MII2 antibodies, mucosal immunity, malaria
1/118
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
119 Terms
which region of an Ab defines the function (class) of the antibody
Constant heavy chain (Fc) region
what defines whether an Ab is membrane bound or secreted
different forms of RNA processing resulting in different carboxyl terminals
does swapping Ab constant Fc region change the specificity for the Ag
no
what are the first Ab isotopes produced by a cell
IgM and IgD
which are the only Abs that can be produced simultaneously by a B cell
IgM and IgD
what Ab is the first response
IgM
how are IgM and IgD produced
differential RNA splicing
how are IgG, IgA, and IgE produced
irreversible change in DNA
can multiple isotopes (G, A or E) be produced at once
no
can a cell produce IgD or IgM after changing to IgG A or E
no
which Ig is responsible for allergies
IgE
which cell controls B cell isotype switching
Th cells
which Ig has the lowest affinity for Ag and why
IgM - produced prior to affinity maturation
what is IgM good at binding to and why
repeating epitopes , due to forming a pentamer
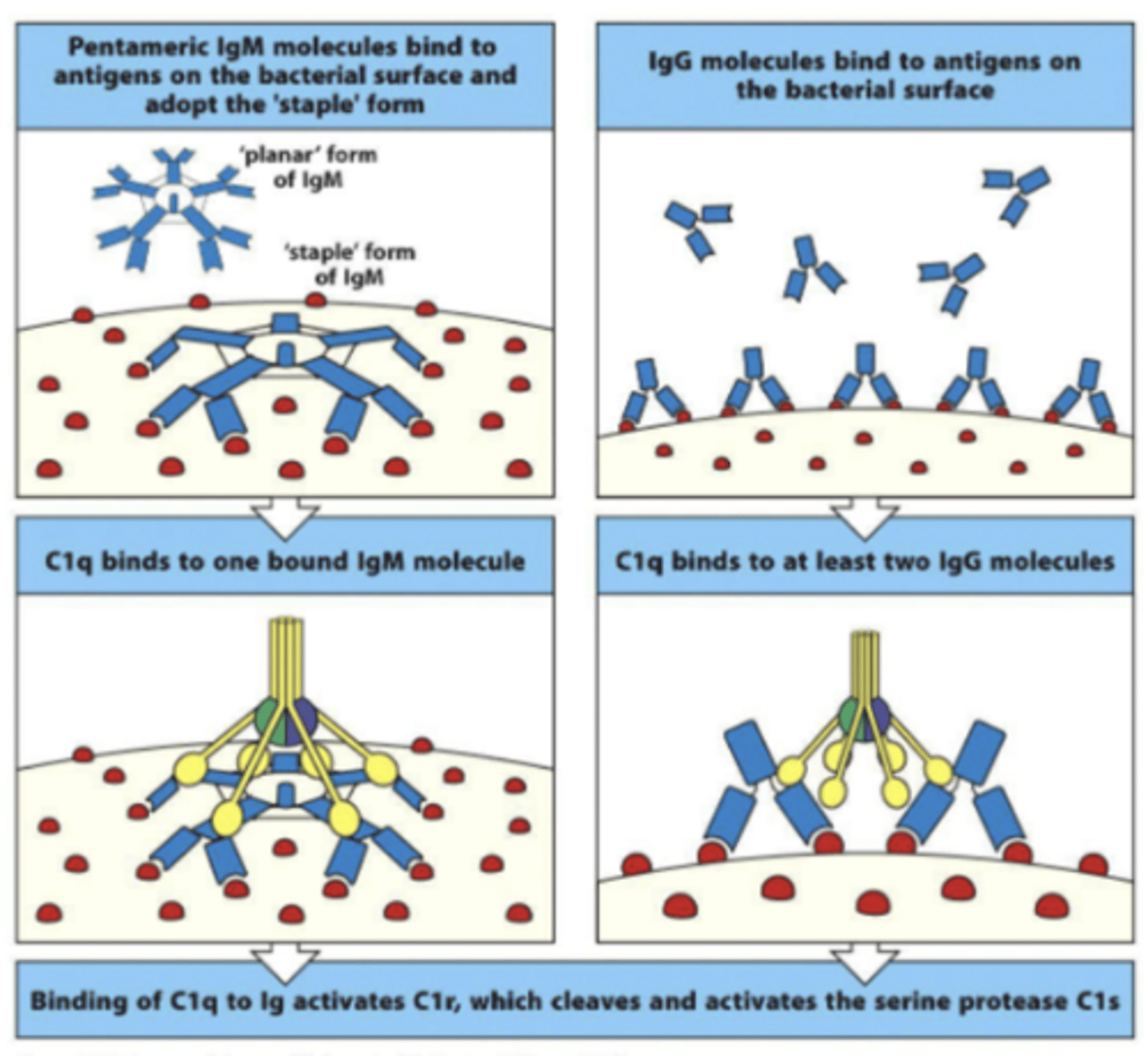
what is IgM good at activating
complement
which pathway do Ab activate the complement via
classical pathway
which region of antibodies activates complement molecules
Fc region
what is a complement fixer
an antibody capable of activating the complement
what makes IgM more effective at activating the complement than IgG
its pentameric structure
what environment is IgA best suited to
mucosal surfaces
which region of IgA facilitates transport across the intestinal epithelium
J chain
what is IgA resistant to (2)
low pH and digestive enzymes
How does IgA keep pathogens away from epithelium?
