The Diels-Alder Reaction of Anthracene and Maleic Anhydride Lab 4
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
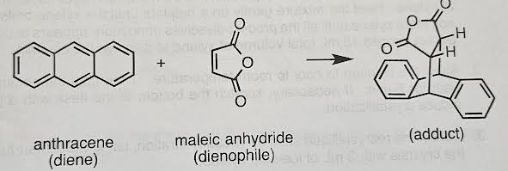
what is the diene?
the 4 pi component. it will have two pi bonds (2 double bonds) dienes are electron rich and are nucleophiles. Electron donating groups will enhance the diene’s reactivity. The diene in this reaction is the anthracene
what is the dienophile?
The dienophile is the 2 pi component that has electron poor double bonds or triple bonds. Dienophiles need at least one strongly electron withdrawing group. Dienophile will be maleic anhydride
stereochemistry of Diels Alder product
the stereochemistry will be the same as the reactants.
why is maleic anhydride a good dienophile
maleic anhydride has two strongly electron-withdrawing groups that are attached to the double bond.
why is anthracene the diene even though it has aromatic rings
it can be thought of an aromatic compound but it isnt as stable as an isolated benzene ring and also it has only 14 pi bonds as opposed to the 18 that would be in 3 aromatic rings
why is the reaction carried out in xylene
1: it has a 140 celcius boiling point which will withstand the reaction temperature. 2: the xylene will not freeze in cold water 3: the reactants are soluble in xylene while the product is not and the product will crystallize out so it is easy to separate the product
The product is an anhydride and anhydrides will react with water in the air, so why if this product still good
This anhydride will react very slowly so its a good product
so what happened?
anthracene. maleic acid, and xylene were refluxed for 30 minutes. The xylene wont boil until 140 celcius so it will provide good reaction temperature. The precipitate was obtained by cooling the mixture in ice bath. The xylene will not freeze and will keep the solutes dissolved so only the adduct will crystallize. Then the adduct was filtered out and recrystallized with xylene.
how to know the reaction was successful from the IR?
The product has carbonyl peaks, ester peaks, and an aromatic ring
why cant we recrystallize with water or alcohol
anhydrides will undergo hydrolysis if it encounters alcohol or water