CHEM 4631 Exam 4 Lecture Slides 26
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
What is the function of the mobile gas carrier in GC?
To carry the sample through the system
What should an ideal mobile gas carrier be?
Inert
Dry
Thermally stable
Cost effective
Safe
Compatible with the detector
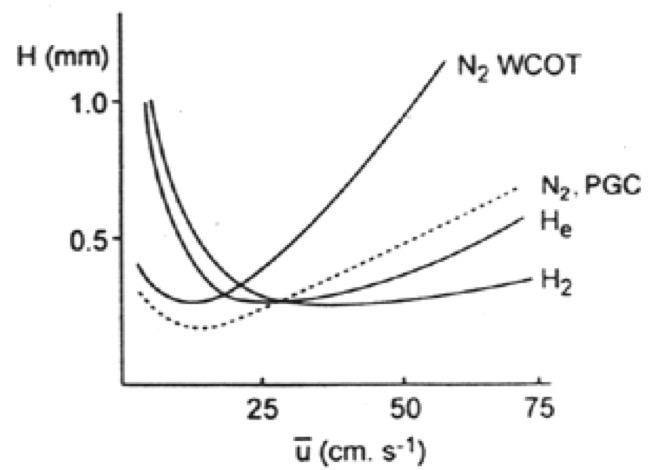
What can be said about N2 when it comes to separation efficiency according to the graph? Why?
N2 has the highest separation efficiency
N2 has a greater molecular weight and smaller diffusion coefficient; so lower B
However, this sacrifices analysis time
What advantage does H and He have over N?
Both have a better analysis time, but a small sacrifice in efficiency.
He → 16-20 cm/sec
H → 35-40 cm/sec
How is deterioration of the Stationary Phase reduced?
A high purity gas needs to be used.
For this reason oxygen and moisture traps in the carrier gas lines are used.
What Two Variables describe the Flow of Carrier Gas?
Flow rate (measured in ml/min)
Pressure drop between the injection port inlet and detector outlet
How is the Carrier Gas regulated?
A flow controller
What is the function of a Scrubber Tubes in GC?
Cleans mobile gas
Gets rid of Oxygen, Hydrocarbons, and Water to prevent contamination interferences in results
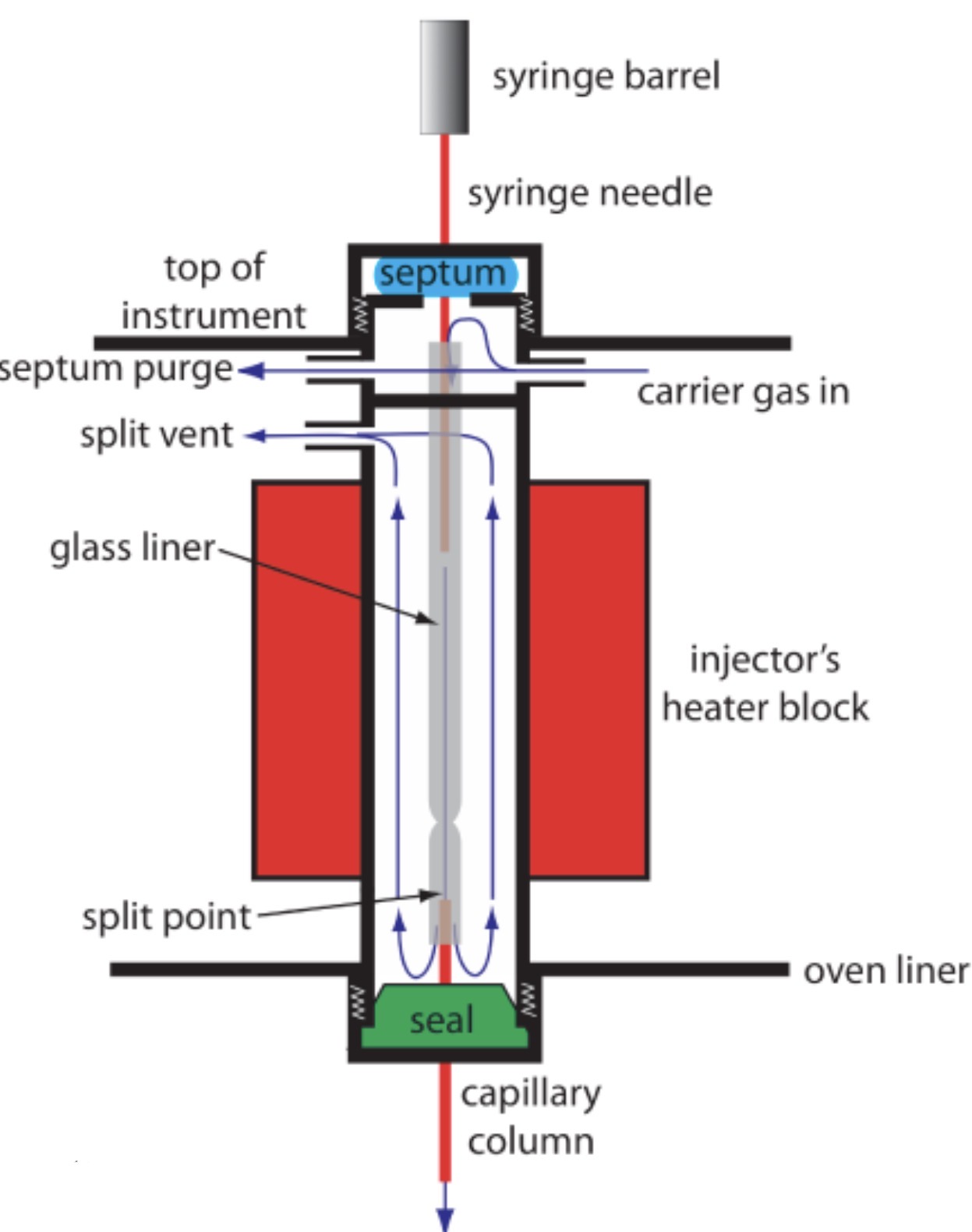
Which parts of a GC system are set at different temperatures?
