Case Series
1/6
Earn XP
Description and Tags
Week 4 -> Dr. Ekong
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Case series
A descriptive, observational study that’s used to illustrate an aspect of a condition, treatment or adverse reaction to treatment, in a group of patients with the disease or outcome of interest.
Data from a cluster or series of cases are reported without a comparison group (i.e., no control group); instead, the exposures of the cases are often described.
e.g., ‘Metoprolol-induced visual hallucinations’
3 patients (case 1, 2, 3) were reporting visual hallucinations
After discontinuation of the metoprolol, the visual disturbances resolved within several days
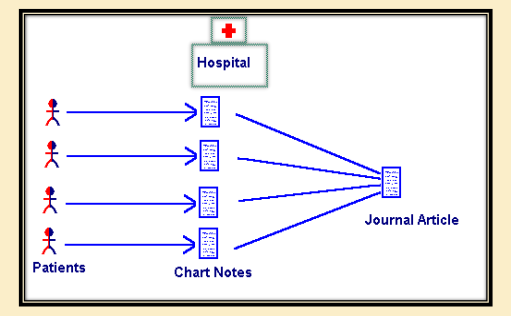
When are case series used?
To describe outcomes of a new treatment
e.g., “A clinician examines a patient receiving a specific medication who develops a rare side effect to the medication. The clinician will search for cases/patients with similar side effects to the medication and identify patterns.” (this process could help given an intervention/recommendation regarding the direction of treatment or diagnosis)
Descriptive/Qualitative Data
Reports of case series usually contain detailed information about the individual patients
Demographic information
Information on diagnosis
Treatment
Response to treatment
Follow-up after treatment
What are the strengths of case series?
Informs patients and physicians about natural history and prognostic factors
The information gained from a case series can be used to generate a hypothesis
It's a feasible study design
Easy, inexpensive, and less time-consuming
Can help answer the question of why this outcome occurred
What are the weaknesses of case series?
There is no comparison (control) group
Selection bias
Cases may not be representative of the population
Outcome may be a chance finding, not characteristic of the disease
Doesn’t have statistical validity
No cause-and-effect relationship
How to minimize bias in a case series study design?
Clear study objective/question
Well-defined study protocol
Clear inclusion and exclusion criteria for study participants
Specified time interval for patient recruitment
Consecutive patient enrollments
Clinically relevant outcomes
Prospective outcome data collection
High follow-up rate
What are the key similarities/differences between a case series and a case-control study?
Case series
Inexpensive
Group of cases (disease); no control group
Collection → present
Purpose → for generating hypothesis
Statistical validity → no !!
Case-control study
Inexpensive
One group has disease and the other is disease-free (control group)
Collection → past
Purpose → for testing hypothesis
Statistical validity → measure of disease frequency and risk (odds ratio)