it clumps pathogens together for explulsion from the body
what makes IgA less likely to cause collateral damage
weak ADCC function and complement fixation
what is IgE suited for
helminth infections
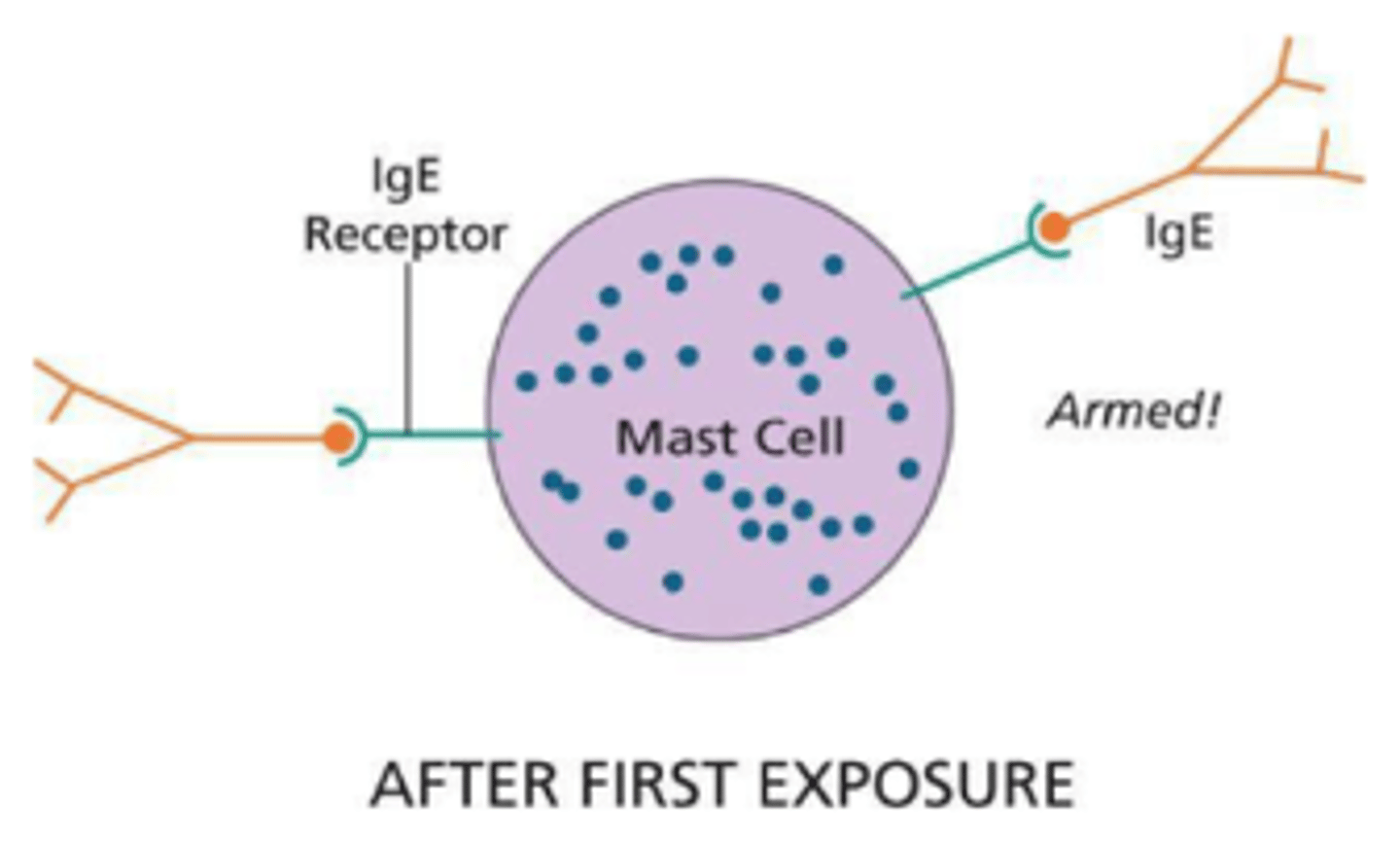
which Ig can make innate cells into memory cells
IgE
how does IgE make innate cells into memory cells
- FceR has high affinity for IgE, and binds monomeric IgE.
