NEW Lab 7
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
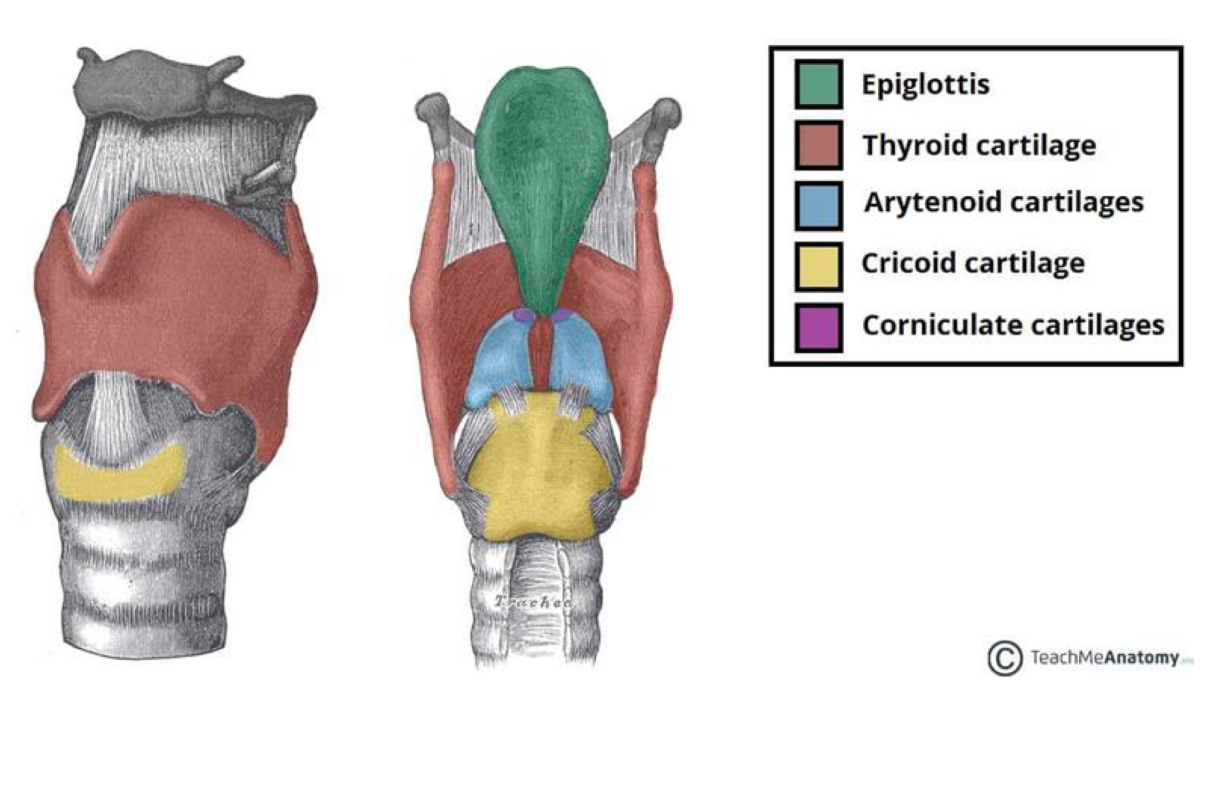
Larynx Anatomy
The Adam’s apple (Laryngeal Prominence) is from an enlargement in the thyroid cartilage!
