H1 Toth- Endomembrane system I + II: key organelles, vesicles, signals and sorting
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
how are proteins secreted in eukaryotes?
the proteins are translocated co-translationally by the ribosomes of the rough ER, either integrating into the membrane or being released into the lumen
during this process, N-linked glycans are added- short oligosaccharides (sugar chains) are coupled to the protein via a nitrogen in an asparagine residue
this is used as a signal throughout the secretion process to determine whether the protein is folded and modified correctly (controlled by chaperone proteins), as the glycan chain is truncated and edited at different points, for quality control
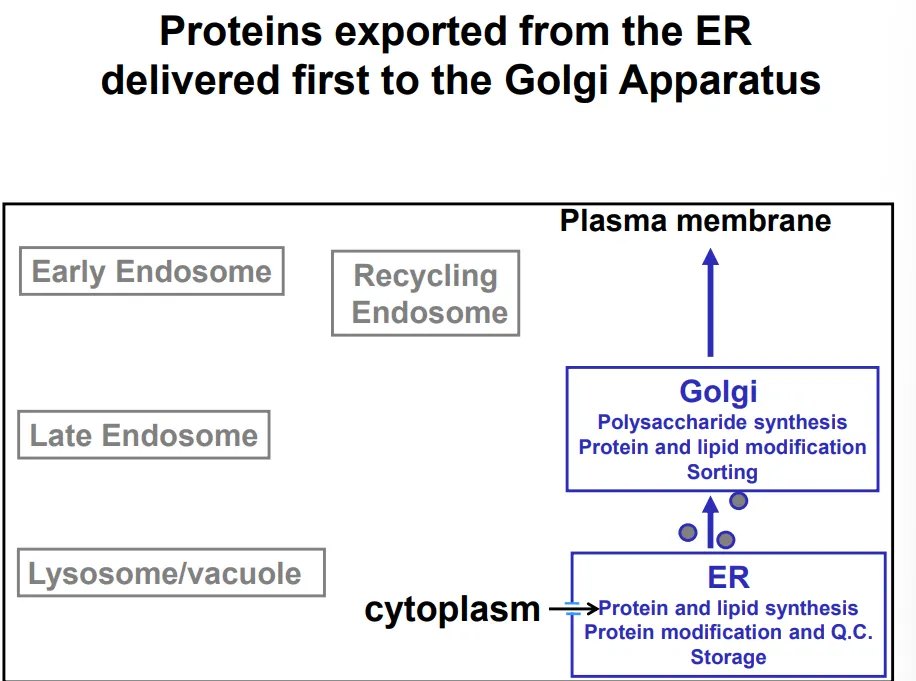
the proteins are transported from the ER to the golgi apparatus by the membrane budding into COP-II vesicles (so that they don’t need to cross membranes to be secreted)- this is also used to transport lipids produced by the smooth ER
the golgi apparatus (cis → medial → trans layers of cisternae) is a polysaccharide factory, which modifies the existing N-glycan chains for signalling and adds further polysaccharides of functional importance for glycoproteins and glycolipids
the vesicles bud off again and release the contents of the lumen (topologically equivalent to extracellular space) out of the cell, either immediately or following an external signal, in the case of regulated secretory vesicles
what is the endocytic pathway in eukaryotes?