Injection Port
Oven
Detector
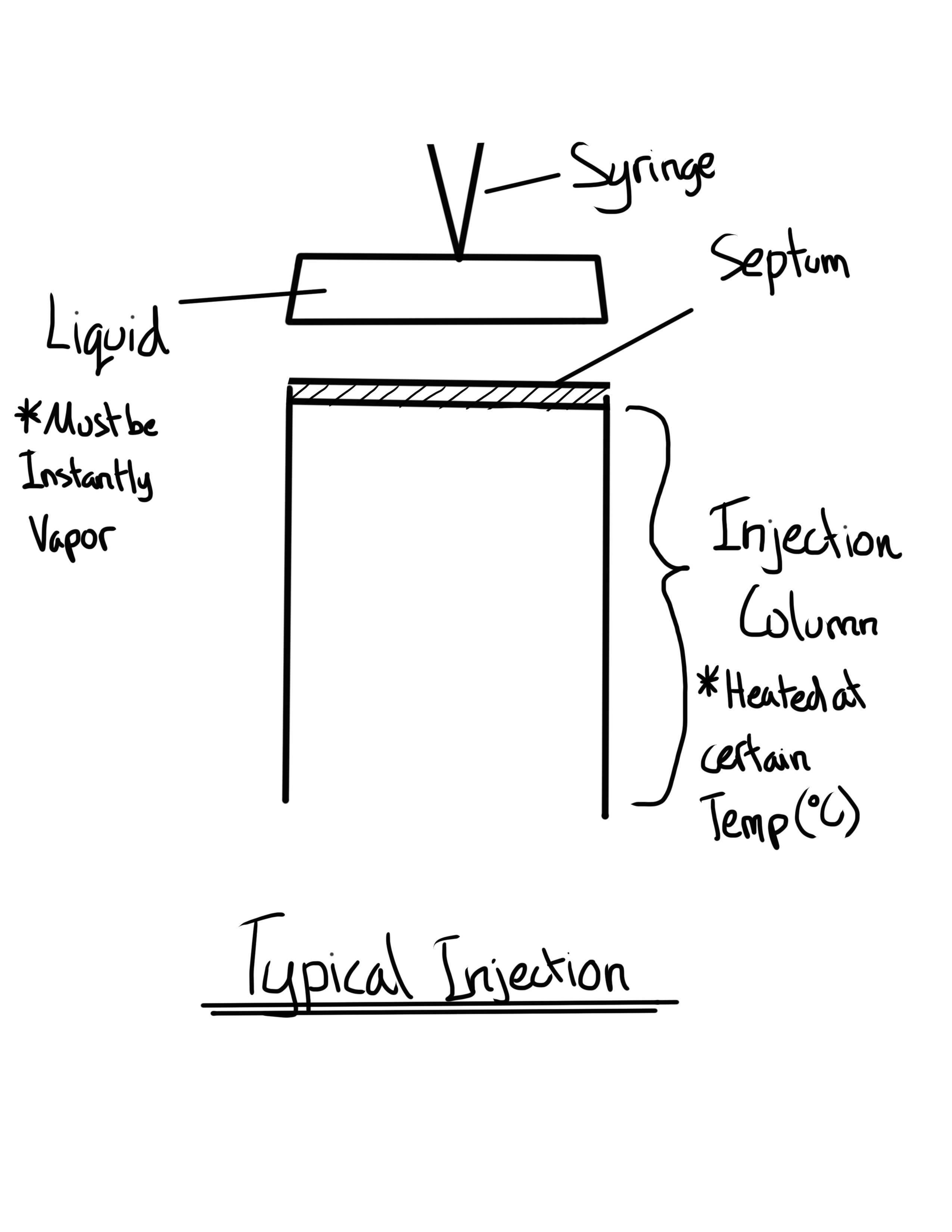
Why is the Injection Port set at a high temperature?
