Proteins and Enzymes Overview
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Proteins' Elements
carbon, hydrogen, oxygen, nitrogen, and sometimes sulfur
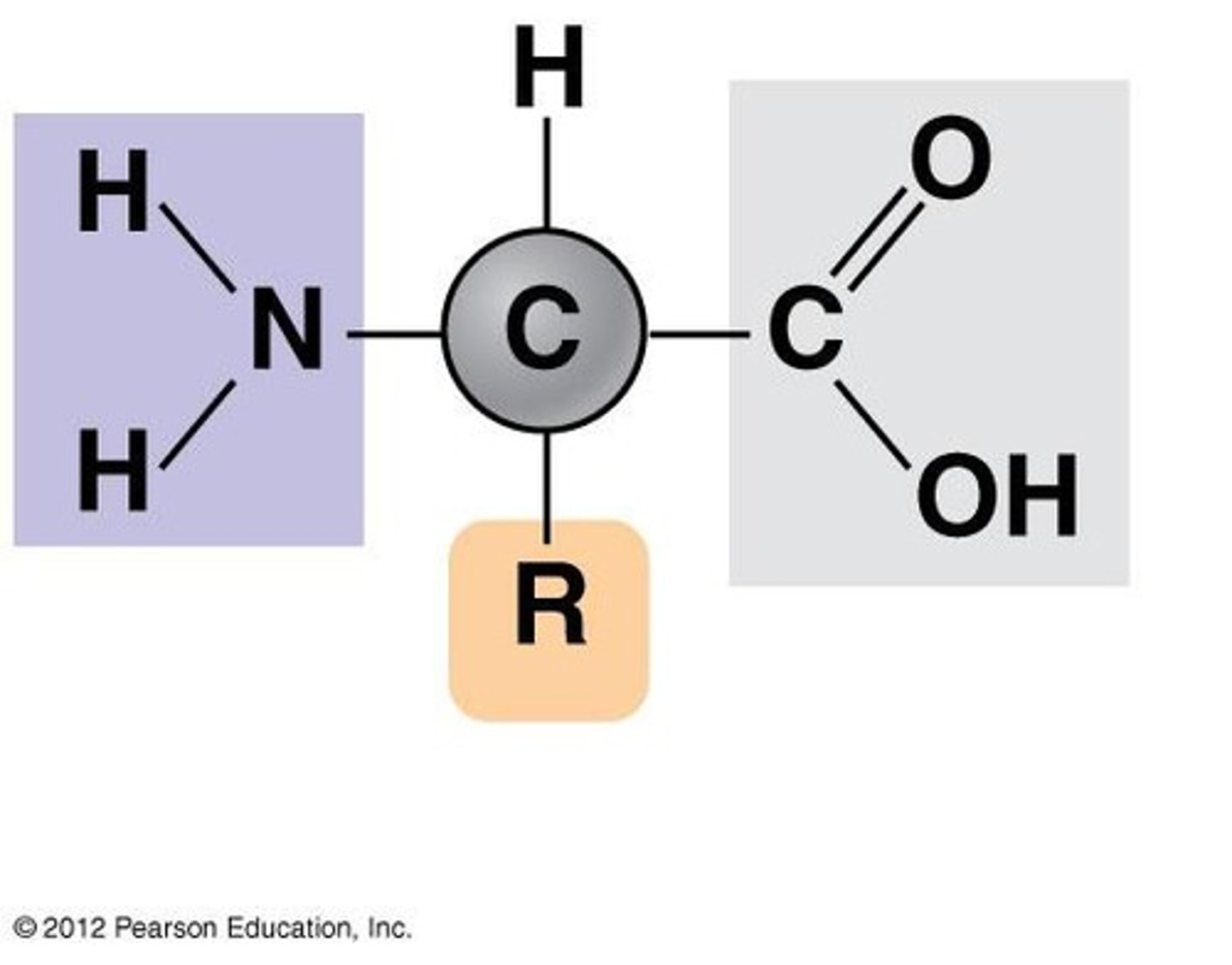
