Environmental Pollution
1/151
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
152 Terms
Run of River hydropower
water is diverted from river, and runs through turbine
pump and store
usually requires building a dam, provides electricity at peak energy demand, and uses excess at night to pump water up
large scale storage
i.e. 3 Gorges dam, but can flood greenery, which causes decomposition of organic material and CO2 can be released
Installed Capacity (Wind Power)
amount of energy we could produce if turbines are working at maximum capacity all of the time
Base load
energy provided doesn’t change over time, for example nuclear power
Variable load
can increase output from energy sources when demand is high
peak demand
when energy demand is highest, i.e. in the morning and around dinner time
acrotelm
upper region of peat, the zone of living plants, can filter water very well and is partially aerated
catotelm
the lower region of peat, no living plants, accumulates plant material (humic material) and stores most of the carbon
Why is peat important in wind energy
the places with high wind are also places where there is a lot of peat (high elevation) if turbines are built, it can increase the amount of CO2 released from peat (b/c if you dry out the peat, increases oxygen into the lower layer and causes aerobic decompositon)
Peatland rewetting
peatland rewetting can cause the humic material to get broken down and escape into drinking water, which then can cause DBPs further down the line and accumulate other contaminants
Deep Geothermal
heat from depths >500m, partly radioactive heat and partly primordial heat
Solution for peat wind farm issue
Floating roads, a road with 1 or 2 layers of geogrids that distribute load evenly and line sides with peat to avoid water run-off
Binary Cycle Geothermal
Geothermal is <150-180 C, so we use a highly efficient heat exhanger to heat water and make steam to turn a turbine. A closed loop cycle with direct return of fluids to depth, so no liquid or gas emissions
Hydrothermal systems
Wet steam vs. dry steam (wet steam is water under high pressure but at surface turns into steam), used to turn a turbine. Can also release gases like CO2, CH4, H2S, NH3
Physical Carbon Capture
Absorption (selexol) and adsorption (zeolites, etc.)
Chemical Carbon Capture
Absorption (using amines) and bonding
Lithium ion batteries
sealed, 80% capacity, lasts 20+ years, 3000-5000 cycles, more expensive upfront, cheaper per kWh
Where is lithium found
lithium can be found in brines and minerals, Chile has most of the world’s Lithium
Environmental Impact of processing Lithium
Land Use - 3124m² per tonne Li, Water use - 469 m³ per tonne Li, CO2 emissions - 5000kg/tonne Li
Tailings
Batteries use secondary metals and mining them and processing creates tailings, exponential growth of tailings,
we leave tailings in sludge ponds or piles and just leave them there - spray water on them which is a huge water sink
Acid mine drainage
cascade effect of acidity, dissolves heavy metals as well
Contamination Factor
CF = Cmetal/Cbackground
measured concentration / pre industrial/natural concentration
low contamination is CF < 1
moderate contamination is 1 < CF < 3
considerable contamination is 3 < CF < 6
high is CF > 6
Li Recovery
pyrometallurgy - combust Li in a furnace at 1000C to recover
hydrometallurgy - recovers desired metals by leaching in acidic or basic solution
biometallurgy - bacteria that can precipitate out metal
Landfill Fires
emerging environmental concern, can release HF (hydrofluoric acid), a major hazard and can cause serious toxic effects
solution to Li-ion battery recycling
Closed loop system - for different types of materials there could be different types of recycling
Vanadium
Important for many high tech and economically significant tech. Vanadium reflow plants are becoming popular
low energy density, more expensive, lasts longer, but requires high purity (rare)
supply risk is high
vanadium is potentially toxic to aquatic organisms
Nuclear power
provides a base load of energy
large amounts of energy released from comparatively small amount of fuel
Uranium
3 main isotopes - U-234, U-235, U-238
U-235 is fissile, used as fuel for nuclear power plants but only makes 0.7% of naturally occuring uranium
Uranium mining
main ores are uraninite and carnotite
open cast mining, using crushing and flotation to separate ores from impurities
Uranium refining
+IV oxidation state - insoluble
+VI oxidation state - soluble
to extract uranium, it is first oxidized, then extracted by adding sulfuric acid
organic solvent is used to separate uranium from other species
after recovery of uranium, it is precipitated
Environmental Impact of Mining and Refining Uranium
fine to coarse particles in slurry
Radium - highly soluble, leaches out
Radon - gaseous, can diffuse out
Enrichment
For energy, you just need 3% enriched uranium (for weapons you need 95%)
Nuclear spent fuel - what to do w it
generates a lot of heat, stored underwater for 6-12 months
cut up into small pieces and dissolved
reprocessed
separation of U and Pu from fission products, extraction w U and Pu going to organic layer, fission products in aqueous layer (TOGDA)
Intermediate level liquid waste (ILLW), High level liquid waste (HLLW), High level solid waste (HLSW)
ILLW - <4×10^4 GBq/m³
HLLW - ~10^7 GBq/m³
HLSW - cladding from fuel rods, etc.