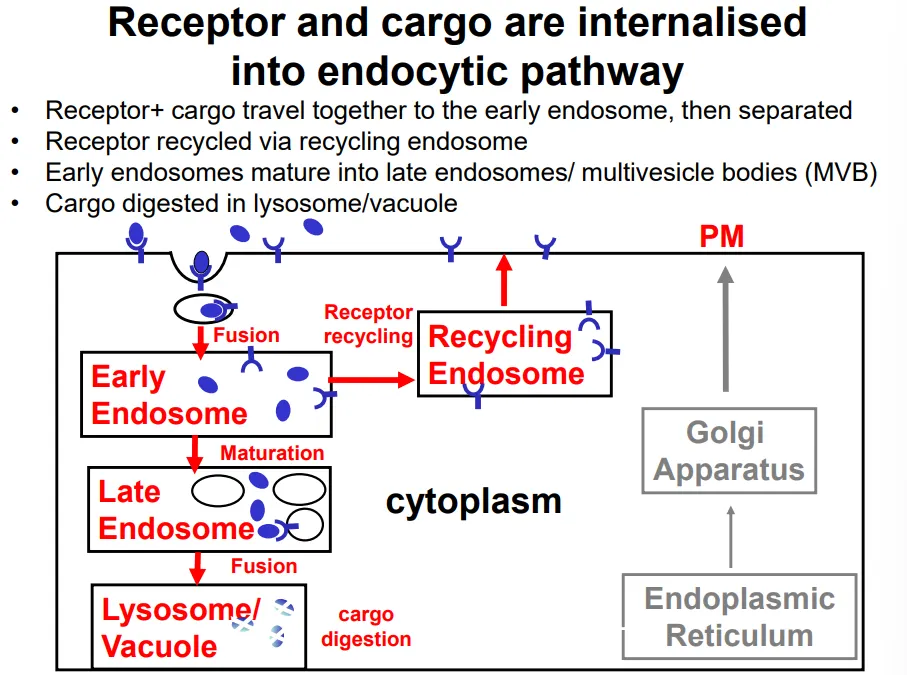
endocytosis and phagocytosis are initiated by specific receptor proteins, which form a receptor-cargo complex
clathrin-coated vesicles bud off the plasma membrane into the cell, producing an endosome
the receptor and cargo dissociate in the early endosome, so that the receptors can be re-inserted into the plasma membrane via a recycling endosome
the cargo remains in the endosome, which matures into a late endosome upon receiving additional contents (eg. enzymes) from the golgi apparatus
exisiting lysosomes fuse to this, releasing further digestive enzymes that degrade the cargo (nutrients, pathogens and signalling molecules)
during maturation, the endosome becomes increasingly acidic until it becomes a lysosome, where the pH is low enough for the digestive enzymes to function (acidic optimum so that they don’t damage the cell earlier)
how are transport vesicles formed and what proteins do each kind of vesicle use in the process?
curvature of membranes is energetically unfavourable, so to form vesicles, they must be forced into deformation by a protein coat
protein coat subunits self-assemble on the cytoplasmic side of the membrane
these are also responsible for collecting and packaging the cargo into the vesicle
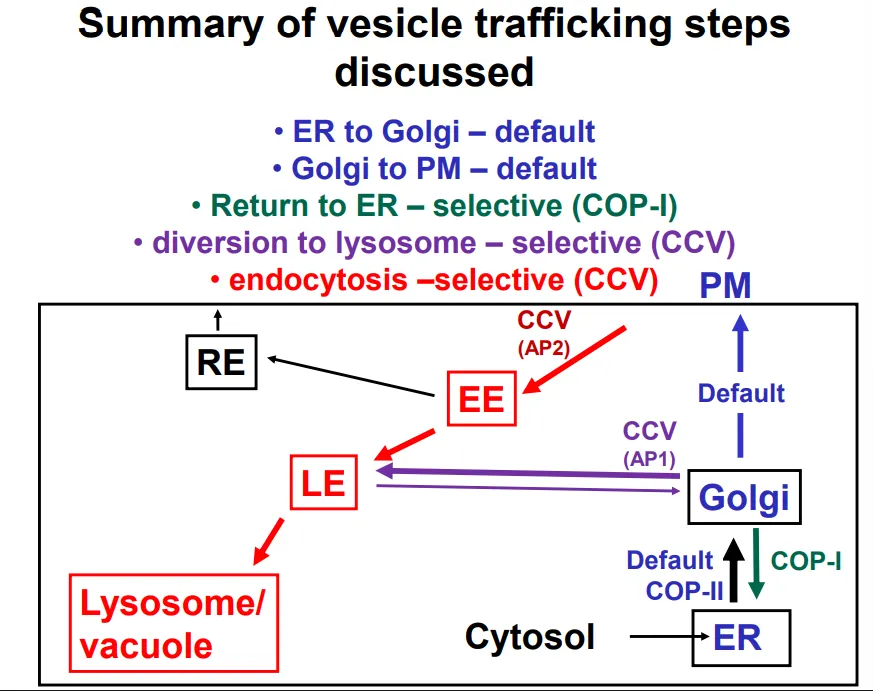
ER → golgi vesicles use COP-II coats
golgi → ER (retrieval) vesicles use COP-I coats
PM → early endosome vesicles use clathrin and adaptin 2 to form clathrin coated vesicles (AP2)
golgi → late endosome vesicles use clathrin and adaptin 1 to form clathrin coated vesicles (AP1)
how are proteins signalled to be retained or exported in the ER?