To instantly vaporize the sample before reaching the column
How does the Injection Process influence Peak Widths?
Dependent on the Injection Length of the Sample
The shorter the length of the column occupied by the injected sample, the shorter the band as it begins and completes the chromatographic process
What is the critical function of the Injection Process in Chromatography?
Introduce the sample so that it occupies the shortest possible length of column
What is the Syringe Technique?
Universal method of introduction is with a through a septum.
Why is reproducibility poor when using Syringe Techniques? How can this be remedied?
Because the volume of gas is temperature dependent
Use of a sampling valve increases precision
What is the Hot Needle Syringe Technique?
Sample in barrel, allow the sample to heat up using the injection prt
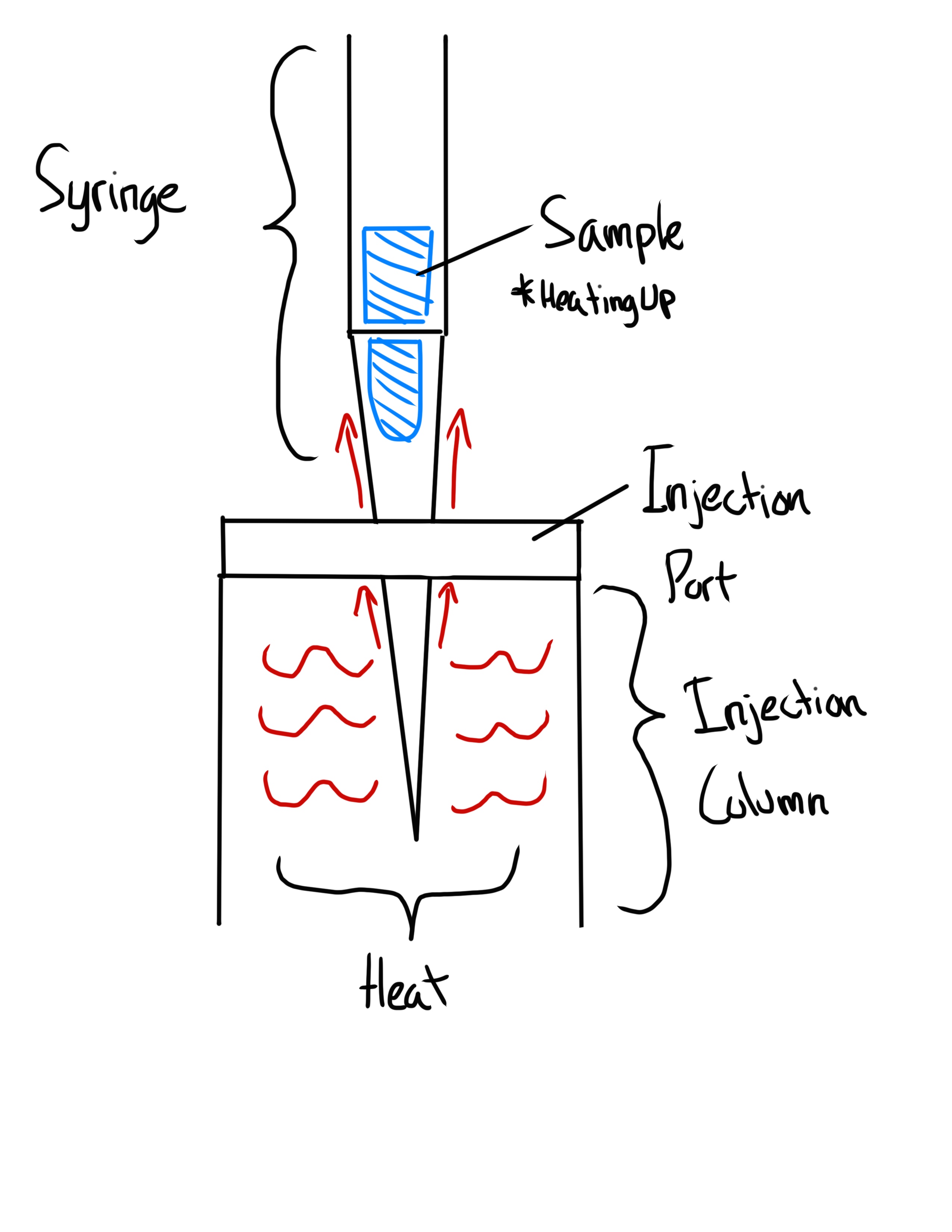
What is the Cold Needle Syringe Technique?