Protein Monomers
Amino acids
Protein Polymers
Polypeptides
Number of Amino Acids
20 different amino acids
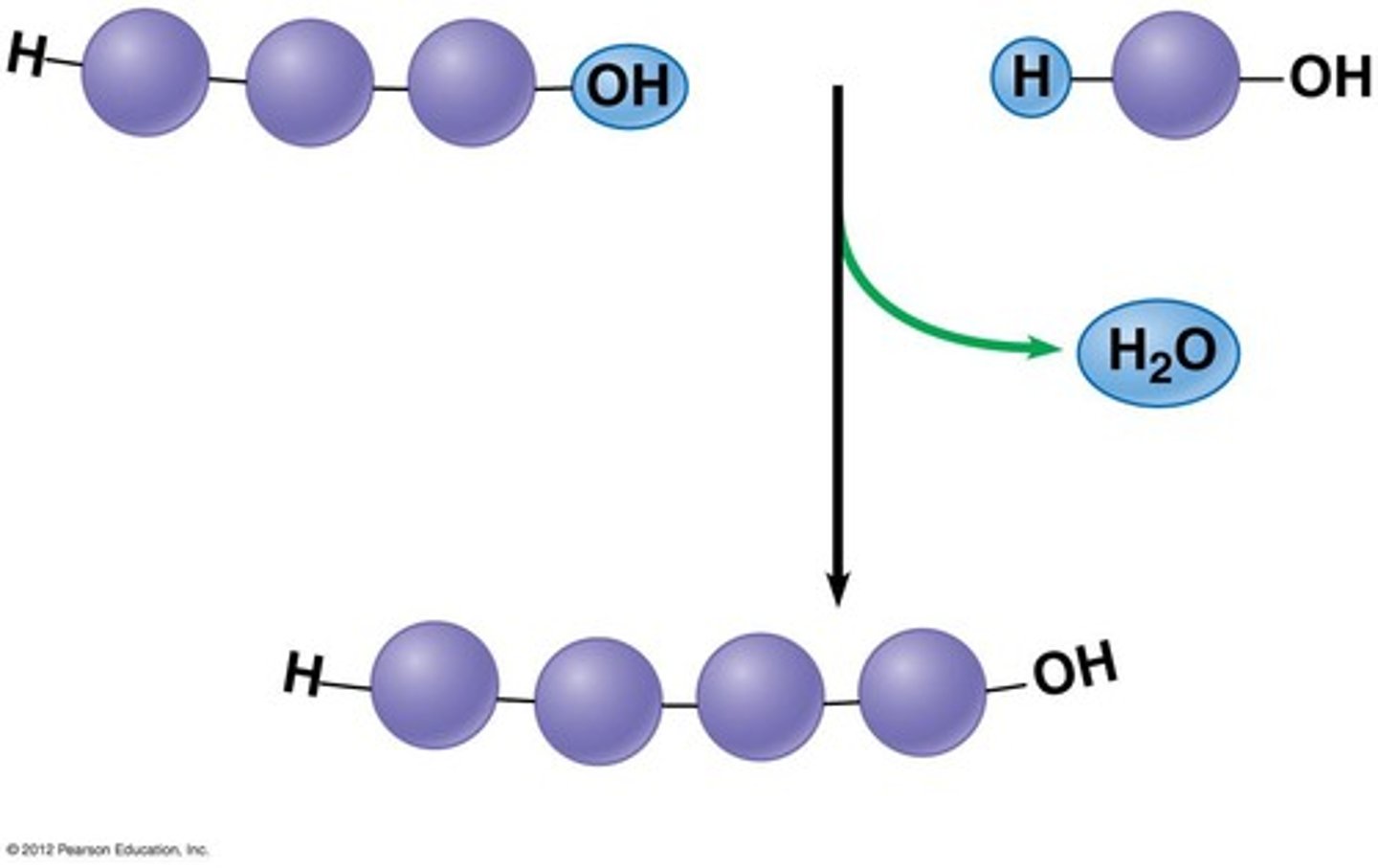
Dehydration Synthesis