export from the ER doesn’t require a signal, it occurs by default ‘bulk flow’ (but some signals may accelerate export)
bacterial proteins that are inserted into eukaryotes (adding a signal for production in the ER, but not a signal for retention) are excreted
resident proteins aren’t prevented from leaving the ER, but they are continually retrieved from the golgi apparatus due to an ER-retrieval signal
these are necessary (deletion of the signal results in secretion instead of residency) and sufficient (the only sequence that is required to cause secretion)
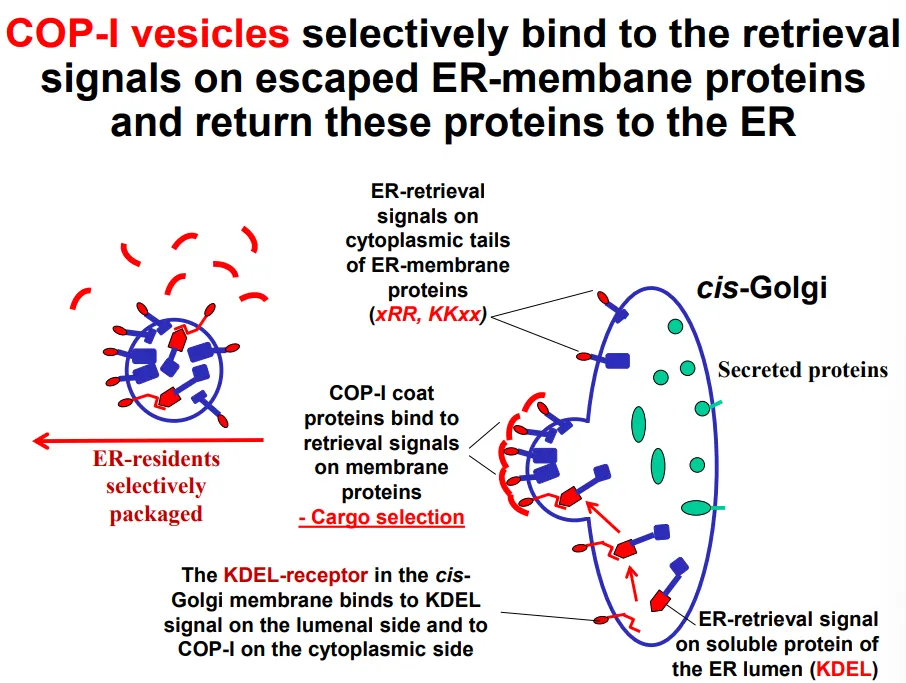
COP-I vesicles selectively bind to retrieval signals (primary structure) for escaped ER resident proteins that are found in the cis-golgi:
diarginine (N terminus) and dilysine (C terminus) motifs on the cytoplasmic side of transmembrane ER resident proteins