Sample is injected immediately without heating up unlike in Hot Needle
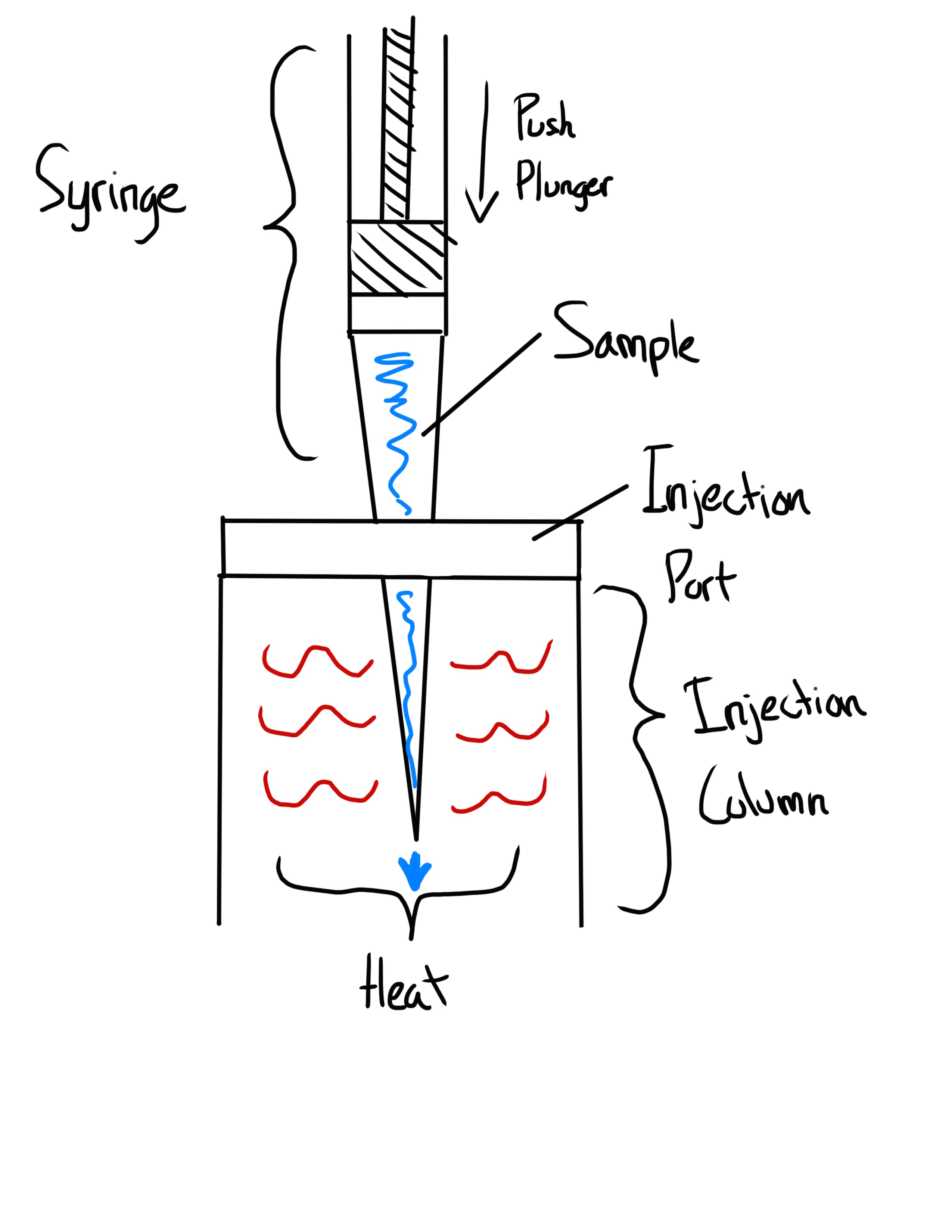
What is the Solvent Flush Syringe Technique?