A chemical reaction where water is removed to bond molecules
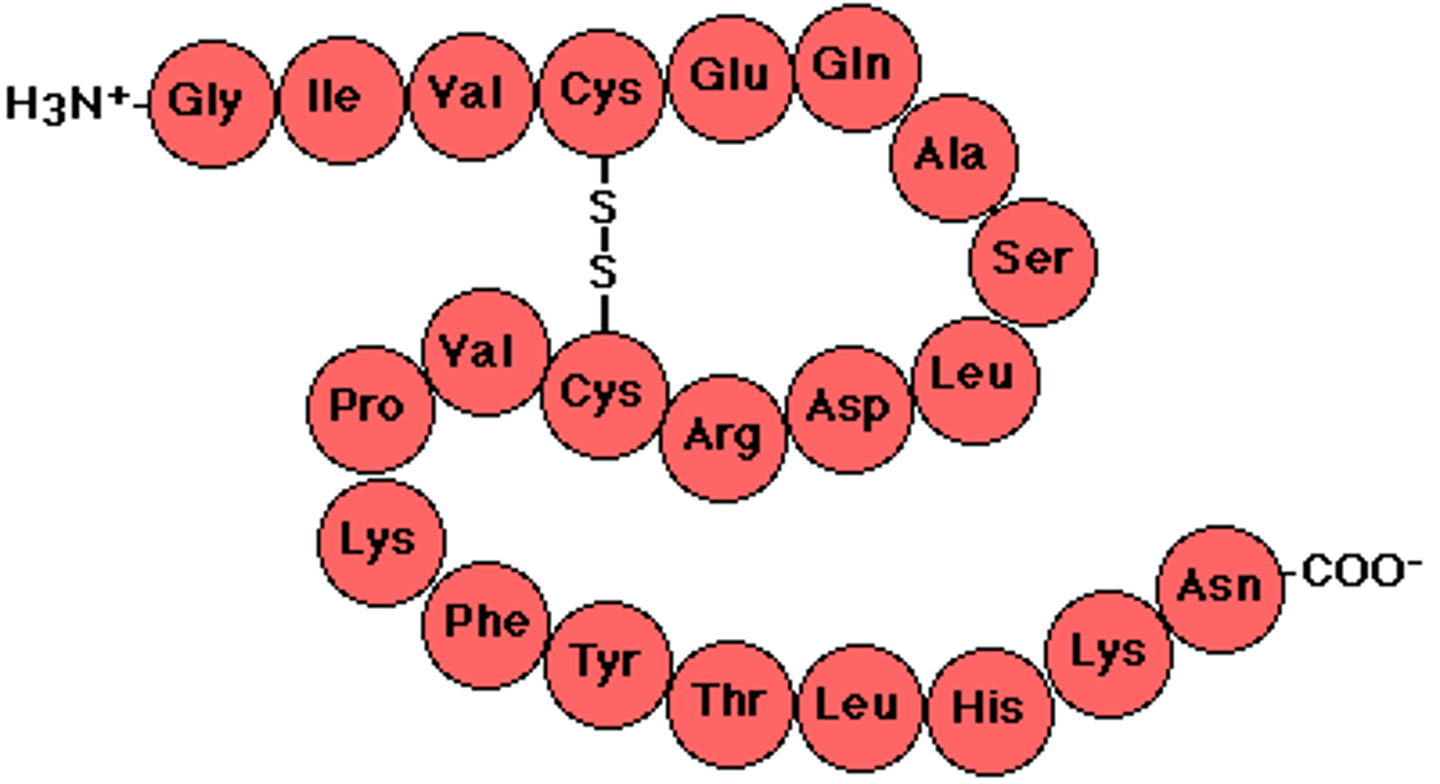
Primary Structure
Linear sequence of amino acids in a protein