- FceR becomes loaded with IgE
- This gives the cell an Ag-specific receptor
which FcR is specific for each Ig

which Igs can opsonise a cell (2)
IgG and IgA (and IgM via the complement)
why can't IgE opsonise
its meant for helminths which are too large to opsonise
how does IgG achieve ADCC
Directs innate cells to kill via interaction with FcR
how does IgE achieve ADCC
kills extracellular pathogens by firing anti-microbial molecules at them
what is the advantage of Ab having two Ag binding sites
Enhanced cross-linking: two binding sites allows a single Ab to bind two distinct epitopes (repeating or adjacent Ags) which is more easily recognised by immune cells
how does AgAb cross linking aid antibody mediated neutralisation (2)
clumps together toxins/pathogens, allowing clearance by immune complexes, and hindering pathogen mobility
why does IgE cause allergies and severe anaphylaxis
When exposed to an allergen Ag, the immune system produces IgE that binds to mast cells.
Upon re-exposure, the Ag cross-links these IgEs, causing mast cells to rapidly release inflammatory mediators (eg histamine), which produce allergic symptoms
what is affinity maturation in Ab
mutations to the V, D, J segments of the Ag binding site changes the affinity of the Ab for its Ag, or which Ag it recognises
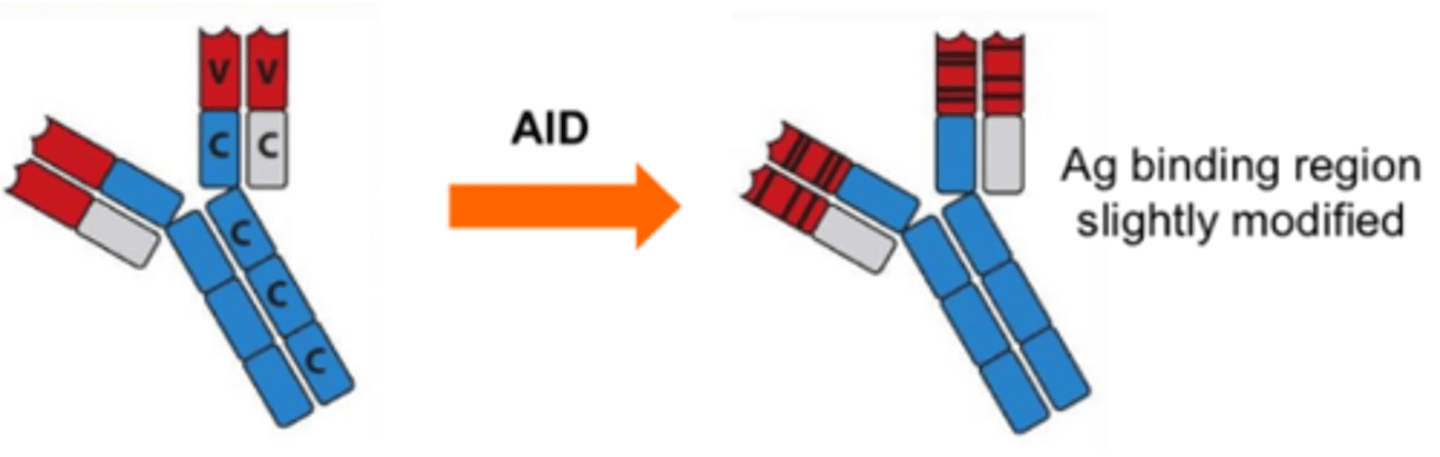
is affinity maturation rare or common
common - 100,000 fold higher rate than normal human cells
what happens if the affinity maturation causes loss of affinity for Ag
the B cell dies
what happens if the affinity maturation causes reduced affinity for Ag
B cell survives but is outcompeted
what happens if the affinity maturation causes increased affinity for Ag
B cell outcompetes others
what is the process of affinity maturation called
somatic hypermutation
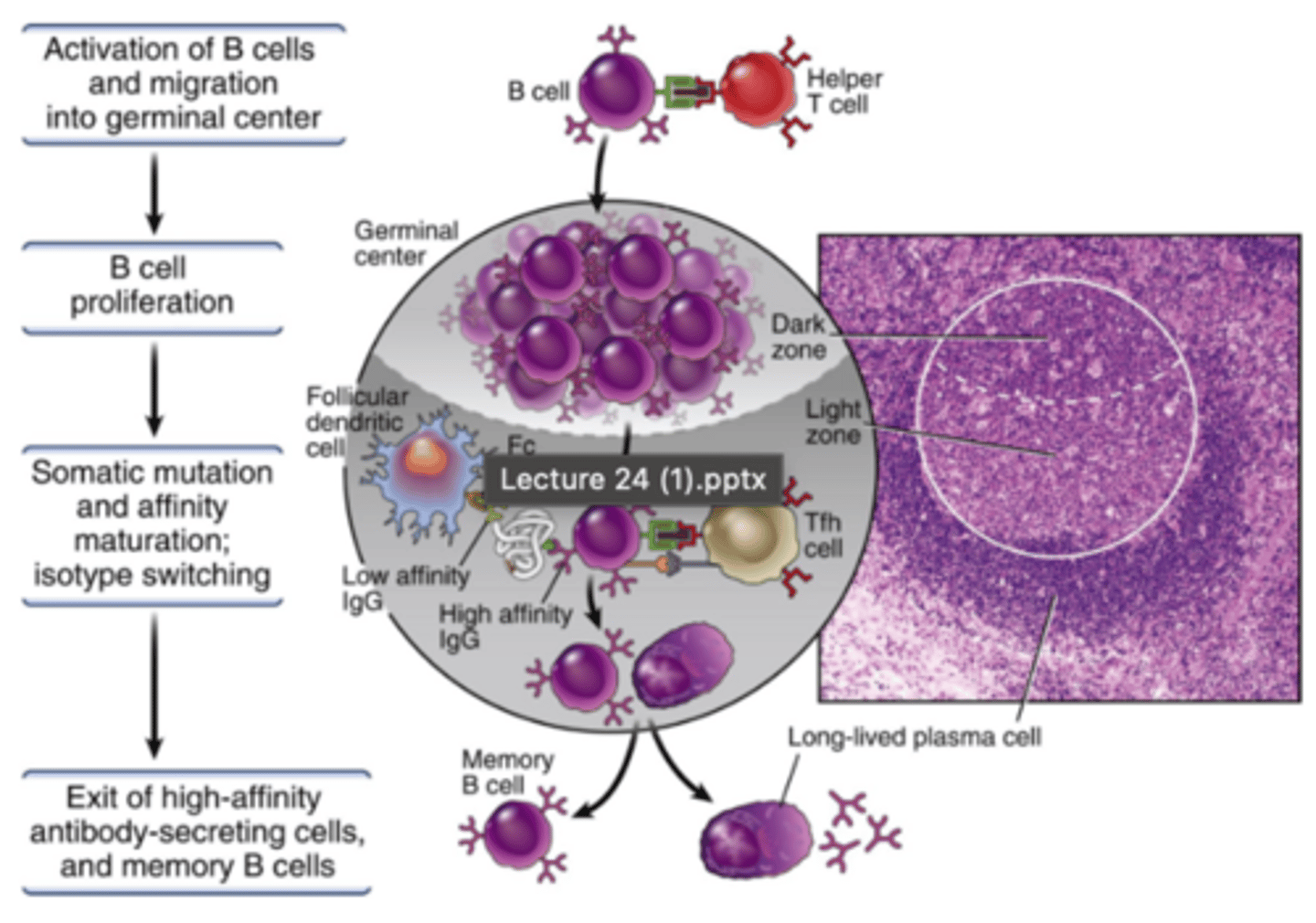
where does affinity maturation take place (2)
germinal centres of the lymph nodes and spleen
what do B cells require to undergo and survive affinity maturation
permission from T cells
what negative thing can occur from affinity maturation
B cells specific for self-Ag (autoimmune disease)
what usually prevents B cells producing Abs specific for self-Ags
B cell must present the Ag it recognises to a T cell.
