DAT Orgo forgettables
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
what does Na2Cr2O7 oxidizes primary alcohols into?
carboxylic acids
what does Na2Cr2O7 oxidizes secondary alcohols into?
ketones
ortho/para directors
(things with lone pairs) -NH, -OH, -OR, -R (alkyl), -benzene ring
meta directors
(things with a double bond to O) -NO2, -CN,
what is another name for asymmetrical carbon
chiral center
protic solvents
solvents that have H atoms bonded to O, N, or S. Examples include: CH3OH, CH3CH2OH, CH3COOH, H2O
aprotic solvents
do not react with nucleophiles, so that they can proceed down the SN2 or E2 pathway. Examples include: DMSO, acetone, DMF, THF, ether
what type of solvents do SN1 and E1 reactions prefer?
polar protic solvents
what type of solvents do SN2 and E2 prefer?
polar aprotic solvents
is NO2 an ortho/para or meta director?
meta director
Is NH2 an ortho/para or meta director?
ortho/para director
what is the rate for SN2 reactions?
methyl > 1 > 2 > 3
what type of reactions use aprotic solvents?
E2 and SN2
are meta directors electron withdrawers or donors?
withdrawers
do meta directors increase or decrease acidity?
increase acidity
are ortho/para directors electron withdrawers or electron donors?
electron donors
do ortho/para directors increase or decrease acidity?
decrease acidity
is CH3 an ortho/para or meta director?
ortho/para
aldol condensation