The epiglottis keeps food from entering the airway
Unpaired: epiglottis, thyroid cartilage, and cricoid cartilage
Paired: Arytenoid cartilage and corniculate cartilage
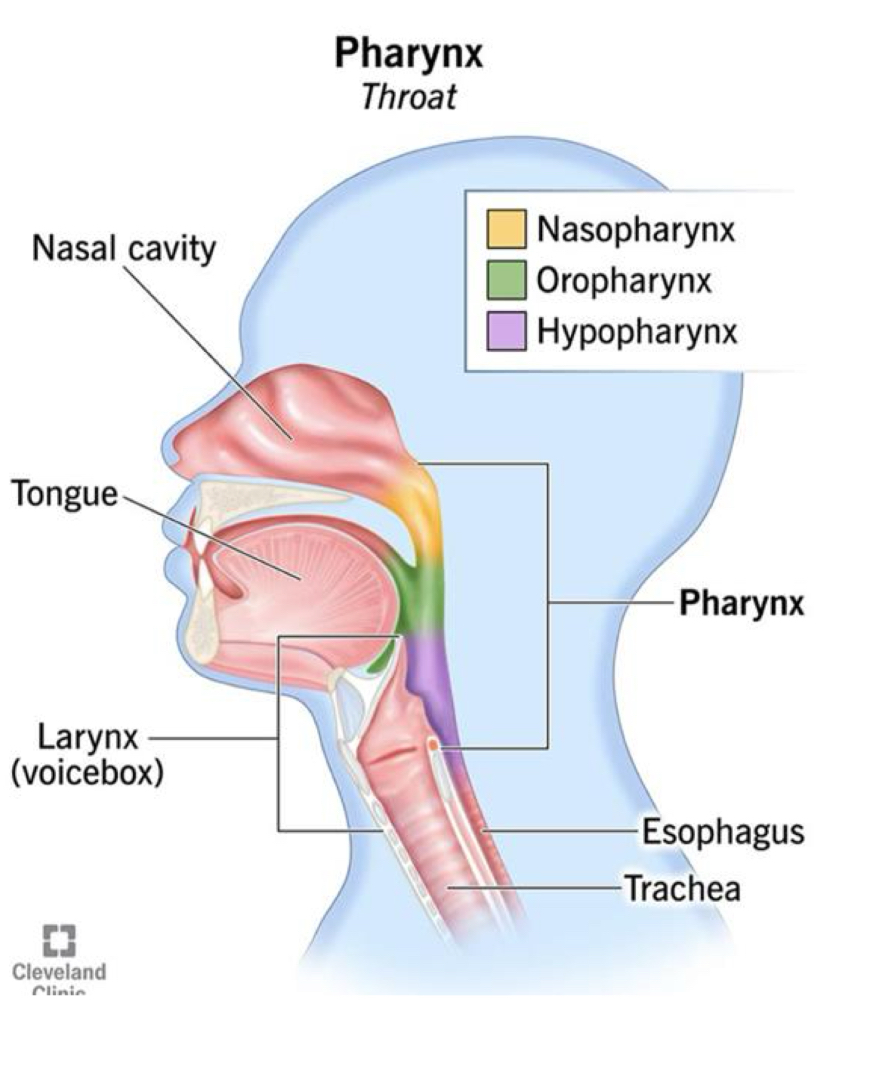
Pharynx Anatomy
The pharynx contains stratified squamous epithelium
The nasal conchae are in the naval cavity and increase surface area to humidify and warm inspired air
The oropharynx is a shared passageway for air and food
The esophagus receives food and lies posterior to the trachea
Respiratory System Overview
Bring oxygen into the body and remove carbon dioxide.
Air moves in and out as the lungs expand and contract during breathing.
Alveoli are tiny air sacs where gas exchange happens.
Surfactant (a lipoprotein complex formed by alveolar cells) is a substance that reduces surface tension in the alveoli
Surfactant helps keep the alveoli from collapsing, keeps the lungs dry, and makes breathing easier.