- If the T cell recognises the Ag: B cell is still microbe specific and can survive
- If the T cell no longer recognises the Ag: B cell has lost specificity or is self-Ag specific so is deleted
do T cells undergo affinity maturation
no
How do clonal expansion, isotype switching and affinity maturation fit together? (picture to see)
what are the three mucosal surfaces of the body
respiratory tract, intestine, urogenital tract
self-reactive T cells can be deleted but what dangerous T cells aren't deleted
T cells that recognise non-self non-dangerous antigens
describe the structure of the intestinal epithelium from surface to mesentery
mucus layer, epthelial cells, lamina proria and peyers patches
what are peyers patches
the areas of lymphoid tissue in the wall of the intestine that are involved in the development of immunity
how do T cells know not to attack food Ags
they will not receive the second signal for danger (B7 upregulation)
what is the result of a T cell being shown a food Ag
it will die or deactivate
what is T cell deactivation called
anergy
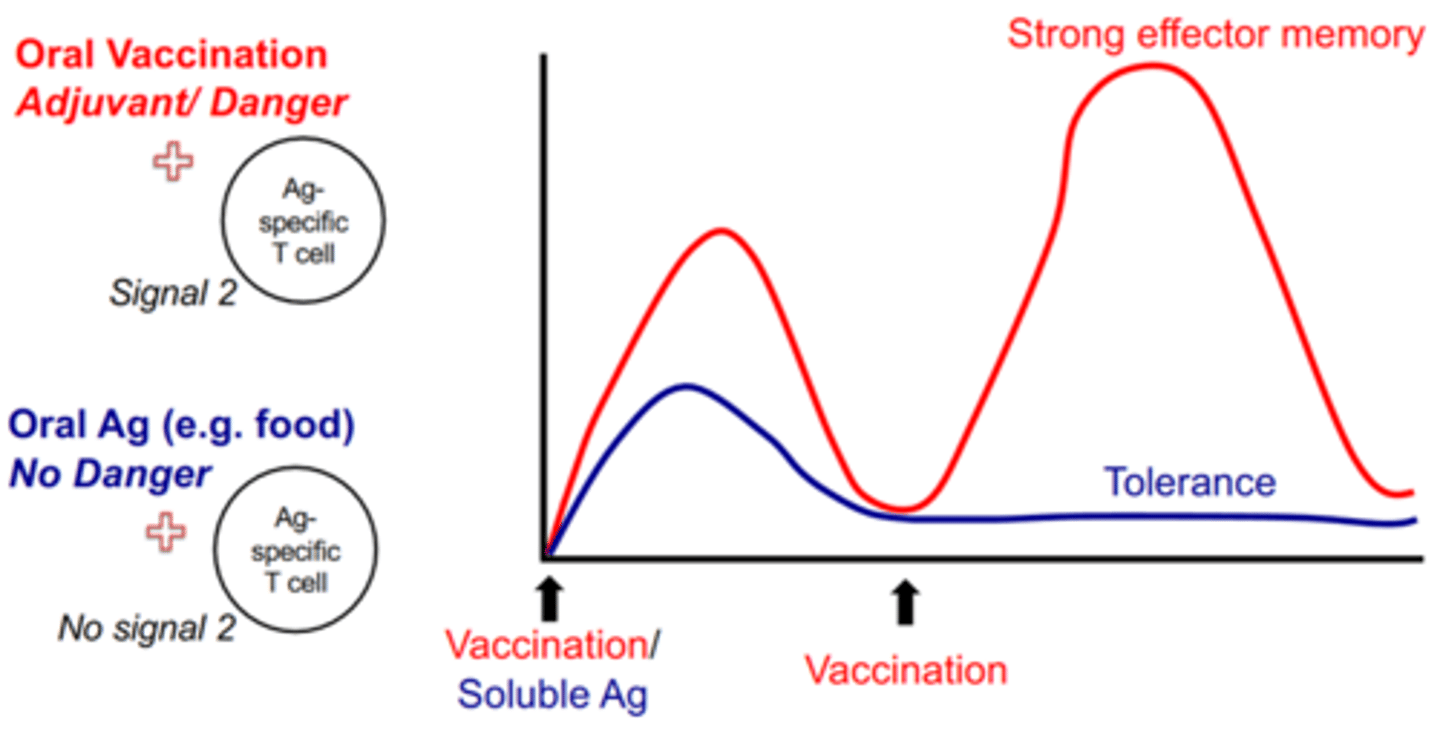
receiving an Ag orally produces what effect. How does this compare to a second vaccination/pathogen occurrence
tolerance on second ingestion. As opposed to a heightened immune response on second infection.
