.
Why is there a small drop in first ionisation energy from phosphorus to sulphur? (4)
- In sulphur, the fourth electron in the 3p subshell pairs up with an electron already present.
- There is slight repulsion between the two negatively charged paired electrons.
- This electron-electron repulsion makes the paired electron easier to remove.
- Consequently, the attraction between the outer electron and the nucleus is weaker in sulphur.