Sample is “Sandwiched” with Solvent
Alternatively: Solvent+Air+Sample+Air
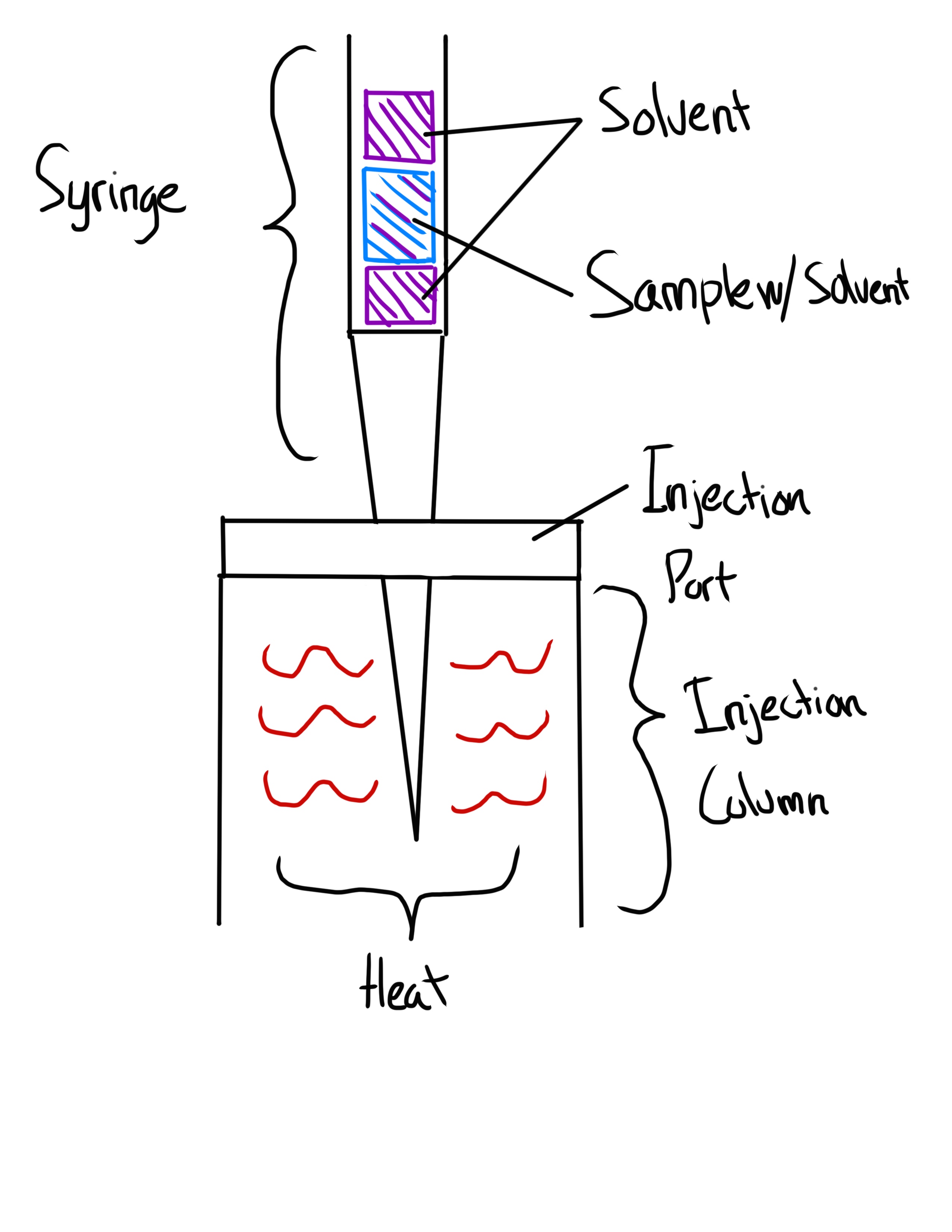
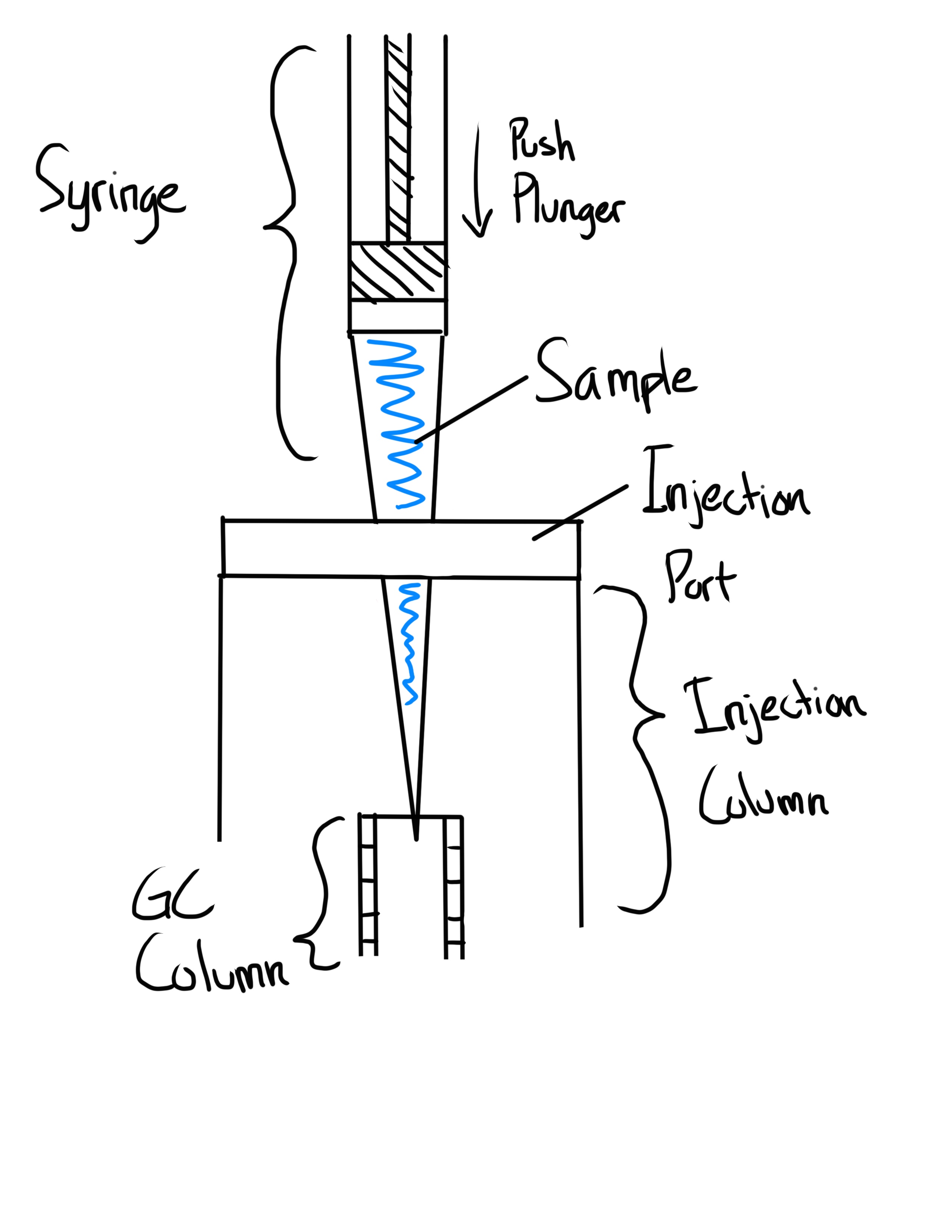
What is the On-Column Syringe Technique?
Sample is directly injected to the column
Most efficient Syringe Technique, but damages the column
What chemical property of a sample determines the syringe technique to use?
Molecular Weight
What is a Split Injection?