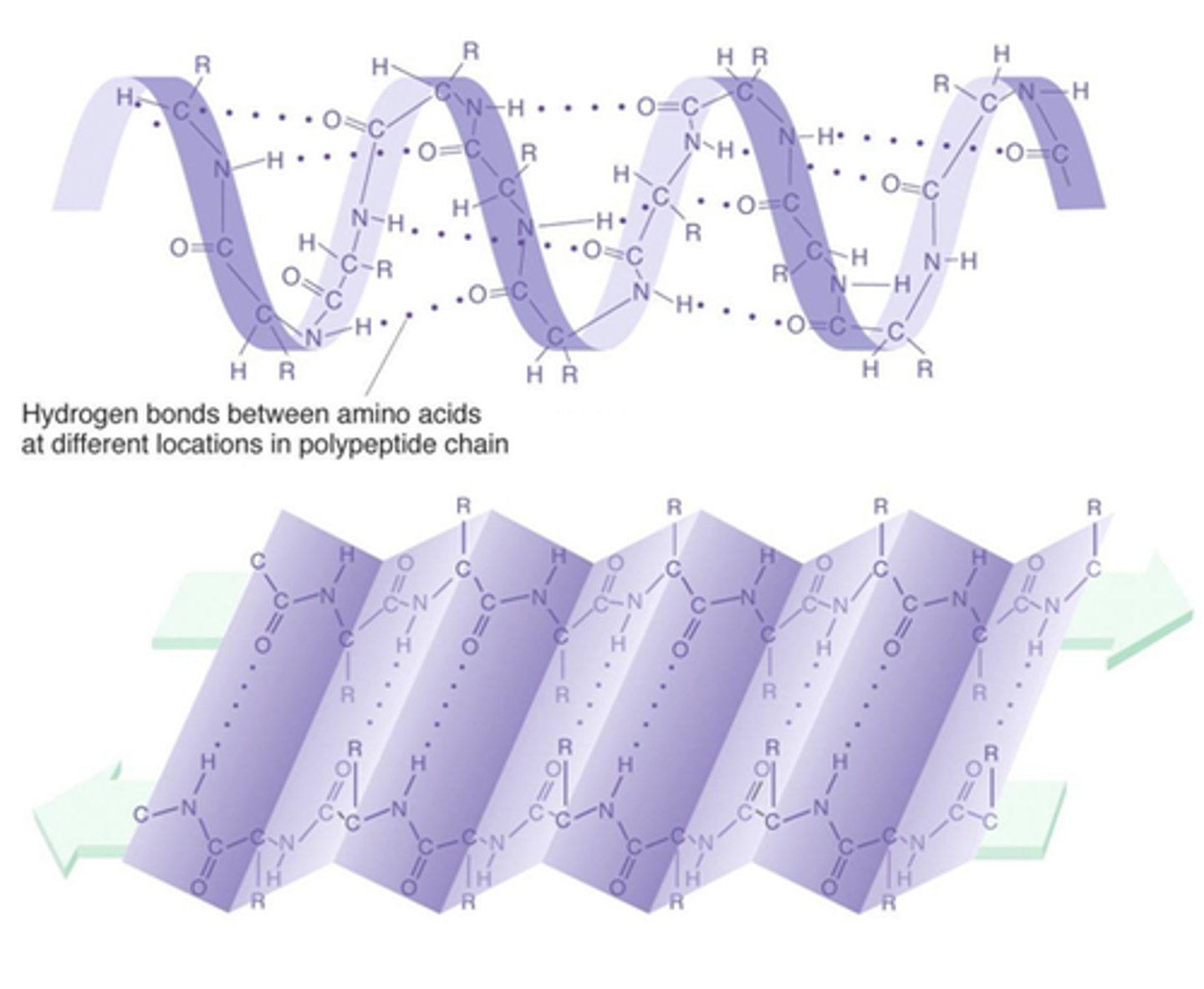
Secondary Structure
Folding of the primary structure due to hydrogen bonds forming between the chain of amino acids; Either an alpha helix or beta pleated sheet.