LLW makes up a lot of nuclear waste, very low activity and can be disposed of by dumping into ocean
Deep disposal of nuclear waste
multi-barrier approach - physical containment, geological isolation, chemical conditioning, stable rocks (300-1000m underground)
absence of large fractures in solid repository matrix
impermeability to water
good heat conductivity
Half-Life equation
A = lambda*N(t)
activity = decay constant (1/s) * atom number
Waste Isolation Pilot Plant
military waste was places in steel canisters, the salt bed was not water free, corroded the waste canisters and there was a major explosion
Future of Nuclear
Thorium-232 is not fissile, but it is fertile (can absorb neutrons and convert into fissile)
3x more abundant than uranium
doesn’t require water as a primary coolant
5x less mining waste than uranium
but also complicated and expensive
Load Factor
(actual generation/installed capacity) *100
Pesticides
bactericide, fungicide, herbicide, insecticides
traditional pesticide problems
usually very toxic to humans and mammals at dosages required to make them effective
non-biodegradable
Desirable characteristics of a pesticide
small amount needed
low toxicity to non-target species
lifetime just long enough to kill target pests
degrades to benign products
does not accumulate in living organisms
does not runoff with water from application site
pests are slow to develop resistance
DDT
agricultural insecticide that opens sodium ion channels in neurons, causing death in insect
silent spring - rachel carson, documented impact of ddt on wildlife
bioconcentration, bioaccumulation & biomagnification
bioconcentration - higher concentration of a chemical in organism than the environment in which it’s exposed to
bioaccumulation - uptake of a chemical by an organism following consumption of a food source
biomagnification - sequence of processes by which higher concentrations of a chemical are reached in organisms higher up in the food chain
How do we detect DDD, DDE, DDT
liquid-liquid extraction using solvent like DCM
gas chromatography
Organophosphates
Insecticides - Parathion and malathion are examples of compounds
parathion - highly toxic to non-target organisms
cholinesterase inhibitor
absorbed through skin and mucous, rapidly metabolized
malathion - low toxicity
Pyrethroids
naturally occurring organic compounds with insecticidal properties
attacks nervous system of all insects - even to beneficial ones
toxic to base of food webs
we detect w conventional methods (GC-MS, FTIR, UV-Vis) or biosensors
Emerging Organic Pollutants
pharmaceutical and personal care products
perfluorinated compounds
endocrine distrupting potential
bioaccumulate/persistent properties
carcinogenic
Disruption of the endocrine system
some chemical mimic natural hormones, others block the effects of a hormone from certain receptors, others stimulate or inhibit the endocrine system and cause overproduction or underproduction of hormones
Ibuprofen
Ibuprofen has R and S form, human body converts R to S form
influent waste treatment waters contain mainly active form of drug + metabolites
high mobility in aquatic environment, concern about fate and effects
Triclosan
non-toxic
lipophilic, accumulates in fatty tissues
may interfere w thyroid hormone metabolism
toxic to algae
PFCs: PFOS & PFAS
PFOS - fire resistant, fabric protection, etc.
persistent, bioaccumulative, and toxic to mammals
causes cancer, endocrine disruption, neonatal mortality, reduced birth size
PFAS is found in drinking water, etc.
can be removed using activated carbon, ion exchange, or membrane filtration
Solubility
mass of solute that can dissolve in water
high solubility - >100 mg/L
moderate solubility - 10-100 mg/L
low solubility - <10 mg/L
Octanol-Water Partitioning
Kow = concentration in octanol / concentration in water
dimensionless ratio expressing distribution of organic pollutant between equal volumes of octanol and water
LogKow < 3 —> organic chemical remains in water
LogKow > 3 —> organic chemical partitions into soil/sediment
log P is equivalent to logKow, relates to the non-ionised form of the substance at pH where non-ionised form dominates
Bioconcentration Factor (BCF)
BCF = concentration in living organism / concentration in water
BCF < 100 - low tendency to bioconcentrate
100 < BCF < low 1000s - medium tendency
BCF > 5000 - high tendency to bioconcentrate
Why is accumulation in living organisms important?