combines two carbonyl groups to form a beta-hydroxyl carbonyl
what is required for aldol condesation to occur?
an alpha hydrogen
what is an alpha hydrogen?
a hydrogen bonded to a carbon that is immediately adjacent to a carbonyl carbon.
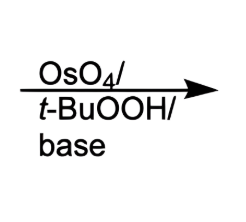
what do these reagents make?
a syn diol (two OHs facing the same direction as each other)
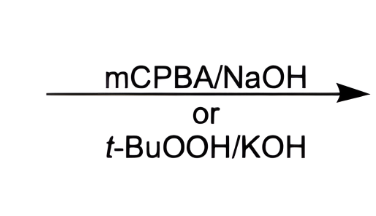
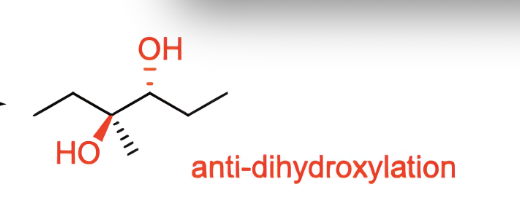
what do these reagents make?
an anti diol
what are swern oxidation and PCC do?
they are oxidizing reagants that change primary alcohols to aldehydes and secondary alcohols to ketones
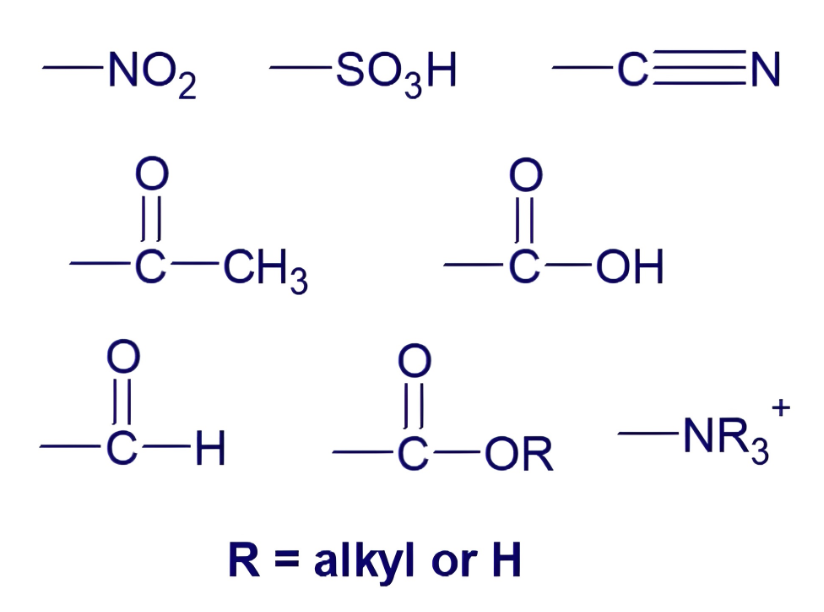
what are these
meta directors
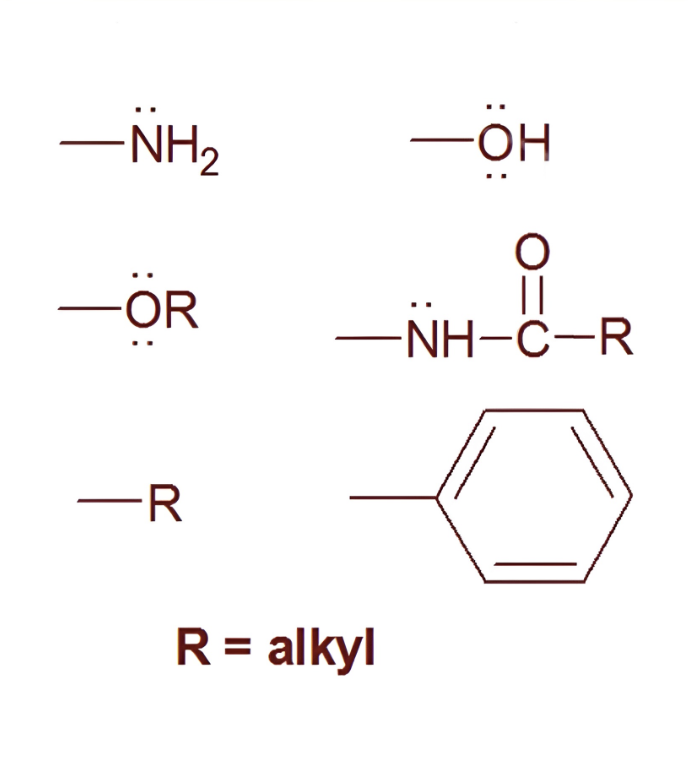
what are these
ortho/para directors
what do electron withdrawing groups (meta groups) do?
draw electron density out of the aromatic pi-system, thereby decreasing the reactivity of the benzene (deactivate rings)
what do electron donating (ortho/para) groups do?
make an aromatic ring more reactive
thermodynamic product
the major product at higher temperatures
which type of reaction gives a stereochemical inversion of the configuration?
SN2
degrees of unsaturation
number of pi bonds + number of rings
which type of solvents are favored for SN2 reactions?
polar aprotic solvents
what does H2, Lindlar do?
converts an alkyne to a Z-alkene
what does Na, NH3 do?