which cytokines does Th1 cells produce
IFN-gamma
which cytokines does Th2 cell producee
IL-4
In coeliac disease, what is activated and what do they produce (3)
Th1 cells produce IFN-y, activated macrophages, B cells
in normal food allergies, what is activated and what do they produce (3)
IL-4, mast cells, igE (from B cells)
where do intestinal commensals live
near the surface of the mucous layer
which type of gut cell produces antimicrobial peptides
paneth cells
which cells transport Ags to the lamina propria
M cells
what is the most prominent antibody in the intestines (reminder)
IgA
what is the special environment found in Peyer's patch
B cells in Peyer's patch have higher rates of somatic hypermutation, making IgA higher affinity for neutralising pathogens
what is the benefit of IgA poor ADCC and complement response
weak responses won't impair the epithelial barrier
comment on the action of these features of mucosal immunity:
1. tight junctions
2. mucus layer
3. anti-microbial peptides
4. IgA
1. tight junctions - prevent epithelium penetration
2. mucus layer - prevents microbes getting to the epithelium
3. anti-microbial peptides - in inner mucus layer, damages microbes that get too close
4. IgA - specialised for neutralising and blocking microbes
what is the result of having no commensals
immune system doesn't develop
how do commensals protect the intestine
they fill up the intestinal niche to prevent pathogenic bacteria surviving
do commensals trigger an immune response?
yes, just not an aggressive response
what causes Appendicitis
blockage in the appendix causing build up of mucus and microbial products and inducing inflammation
what is the result of inflammation in appendicitis