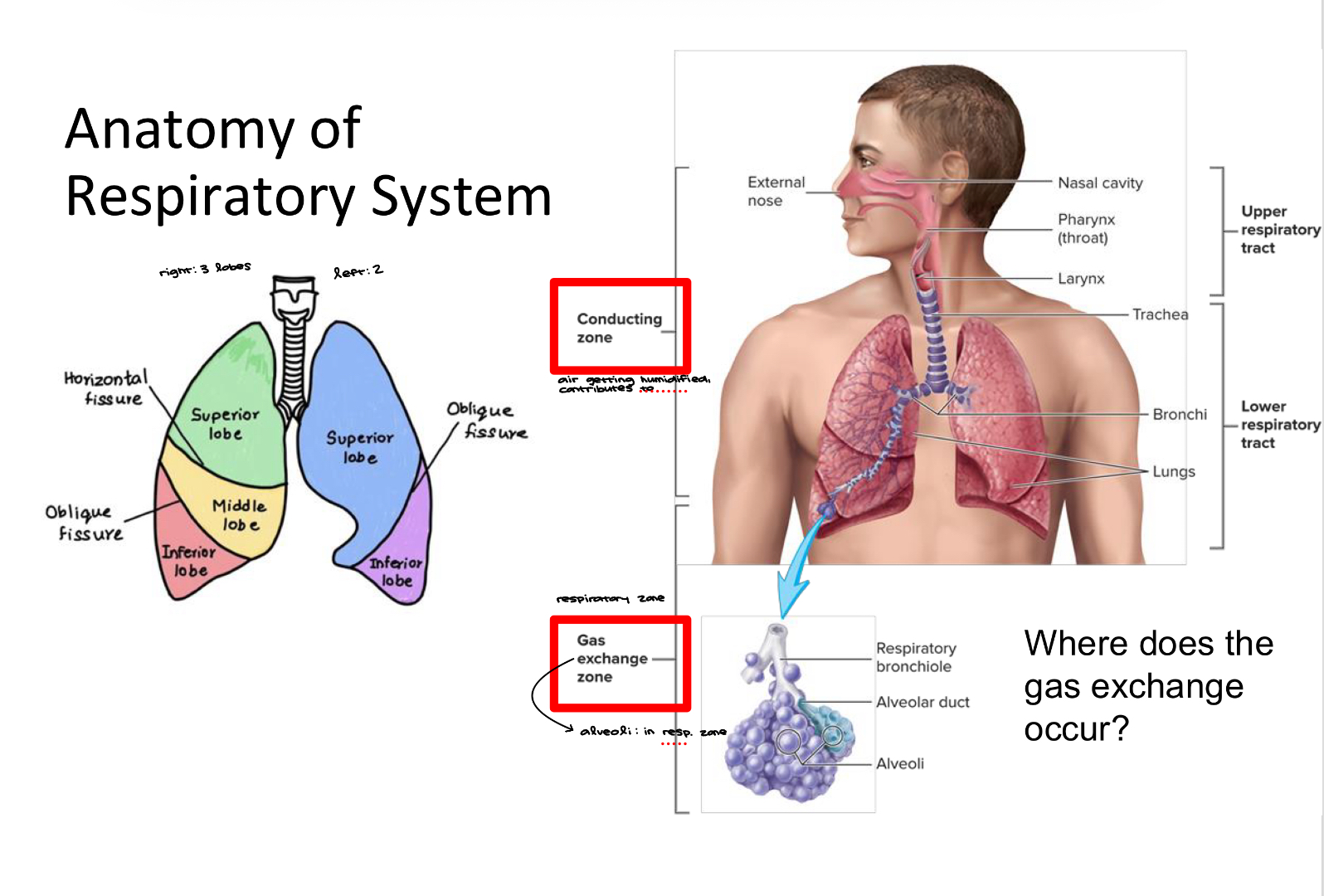
Anatomy of the Respiratory System
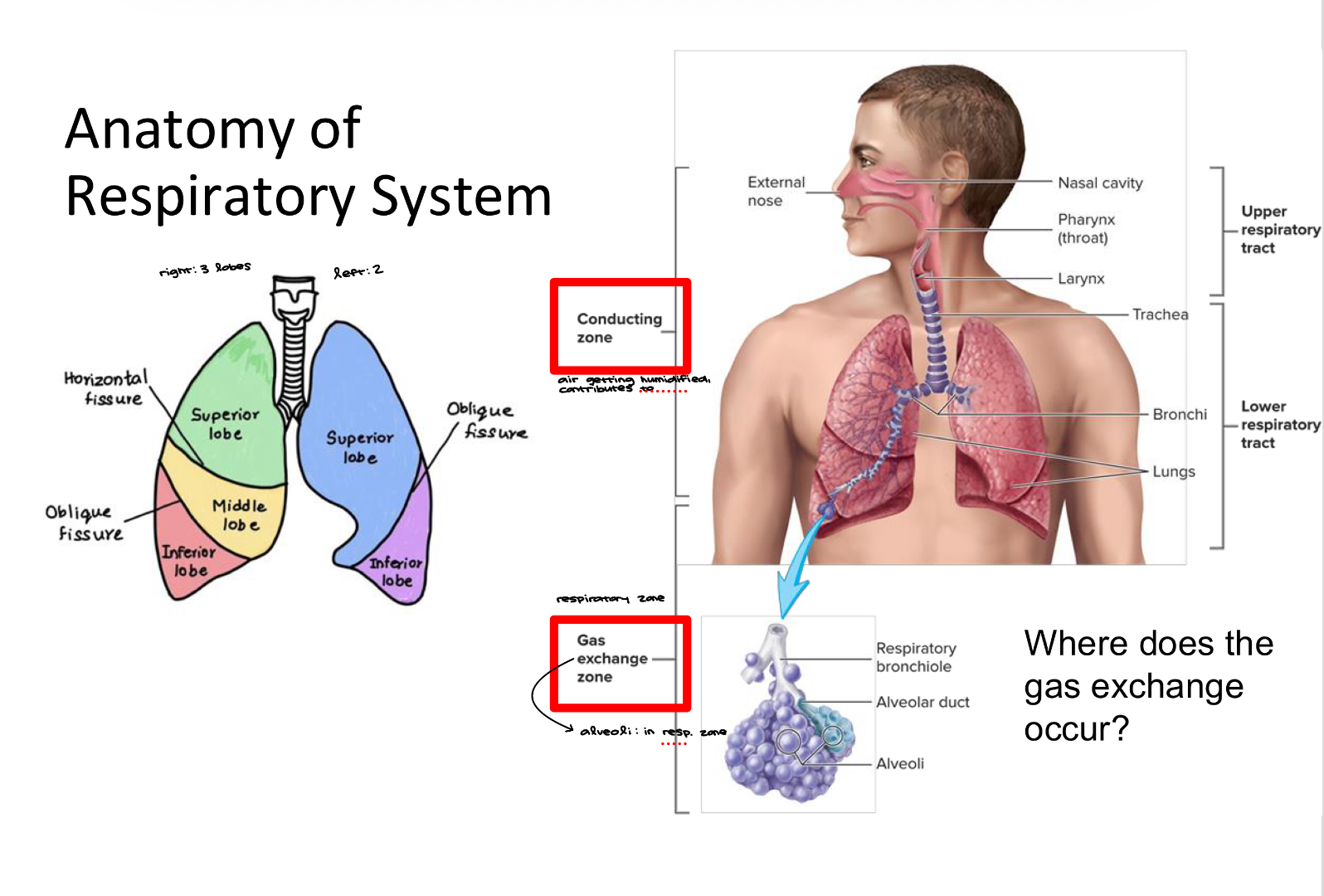
Anatomy of the Respiratory System 2
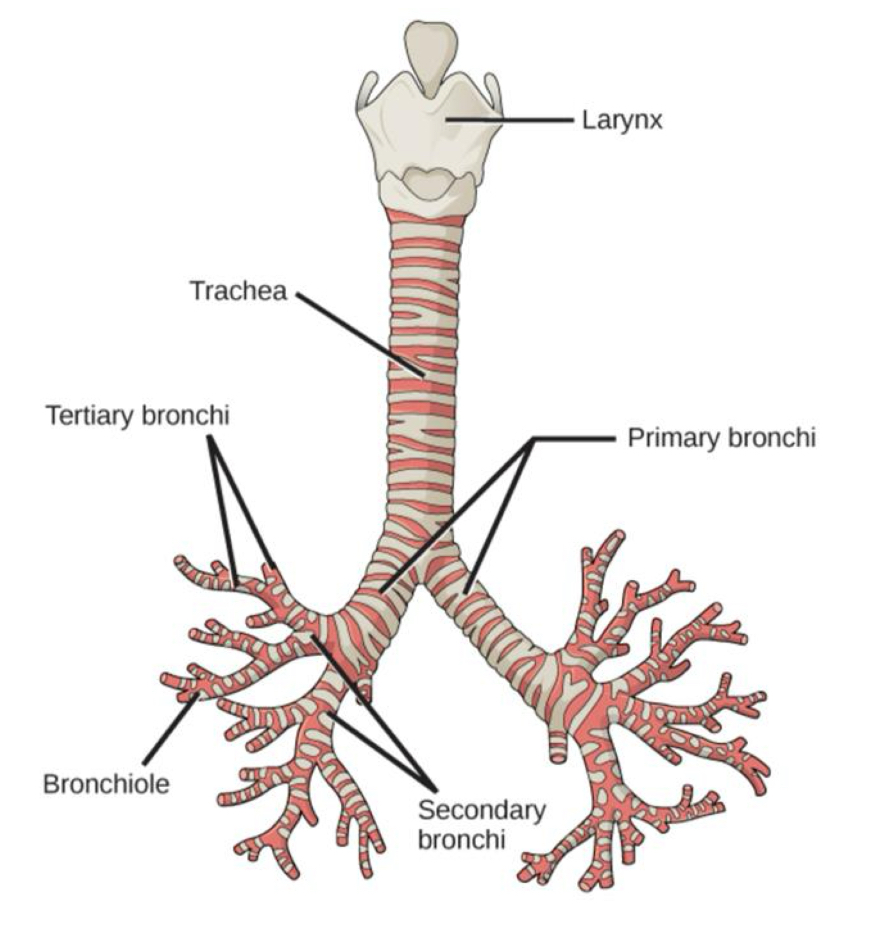
Anatomy of the Respiratory System 3
Gas exchange occurs in the bronchioles and the alveoli
