Biology Module A
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
What are the four classes of biological macromolecules?
Carbohydrates, proteins, lipids, nucleic acids.
What is a monomer?
A small molecule that can join with others to form a polymer.
What is a polymer?
A large molecule made from repeating monomers.
What is a condensation reaction?
A reaction where two molecules join and water is released.
What is another name for a condensation reaction?
Dehydration reaction.
What is hydrolysis?
A reaction where water breaks a bond.
Why do organisms use condensation reactions?
To build macromolecules.
Why do organisms use hydrolysis reactions?
To break molecules for digestion or energy.
What is a monosaccharide?
A single sugar molecule, such as glucose.
What are two features that distinguish carbohydrates?
Number of carbons and the location of the carbonyl group.
What is a disaccharide?
Two monosaccharides joined together.
What bond joins sugars together?
Glycosidic bond.
What reaction forms glycosidic bonds?
Condensation reaction.
What is a polysaccharide?
A polymer made of many sugars.
What is an example of an energy storage polysaccharide in plants?
Starch.
What is an example of a structural polysaccharide in plants?
Cellulose.
Why can humans digest starch but not cellulose?
Humans cannot break β-1,4 glycosidic bonds.
Which is broken down faster: amylose or amylopectin?
Amylopectin, because it is branched.
What are proteins made of?
Amino acids.
How many standard amino acids exist?
20.
What are the four components of an amino acid?
Amino group, carboxyl group, hydrogen, and R group.
What determines amino acid properties?
The R group (side chain).
Which amino acid is not chiral?
Glycine.
What bond links amino acids?
Peptide bond.
How is a peptide bond formed?
Condensation reaction.
What reaction breaks peptide bonds?
Hydrolysis.
What is pH?
A measure of H⁺ concentration.
What is pKa?
The pH where half the molecules are protonated; it measures the strength of an acid.
What happens when pH < pKa?
The molecule is protonated.
What happens when pH > pKa?
The molecule is deprotonated.
What is primary protein structure?
The amino acid sequence.
What is secondary protein structure?
Local folding patterns in proteins, such as α-helix and β-sheet.
What stabilizes secondary protein structure?
Hydrogen bonds.
What is tertiary protein structure?
The overall 3D folding of a protein.
What is quaternary protein structure?
Multiple polypeptide chains forming one protein.
What is the function of hemoglobin?
Transports oxygen in blood.
What is the function of myoglobin?
Stores oxygen in muscle.
What mutation causes sickle cell disease?
Glutamate to Valine.
What is denaturation?
The loss of a protein's 3D structure.
Do peptide bonds break during denaturation?
No.
What is insulin?
A hormone that controls blood glucose.
What type of bonds stabilize insulin?
Disulfide bonds.
What is the structure of collagen?
A triple helix of three alpha-chains twisted into a right-handed supercoil.
Why is glycine common in collagen?
It is small enough to fit inside the tight triple helix.
What vitamin is required for collagen stability?
Vitamin C.
What is a competitive inhibitor?
An inhibitor that binds to the active site.
What is a non-competitive inhibitor?
An inhibitor that binds away from the active site and changes the enzyme's shape.
What is feedback inhibition?
When the product of a pathway inhibits an earlier enzyme.
What are the three main classes of lipids?
Fats, phospholipids, and steroids.
What is a triglyceride?
Glycerol plus three fatty acids.
What bond links fatty acids to glycerol?
Ester bond.
What is the difference between saturated and unsaturated fats?
Saturated fats have no double bonds; unsaturated fats contain double bonds.
What is a phospholipid?
A lipid with a hydrophilic head and hydrophobic tails.
Why do phospholipids form membranes?
They are amphipathic.
What structural feature do hemoglobin and myoglobin both contain to bind oxygen?
A heme group containing iron (Fe²⁺).