turns alkyne into E-alkene
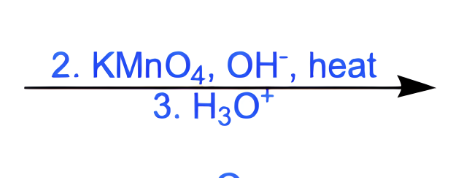
what does this do
makes a carboxylic acid
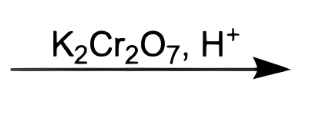
what does this do
convert secondary alcohol to a ketone
the more s character…
the more acidic
electrophilic aromatic substitution
a type of chemical reaction in which an atom, usually a hydrogen, attached to an aromatic ring is replaced by an electrophile
electrophile
has atom with + charge, has electron withdrawing group
nucleophile
has a - charge, has nonbonding electron pairs
what is true about SN1 reactions and aromaticity
aromatic compounds undergo SN1 reactions faster than anti-aromatic compounds because they are much more stable
wolff-kishner
treats a ketone or aldehyde, reduces all the way down to CH2
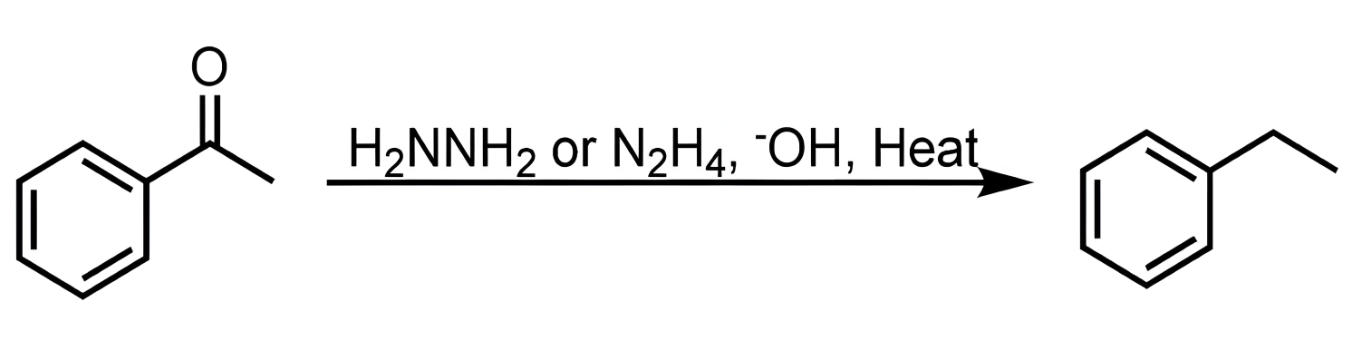
how do you determine which nucleophile is strongest?
whichever one has the strongest, most concentrated lone pair with no resonance stabilization
decantation
the process of tilting a mixture to pour out the top layer without disturbing the bottom layer. This only works if you have two immiscible components
recrystallization
process used to purify solid compounds. If you have a solid dissolved in a liquid, you could use recrystallization to heat the solution, then gradually cool it to precipitate the solid out
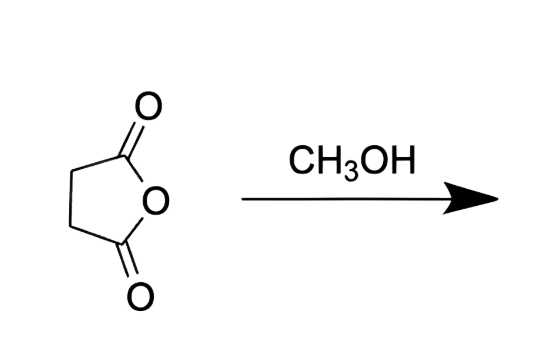
what does this reagent do?
puts an OH and ester on the same molecule
the higher the energy conformation…
the less stable the compound is
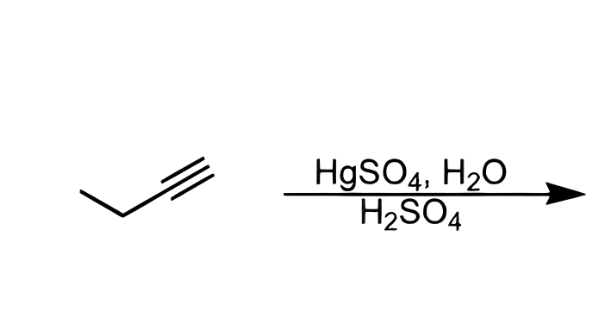
what does this reagent do?
creates a ketone
extraction
a procedure used to separate substances based on their solubility in different aqueous vs. organic solvents
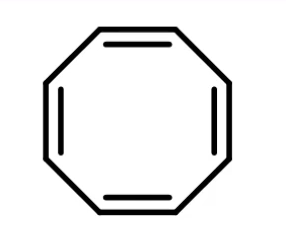
what is this
non-aromatic
Which of the following is the most basic?
H3C-