KDEL receptor proteins in the cis-golgi membrane bind to the KDEL sequences (C-terminus) of luminal ER resident proteins
this is cargo selection, which then triggers vesicle formation
the KDEL receptor proteins dissociate from the cargo upon return to the ER due to pH differences between the ER and golgi lumen (the ER is less acidic, which discourages the association), so the receptors can be recycled to the golgi in COP-II vesicles
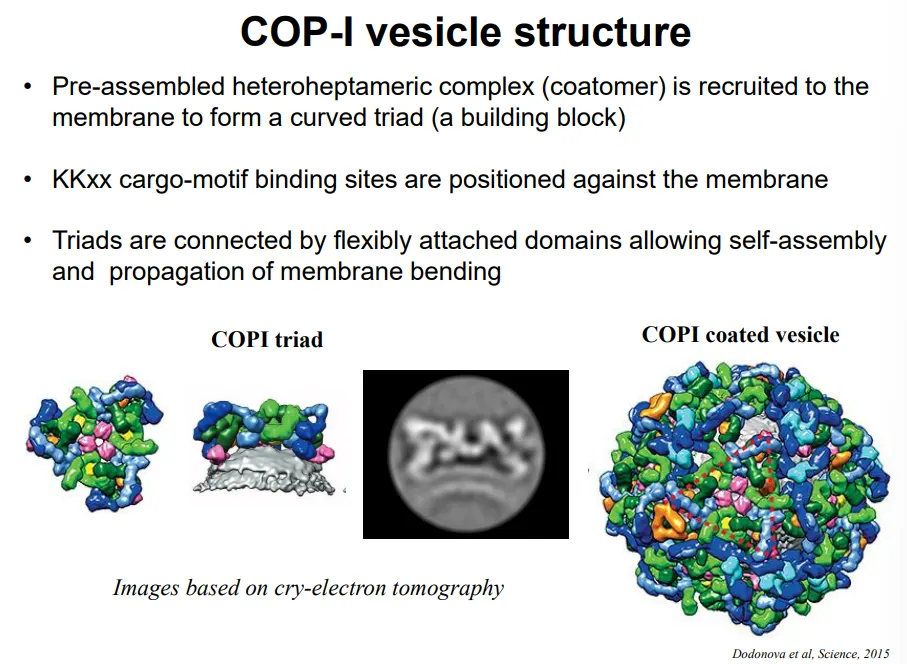
what is the structure of COP-I vesicles?
pre-assembled COP-I triads are present in the cytoplasm
these complexes have a natural curvature
multiple triads assemble to cause membrane bending, with the cargo-motif binding sites positioned against the membrane for cargo selection
these are flexibly bound together so that the size of the vesicles can change
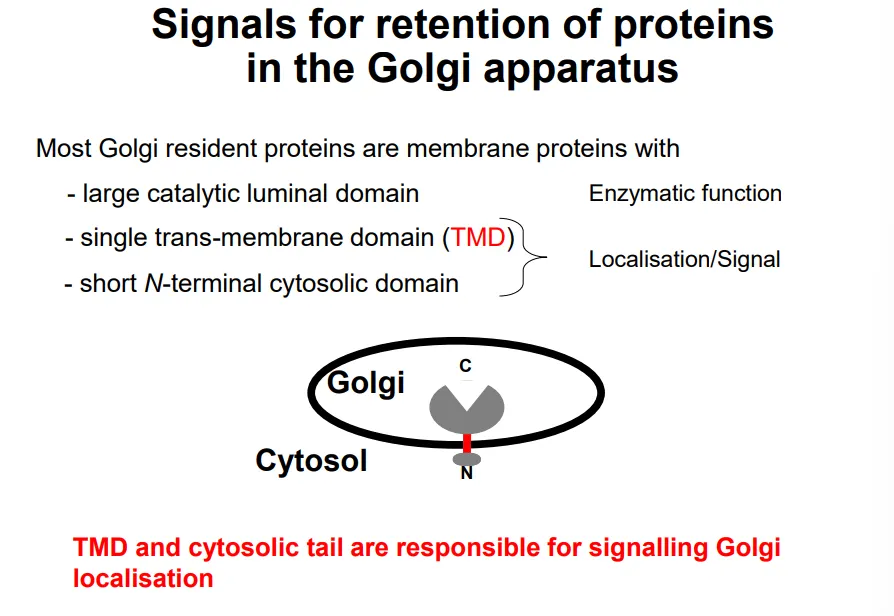
how are proteins signalled to be retained or exported in the golgi apparatus?