intestinal barrier loses integrity, commensals invade lamina propria, appendix bursts
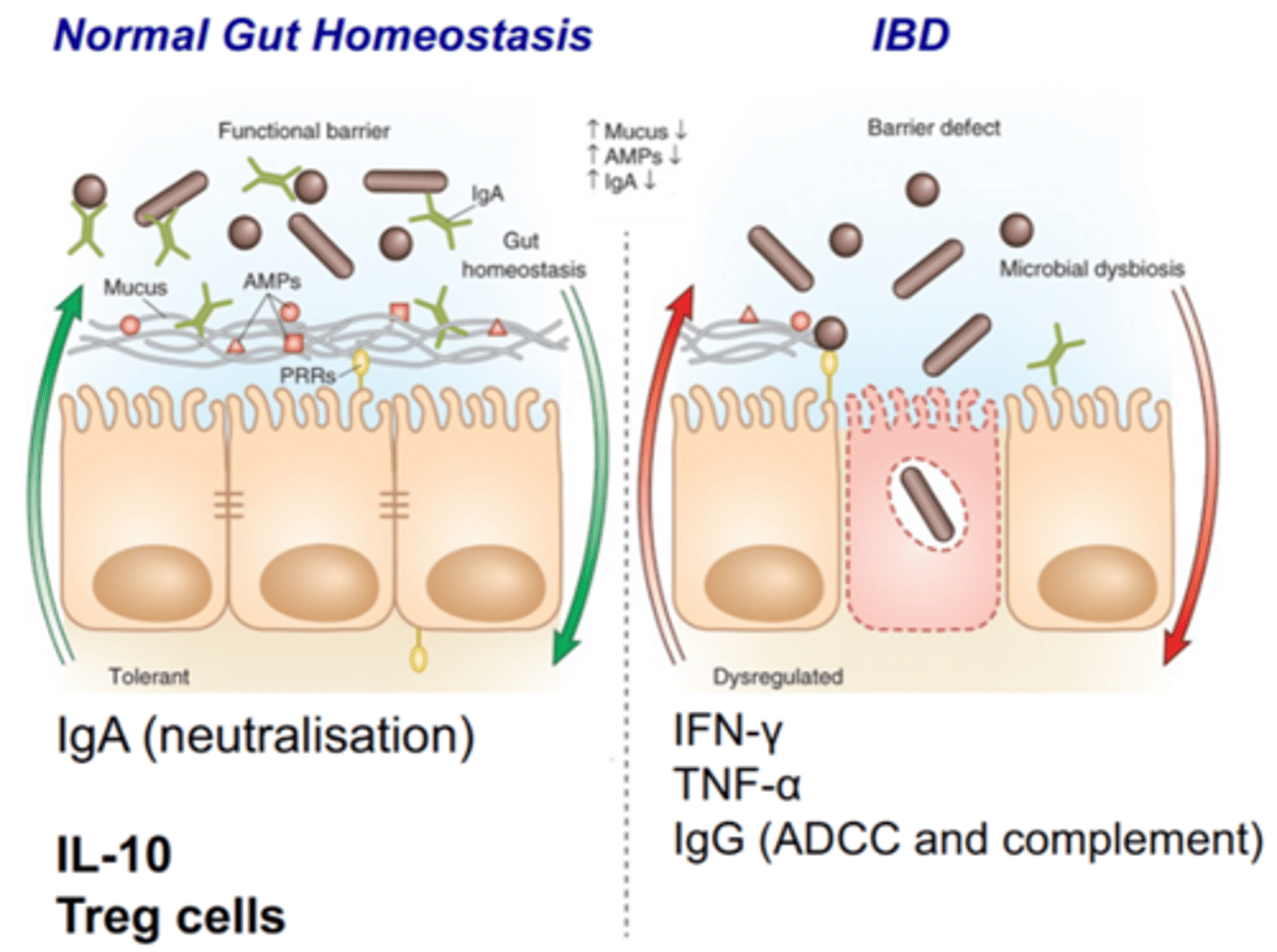
what is inflammatory bowel disease linked to
over-enthusiastic immune responses against commensals
what is the difference between a normal gut commensal response and an IBD commensal response
normal: IgA release, then IL-10 and Treg cells for inhibition
IBD: IFN-y, TNF-a, IgG (ADCC and complement) activation
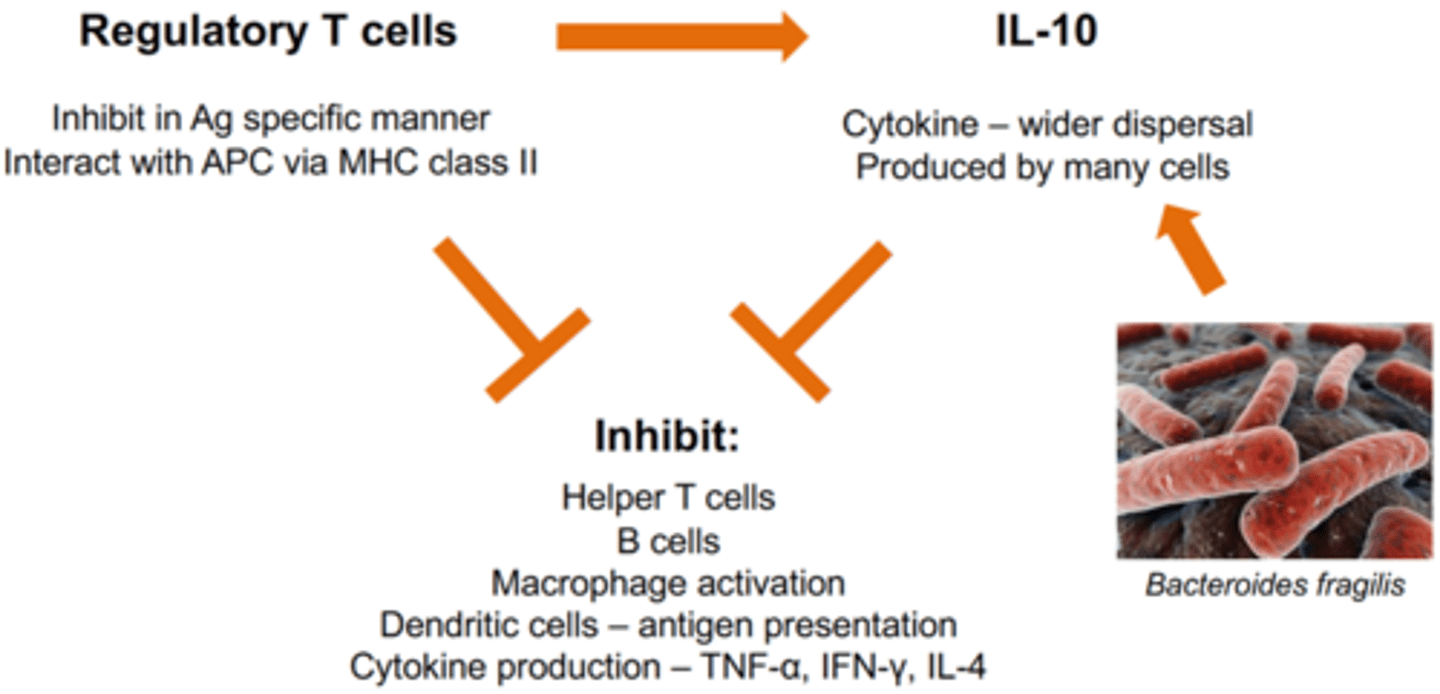
which two immune molecules regulate the immune response
Regulatory T cells and IL-10
what do Treg cells produce
IL-10
what is the inhibitory effect of Treg cells and IL-10 (5 targets)
inhibits:
helper T cells
B cells
macrophage activation
antigen presentation
cytokine production (TNF-a, IFN-y, IL-4)
allergies developed correlate with what childhood factor
interaction with different commensal communities
how long will plasmodium develop in humans
11-14 days
what stage of development does plasmodium enter the human skin
sporozoites
where in the human does plasmodium replication take place
liver
what cells does plasmodium invade
red blood cells
which stage of the plasmodium human cycle causes fever symptoms
erythrocytic cycle
how do the sporozoites reach the liver
carried by blood flow
what type of cell do the sporozoites infect in the liver
hepatocytes
what are hepatocytes
specialised epithelial cells important for protein synthesis and storage, production of bile, synthesis of blood clotting factors
are many hepatocytes infected
no, only about 100, but each cycle in the liver forms up to 90,000 merozoites
what do infected hepatocytes release
type I inferferon
what does type I IFN do in this environment and what effect does it have (2)
