Chem exam 3
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
amines
R-NH2
Have a lower priority than -OH
1º, 2º, 3º amine
depends on how many carbons are attached to the nitrogen
aldehydes
alkanal
R-CHO
can reduce and oxidize
ketones
alkanone
R-C=O-R
can reduce
carboxylic acids
alkanoic acids
R-COOH
undergo: conjugate acid-base reactions
conjugate acid-base reactions
Drop the -ic and add -oate to the chain and name the ion
fats and oils
carboxylic acids that are 10-20 carbons long
H2O soluble salts
Na+, K+, NH4+
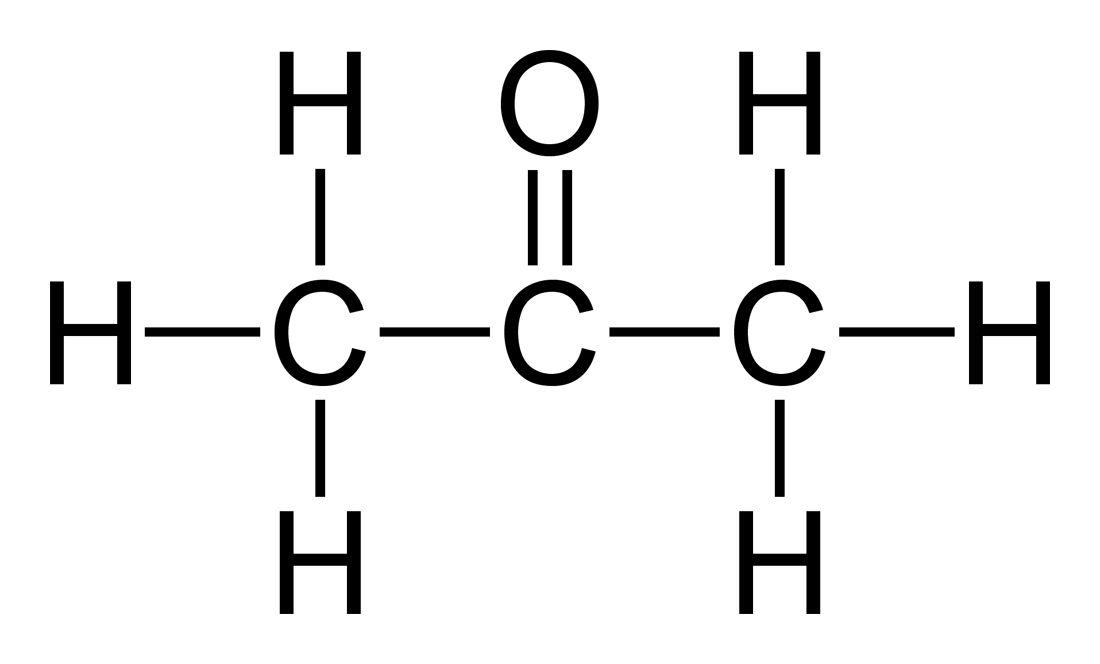
acetone
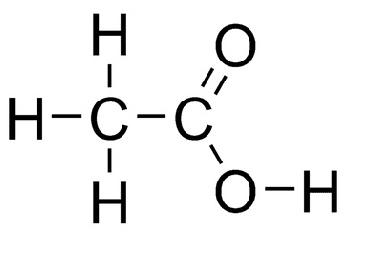
acetic acid
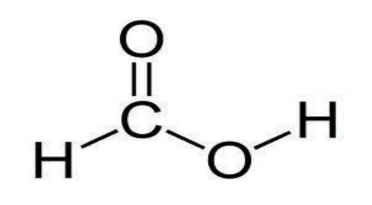
formic acid
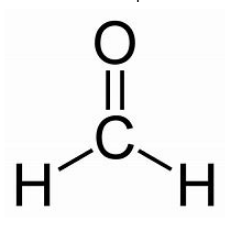
formaldehyde
Esters
R-C(=O)-O-R
drop the -ic and add -ate
Amide
R-C(=O)-N
Carbohydrates
CxH2Oy
all natural sugars have penultimate carbon -OH ground on the right side
DSugar
penultimate -OH on the right side
LSugar
penultimate -OH on the left side
pyranose
6 membered ring
O at 1 o’clock
alpha sugar
-OH oriented down at carbon 1
beta sugar
-OH oriented up at carbon 1
furanose
5 membered ring