After the sample is evaporated and mixed with the carrier gas, the sample splits into two unequal portions.
The larger portion is “purged” (waste)
The smaller portion goes into the column
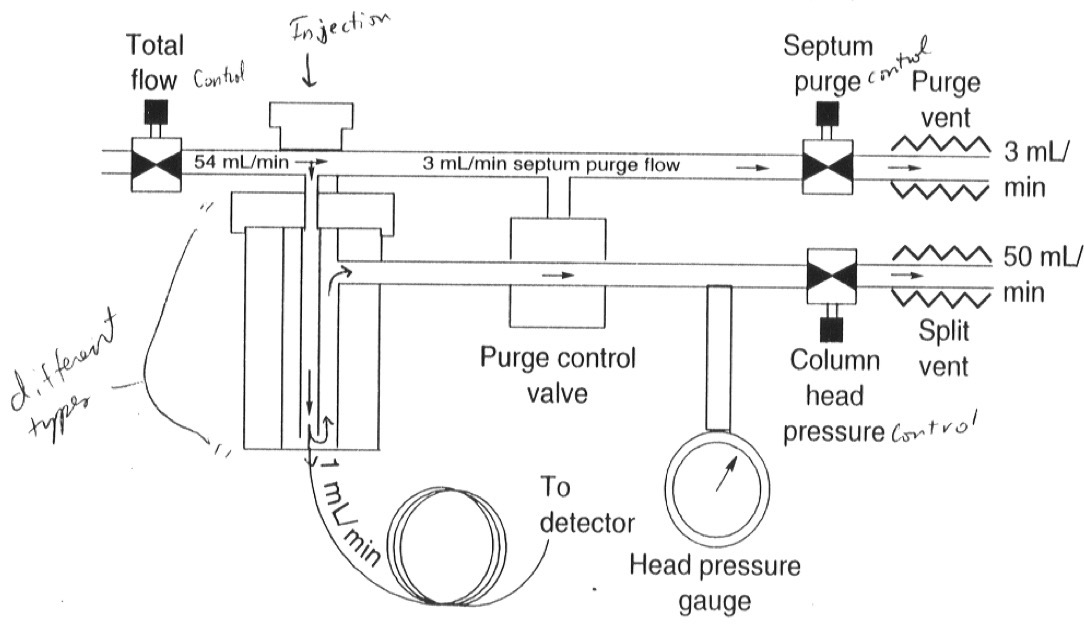
What is Split Injection used for and why?
Highly concentrated samples
Capillary Columns have limited sample capacity
Prevents overload of the column
What is the equation for the Split Ratio of a Split Injection?