toxicity
chemical burdens reduce fitness and resilience
food chain impacts
endocrine disruption
Vapour Pressure
pressure when liquid is at equilibrium with it’s vapour
<0.01 kPa = nonvolatile
> 0.01 kPa = volatile
Henry’s Law Constant
Kh = partial pressure in atmosphere / concentration in water
Kh = p(organic chemical)/ organic chemical
Kh > 100 Pam³ /mol —> volatile
Kh < 100 Pa m³/mol —> nonvolatile
Degradation Times
pathways: photolysis (photodegradation), hydrolysis, biodegradation
Environmental half life: t ½
Degradation Time 50%: DT50
Persistence - Resistance to Breakdown
Water DT50 > 2 months
Soil DT50 > 6 months
sediment DT50 > 6 months
Ionisation - Dissociation pKa & pH
organic chemicals may dissociate, can influence fate, behaviour, and toxicity
ionisation is driven by pH
pKa is pH at which half of chemical is ionised
pKa = -logKa
for neutral chemicals that ionise by releasing H+
pH < pKa = majority of chemical is not ionised (HA)
pH > pKa = majority of chemical is ionised (A-)
Biogeochemical cycles
Nature has slow occurring ones for trace elements and heavy metals
these cycles are controlled by environmental conditions
trace elements and heavy metals cannot be degraded like some organic pollutants
Inorganic contaminants
metals and their salts
inorganic fertilizers
sulfides
ammonia and oxides of nitrogen
acids and bases
Metals - pollutants
naturally occuring and anthropogenic sources
speciation of metals dictates behaviour
oxidation state, complexation, pH, redox conditions —> determine mobility, toxicity, and bioavailability
Discoloured tap water
Mn and Fe have soluble and insoluble forms in aquatic systems
Fe(II) is soluble, oxidizes rapidly to form insoluble Fe(III)
Mn(II) is soluble, oxidised much more slowly to insoluble Mn(III)/(IV) oxides
soluble Mn is difficult to remove during water treatment
generally, reduced species are more likely to be soluble
Eh-pH Diagrams
dotted lines represent the redox limits that liquid H2O can exist at
each field shows pH and redox values a certain species is stable at
species oxidation state controls mobility
Why is Mn in drinking water a problem?
>50 microgram/L causes aesthetic concerns
at higher concentrations, potential for adverse effects upon human health
manganism, parkinson like disorder
decline in intellect in children
WHO limit is 400 microgram/L
What controls dissolved Mn?
Geology
rock weathering is ongoing source
redox related processes mean Mn concentrations may not directly control those in surface water/sediment/soil
Soils
Mn concentrations reflect soil type
redox potential and pH control soluble Mn inputs into surface waters
in addition, soil OM content can help stabilize soluble Mn and mobilize insoluble oxides
Vegetation
may impact Mn runoff into surface waters through soil feedback
coniferous forests tend to result in greater Mn mobilization, acidic soil environments
Mn Pathway into water bodies
Redox cycling between Mn(II) (soluble) and Mn(IV)O2 and Mn(III) (both insoluble) —> due to acidic v. alkaline conditions and microbial activity
goes into water from surface runoff and groundwater seepage
Manganese Annual Cycle in reservoirs
Lakes/Reservoirs have seasonal overturn
heating throughout spring and summer creates thermocline
causes anoxic conditions in bottom water during summer
as water cools, the thermocline breaks down and water recirculates
dissolved Mn spikes from Jun-Oct
Internal Loading for Mn
Mn stored in bottom sediments is released during thermal stratification and anoxia
Algal production relation to Mn + As
Can drive Mn cycling in lakes, surface photosynthesis promotes Mn oxidation, sinking organic matter fuels Mn reduction
As adsorbs to Fe and Mn, algal deposition enhances Fe/Mn reduction, increasing As mobilisation
Speciation determines mobility and toxicity
Redox stratification - Mn
Creates a Mn shuttle: Mn2+ oxidises in oxic waters and is reduced back to Mn 2+ in anoxic layers
Carbon cycling - Mn
Mn cycling is coupled to carbon cycling, changes in productivity directly regulate Mn redox dynamics
In-Situ solutions
HOx system - oxygenates bottom of lake, but Mn continues to leak out of bottom sediments