the large luminal domain of golgi resident proteins is functional enzymatically eg. to make polysaccharides
this is attached to a trans-membrane domain and a short N-terminal cytoplasmic domain, which act as a secondary structure signal
the different organelles in the secretion pathway have different lipid compositions, so the membrane thicknesses increase from ER → golgi → PM
this means that the TMDs of membrane proteins also increase in length, so they may be rejected if they are foreign to each organelle (eg. the hydrophobic region isn’t long enough, so charged residues clash with the fatty acid tails)
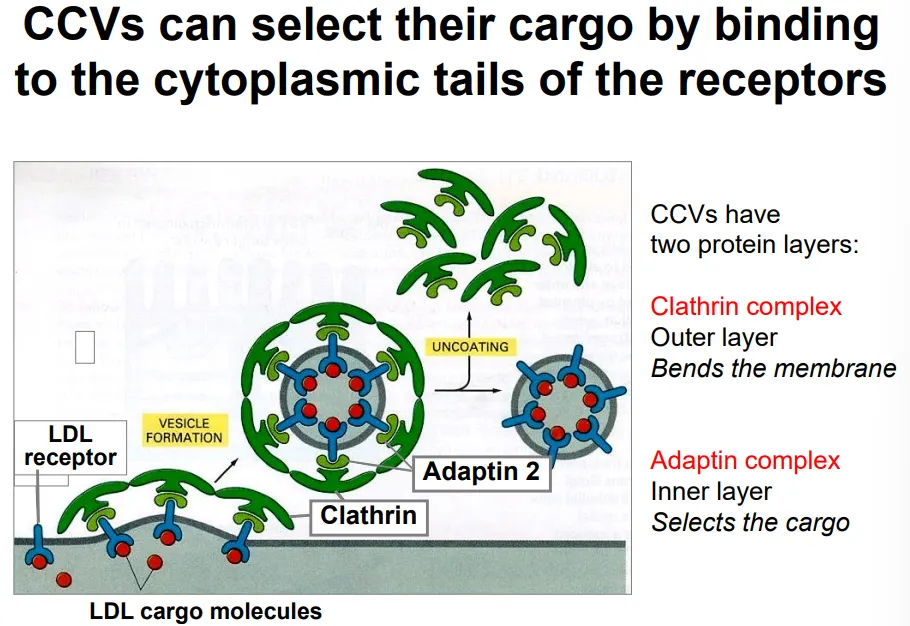
how does endocytosis occur selectively? use cholesterol as an example
cholesterol is transported through the blood as part of low density lipoprotein particles
these contain a large protein signal molecule, which binds to LDL receptors on the plasma membrane of target cells
these receptors bind to adaptin 2 complexes (cargo selection) in the cytoplasm, which in turn attach to clathrin triskelion complexes that cause membrane bending by spontaneous self-assembly, to produce a clathrin coated vesicle (CCV)
the clathrin and adaptin dissociate (using energy) from the vesicle to allow fusion to the early endosome, where the LDL receptors dissociate due to the lower pH so that they can be recycled back via recycling endosomes
how are proteins signalled to be transported from the golgi apparatus to the late endosome?
digestive hydrolases made in the ER have a signal patch in their tertiary structure, which is recognised by enzymes in the cis-golgi
these enzymes phosphorylate a mannose residue on the N-glycan chain of the protein
this modification is recognised by a mannose-6-P receptor in the trans-golgi, which binds to the glycan chain as well as adaptin 1 complexes from the cytoplasm
this causes the formation of clathrin coated vesicles, which transport the cargo to the late endosome
here the Man-6P receptor dissociates due to a decrease in pH, so that it can be recycled back to the trans-golgi
these hydrolase proteins are activated by proteases found in the lysosome, by proteolytic cleavage, so that they don’t cause damage to the cell earlier in the process
how are transport vesicles targeted to the correct destination?
the vesicle temporarily docks to a membrane
here its specific v-SNARE (vesicular) proteins test their interactions with the t-SNARE (target) proteins on the membrane
if the t and v-SNARE proteins are complementary to each other, they form a very tight coiled-coil structure, which brings the vesicle close to the target membrane so that fusion can happen
if not, they won’t coil and the temporary docking will release the vesicle to try elsewhere
after fusion, NSF complexes remove the v-SNARE proteins from the target membranes, using ATP, to maintain compartment identity