Tertiary Structure
Folding of the secondary structure by interaction of the R groups; 3D structure of folded protein
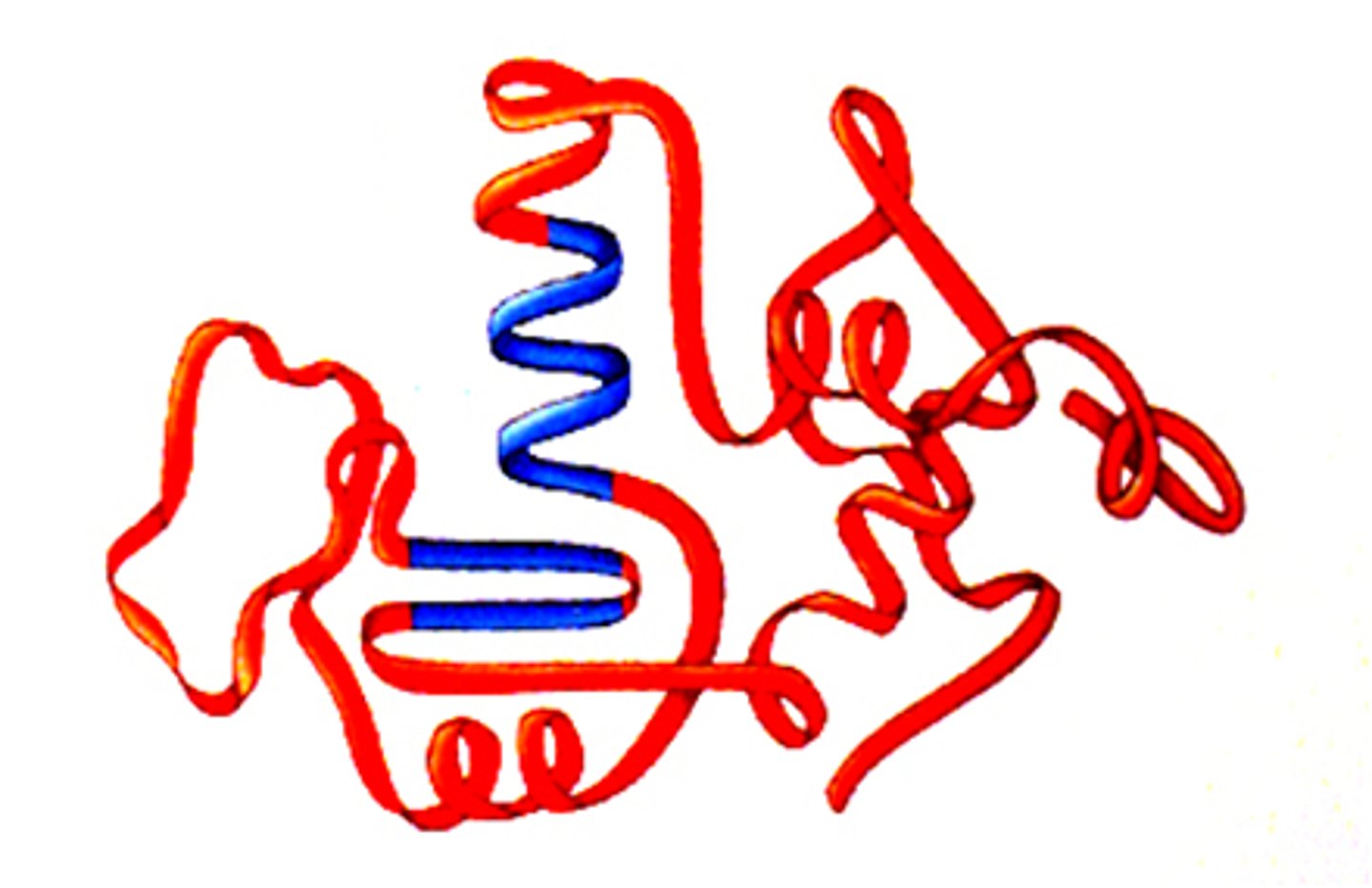
Quaternary Structure