What is the disadvantage of a Split Injection?
High probability of the split inlets suffering from sample discrimination
What is a Splitless Injection used for?
For ultratrace analysis for complex samples with wide range of boiling points
Bonded Columns as most of the solvent goes into the column
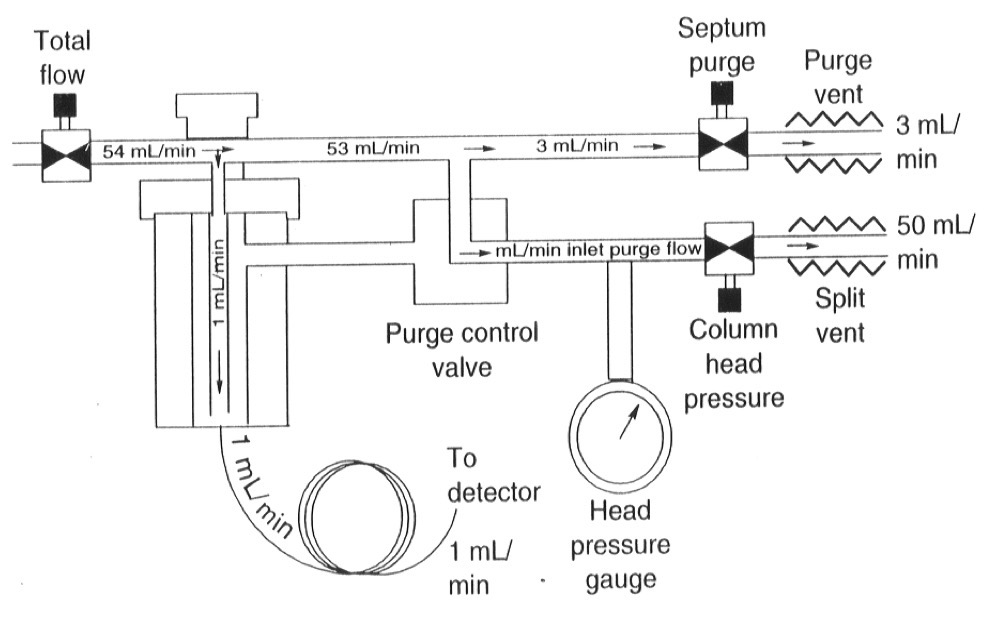
What are the two types of Splitless Injection?
Programmed Temperature Vaporization (PTV)
Cold-on-column injection (On-Column)
What happens in a PTV?
Cold initial column temperature condenses the analytes into a narrow band at the column head. As the column is heated, the analytes begin to move down the column one by one, determined by their heat of vaporization from the stationary phase to the mobile phase.
It is a universal injection system
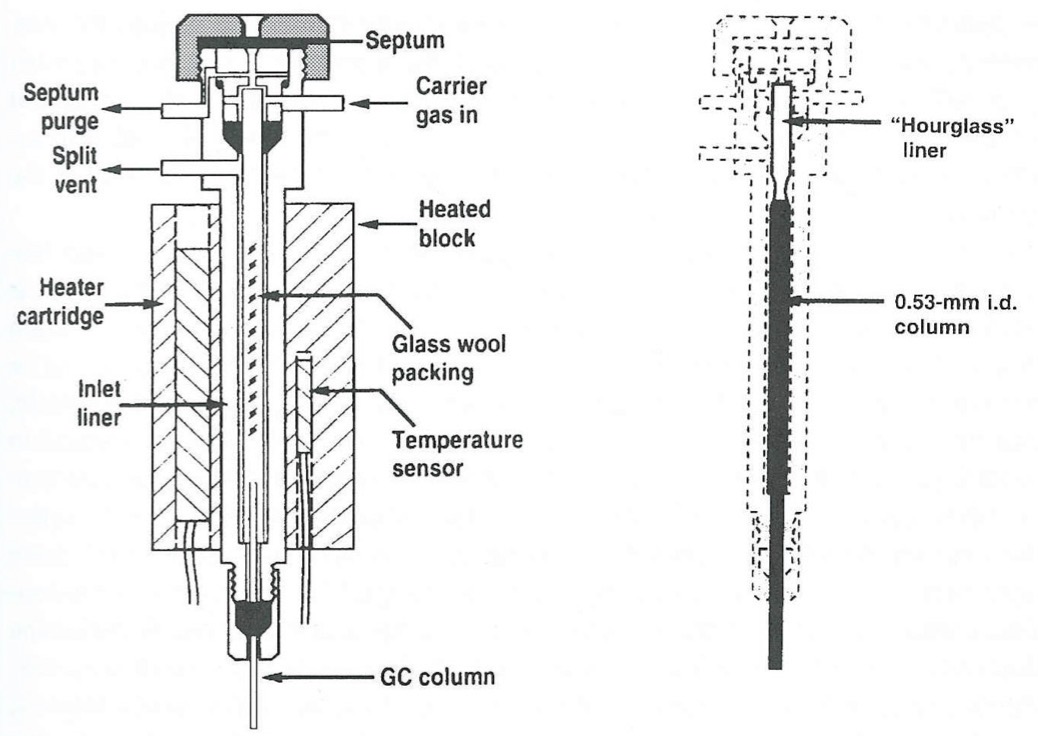
What happens in a Cold-On-Column Injection?