Intrapulmonary pressure: the pressure in the alveoli
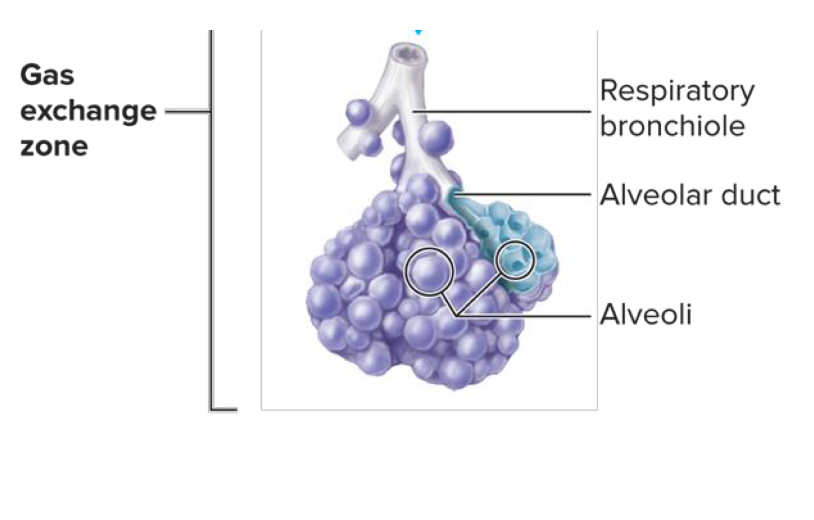
Physics of Gas Exchange
Henry’s Law: a gas will dissolve into a liquid in proportion to its partial pressure and at a speed dependent on the concentration difference between the gas and liquid
Dalton’s Law of Partial Pressures: the total pressure exerted by a mixture of gases will equal the sum of the partial pressures exerted independently of each of the gases in the mixture
Boyle’s Law: at constant temperature, the pressure of gas varies inversely with its volume