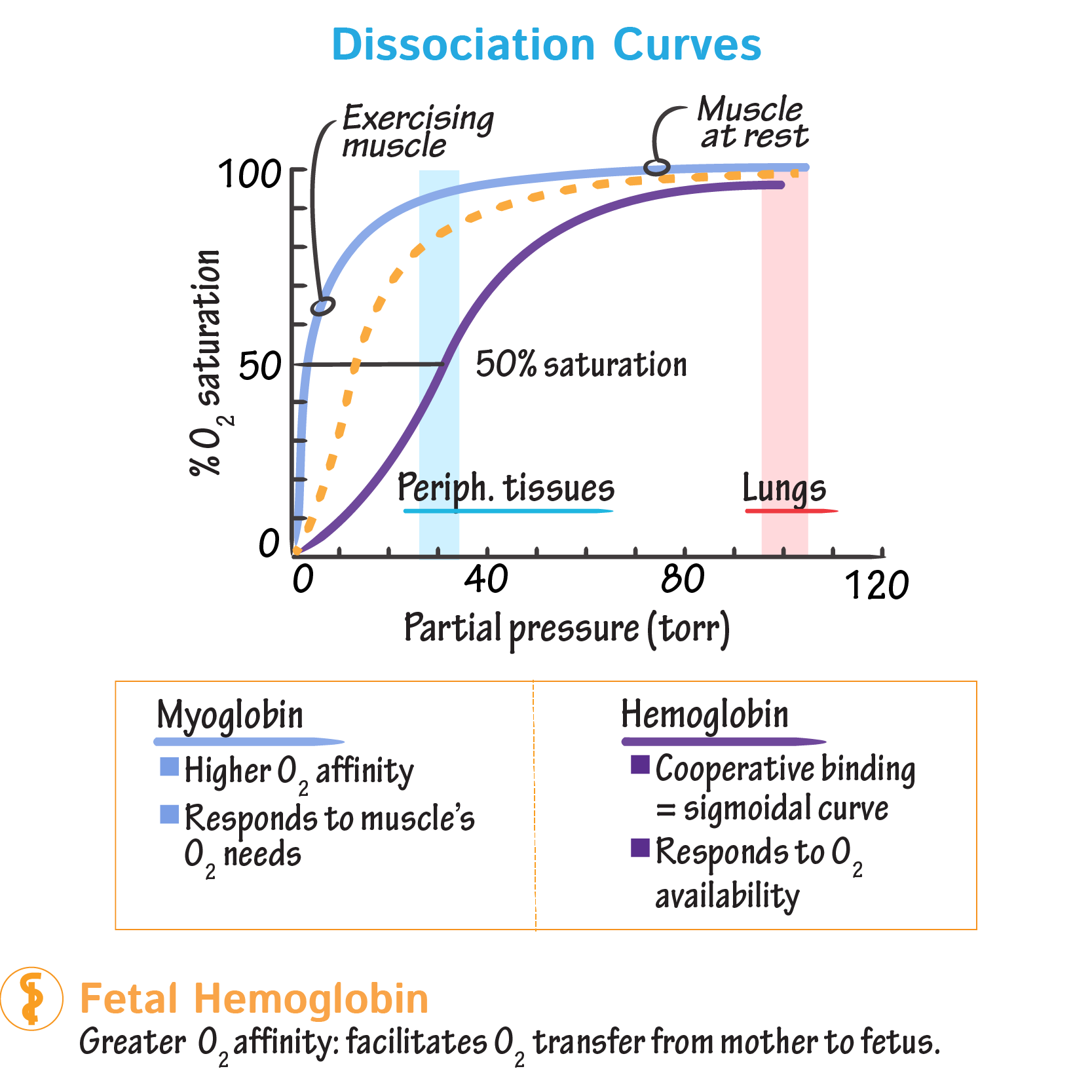
What is the difference between the oxygen binding curves of myoglobin and hemoglobin?
Myoglobin is hyperbolic (one subunit); hemoglobin is sigmoidal (four subunits, showing cooperativity).
What is Km in enzyme kinetics?
The substrate concentration at which the reaction rate is half of Vmax; indicates affinity.
What is kcat?
The catalytic speed; the number of substrate molecules converted to product per enzyme per second.
Tetrameric vs monomeric quaternary structures and give an example
tetrameric = 4 polypeptides ex. hemoglobin
monomeric = 1 chain ex. myoglobin
what are the shapes that a teritiary structure can be
globular = spherical or fibrous = long and thin
what are secondary structures stabilized by?
hydrogen bonds
alpha helix vs beta sheets
alpha helix: stabilized by hydrogen bonds between nearby residues
beta sheet: stabilized by hydrogen bonds between adjacent segments that may not be nearby
what are the ends of the primary structure of an amino acid called?
amino end, carboboxy terminal end
Explain oxygen saturation curve (hemoglobin & myoglobin)
the less O2 there is the more will unbind from hemoglobin and vice versa
when pO2 rises hemoglobin becomes more loaded (saturated) with O2
what does it mean when two proteins are homologous?
they have similar primary, secondary, and tertiary structures (meaning likely have similar functions) BUT differ in quaternary structure
why is carbon monoxide a risk to humans?
because it’s similar in structure to o2 and myoglobin + hemoglobin will bind strongly to it. it can outcompete o2 so that we dont have enough of it
what happens during denaturation?
the peptide bonds don’t break but the hydrogen bonds do. 3d structure is modified but polypeptide structure isn’t
what is a native structure (protein)
protein that is completely folded and fully active
what is tropocollagen vs procollagen
procollagen is inactive and has the c-terminal peptides still attached
tropocollage is active because the procollagen peptidase removed the c-terminal peptides (unstructured polypeptide that prevents fibril formation)
what is the smallest amino acid?
glycine