Resmix - causes clouds of resuspended sediment
Water lifting aerators - reduce elevated levels of Mn and Fe, prevented hypoxia and caused release of reduced Mn and Fe species
Spectroscopy
Sample preparatiom
Atomization
Light absorption
Detection and quantification
Calibration and Analysis
Colorimetry
Sample preparation, reagent addition, measurement, and calibration curve
beer lambert law: A = epsilon*c*l
ICP-MS
Ionises sample in plasma, measures mass to charge ratio
Can detect multiple elements, precise
Pb in drinking water
Current and legacy sources can be mobilised into freshwaters
Main method of controlling Pb is phosphoric scid, which reacts w highly soluble PbCO3 to make a stable solid btw. pH 6-10
Phosphate creates protective layer inside water distribution pipes
Mn future risk
May pose a risk to potable water due to in-pipe cycling
Speciation controls mobility and toxicity
Chemical form determines solubility, sorption, redox behaviour, and biological uptake
Redox conditions govern metal release
Oxic conditions stabilize Fe/Mn oxides, anoxia drives reductive dissolution and metal mobilisatiom
Organic matter links biogeochemistry to contamination
Eutrophication increases oxygen demand, which shifts redox amd releases sorbed metals like As
Solid phases are dynamic, not permanent links
Metals adsorbed to sediments or incorporated into corrosion scale can be released when chemistry changes
Water chemistry drives infrastructure corrosiom
Chloride, alkalinity, oxidants, and corrosion inhibitors determine metal release from pioes
Small chemical shifts can have large public health impacts
Changes in source water or trestment can destabilise equilibria and increase dissolved metal exposure
Nanoparticles
Structures w at least one dimension of 100 nm or less
Nanomaterials
Nanoparticles in a form that serves a particular function
Nanoproducts
Commercial products that include or incorporate nanomaterials distributed in a matrix
Nanomaterials are categorized into 4 types
inorganic based
carbon based
organic
composite based
Nanomaterial size dependent chemical characteristics
as we increase diameter, we have lower surface bonding area/reactivity
melting point increases, energy needed increases
As adsorption on iron oxide
The smaller diameter means As is inside lattice, larger diameter particles localize As on surface
Nanoparticles - Physicochemical properties - shape
sphere
rod
sheet
Nanoparticles - coatings
various coatings are used with different properties, they are important for dictating environmental fate
citrate, pvp, DEXTRAN, casein
Nanoparticles - how do we measure?
number of particles can vary greatly but yield same overall mass based concentration
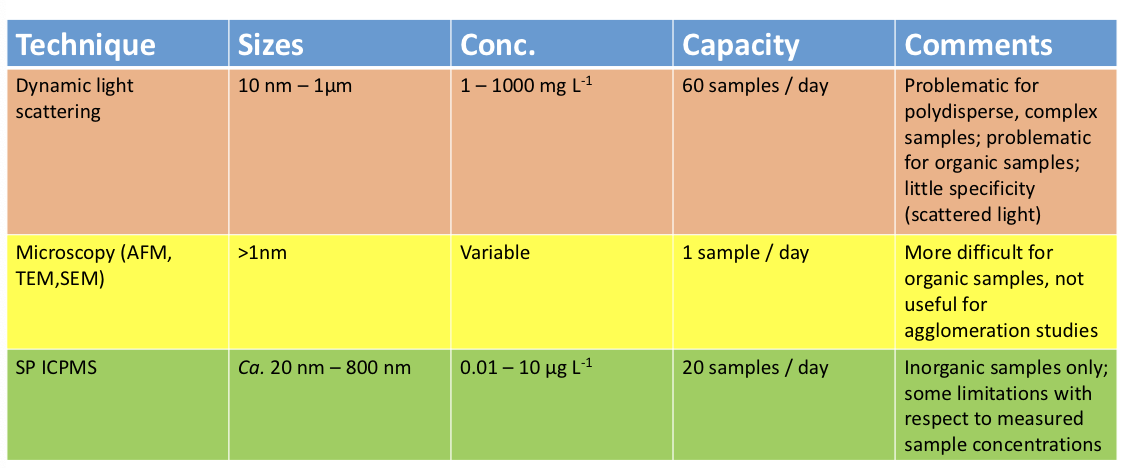
DLS (Dynamic Light Scattering)
laser beam is sent to sample, diffusion coefficient of particles is determined
related to the hydrodynamic diameter of particles thru stokes-einstein eq.
provides average particle size and size distribution
TEM (Transmission electron microscopy)
samples are fixed using grid preparation techniques
measures width of particles
AFM (Atomic Force Microscopy)
sharp tip that interacts with sample surface
measures topographical features to about 1nm in all 3 axes
SEC (Size Exclusion Chromatography)
separates particles based on hydrodynamic volume
more effective for spherical and homogenous size - less effective for wide size range
SP-ICPMS
should not be acidified, continuous signal of lower intensity
pulses of higher intensity are nanoparticles
Nanoparticles - main problem is distinguishing smallest particles from background noise
Dmin = (6×3sigma)/(R* fa* rho * pi)
sigma = sd of background
R = instrument sensitivity
fa = elemental mass fraction
rho = particle density
strategies to reduce background
ion exchange resin can remove charged molecules from solution
Techniques for detecting and quantifying nanoparticles
Natural, incidental, and engineered nanoparticles
natural: made by nature through biogeochemical or mechanical processes
incidental: unintentionally produced by anthropogenic processes
engineered: conceived, designed and intentionally produced by humans