Reduces solute decomposition and discriminatory effects if the injection is rapid.
What is the disadvantage of a Cold-On-Column Injection?
Non-volatile material in the sample will enter the column and remain there
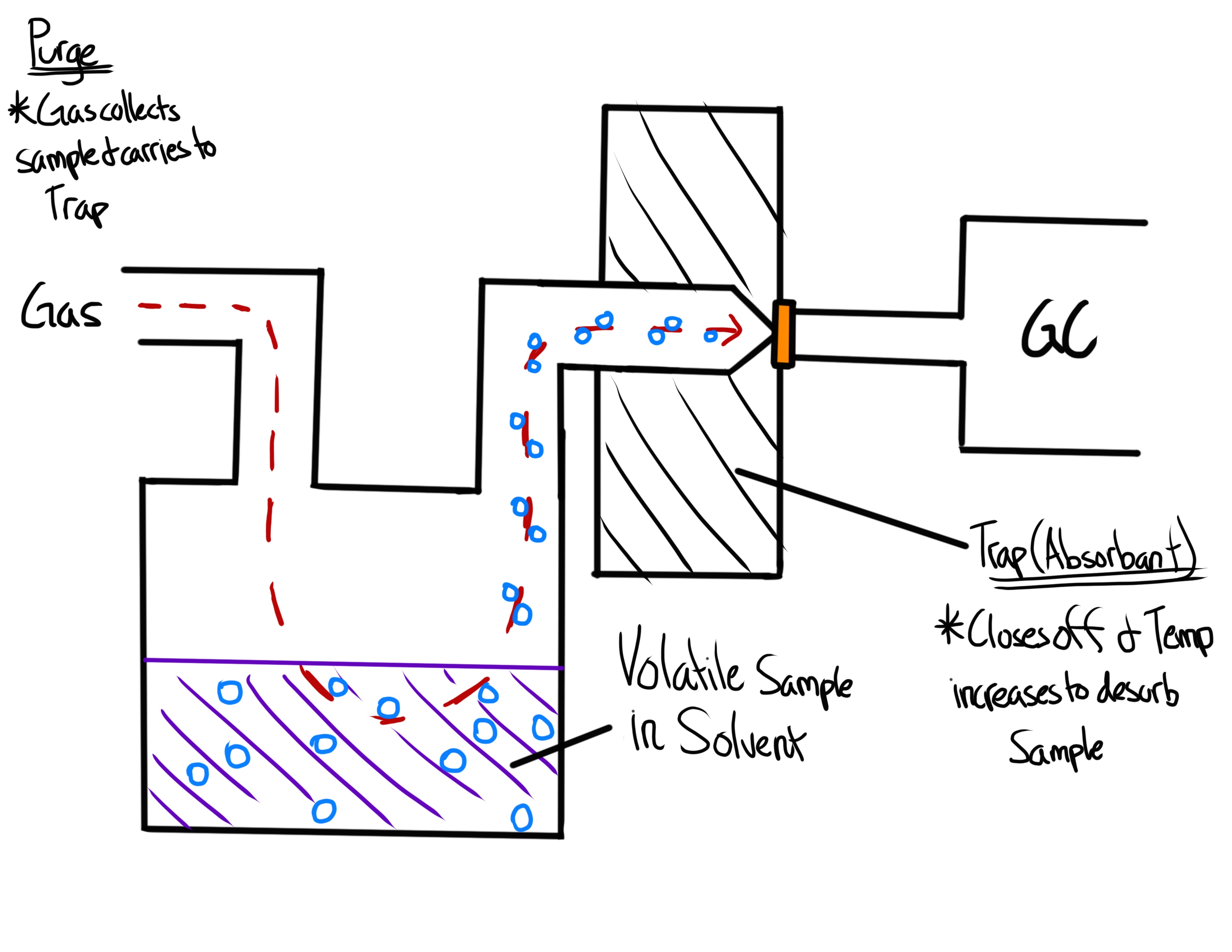
What is the Purge and Trap Method used for?
Analyzing low levels of volatile organic compounds from matrices
What happens in the Purge and Trap Method?
A sample is mixed with a purge gas and is purged at a specific temp.
The sample is carried by the purge gas and into a trapping material
After enough sample is collected, the trap is heated and the sample is desorbed and swept to the column by the carrier gas