signals to neighbouring hepatocytes to reduce chance of infection and increase antigen presentation
reminder: what are the two mechanisms of CTL killing
FAS/FASL
perforin and granzyme B
what happens to the liver during infection and why
hepatomegaly - enlargement of the liver
due to infection and inflammation
what other organ is enlarged during malaria infection
spleen (splenomegaly)
what is the spleens role during malaria infection (3)
filtration of parasitised red blood cells
immune cell activation
haematopoiesis
what is severe malarial anaemia defined by and what causes it
in children mainly:
driven by both increased destruction of red blood cells (RBCs) and reduced production of new RBCs
defined by hemoglobin (Hb) concentrations <5 g/dL
what are the mechanisms that lead to reduced RBCs (4)
1. lysis of parasited RBCs
2. removal of parasited RBCs in the spleen
3. decreased/suppressed erythropoiesis
4. removal of uninfected RBCs
what is the process of RBC lysis
The parasitophorous vacuole membrane ruptures, freeing the merozoites in the RBC
The RBC will swell and the cytoplasm degrades. The lysis that occurs is explosive, scattering the merozoites.
what is also released by the RBC along with the parasite (4)
PAMPs and DAMPs, proteases, haemoglobin and haemozoin
what is haemozoin (Hz)
a product of haemoglobin metabolism by plasmodia.
a crystalline with a brown pigment
what is the purpose of Hz
free haem released by Hb digestion is toxic, so these crystalline byproducts are formed to help the parasites survive
how does haemozoin increase symptoms
it is immunostimulatory (produces lots of cytokines) - can cause chronic inflammation
what is the effect of removing phagocytes on malaria infection
decrease in anaemia, increase in parasitaemia