Two or more proteins (tertiary structures) joined together
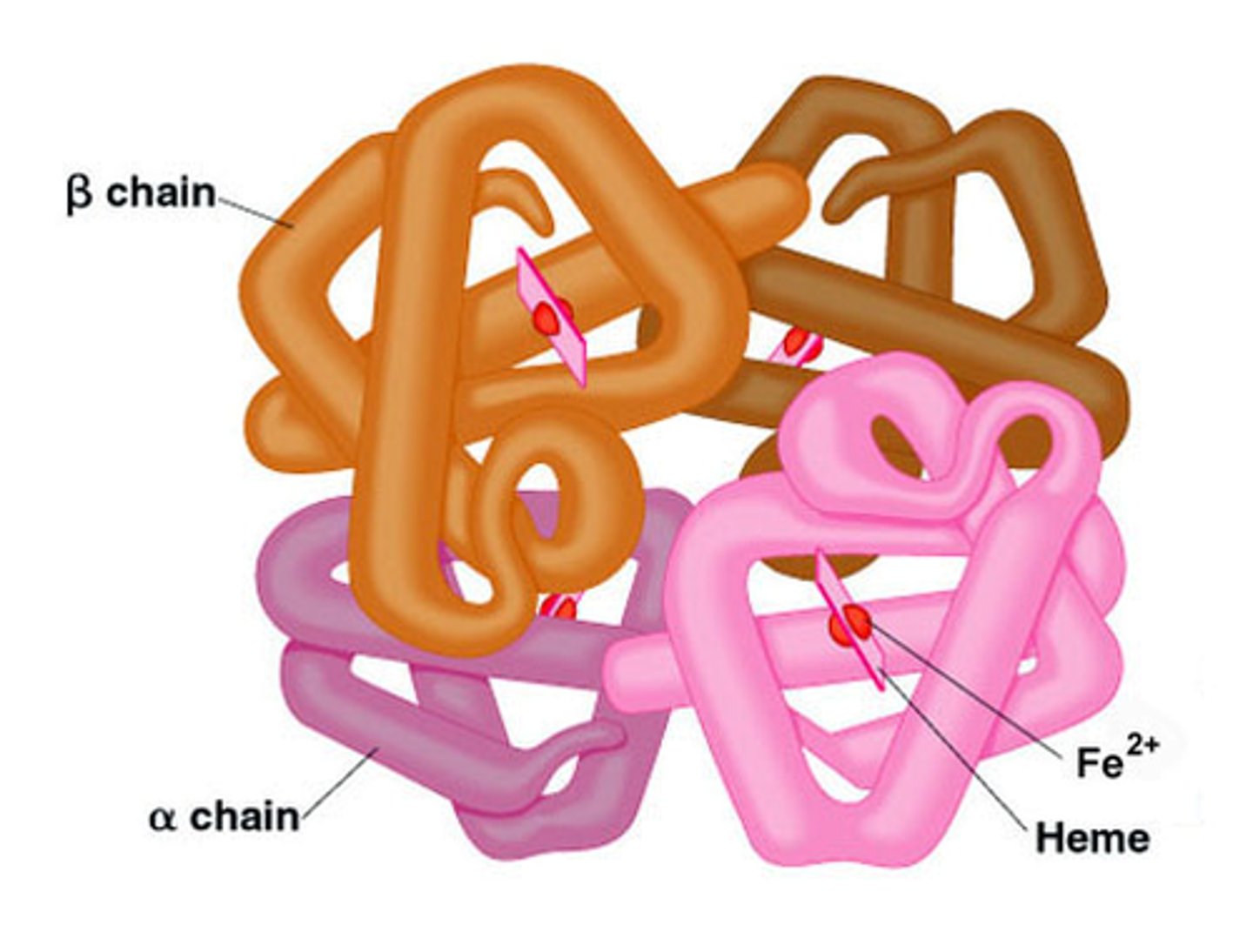
Fibrous Proteins
Polypeptide chains arranged as strands or sheets; providing structural support

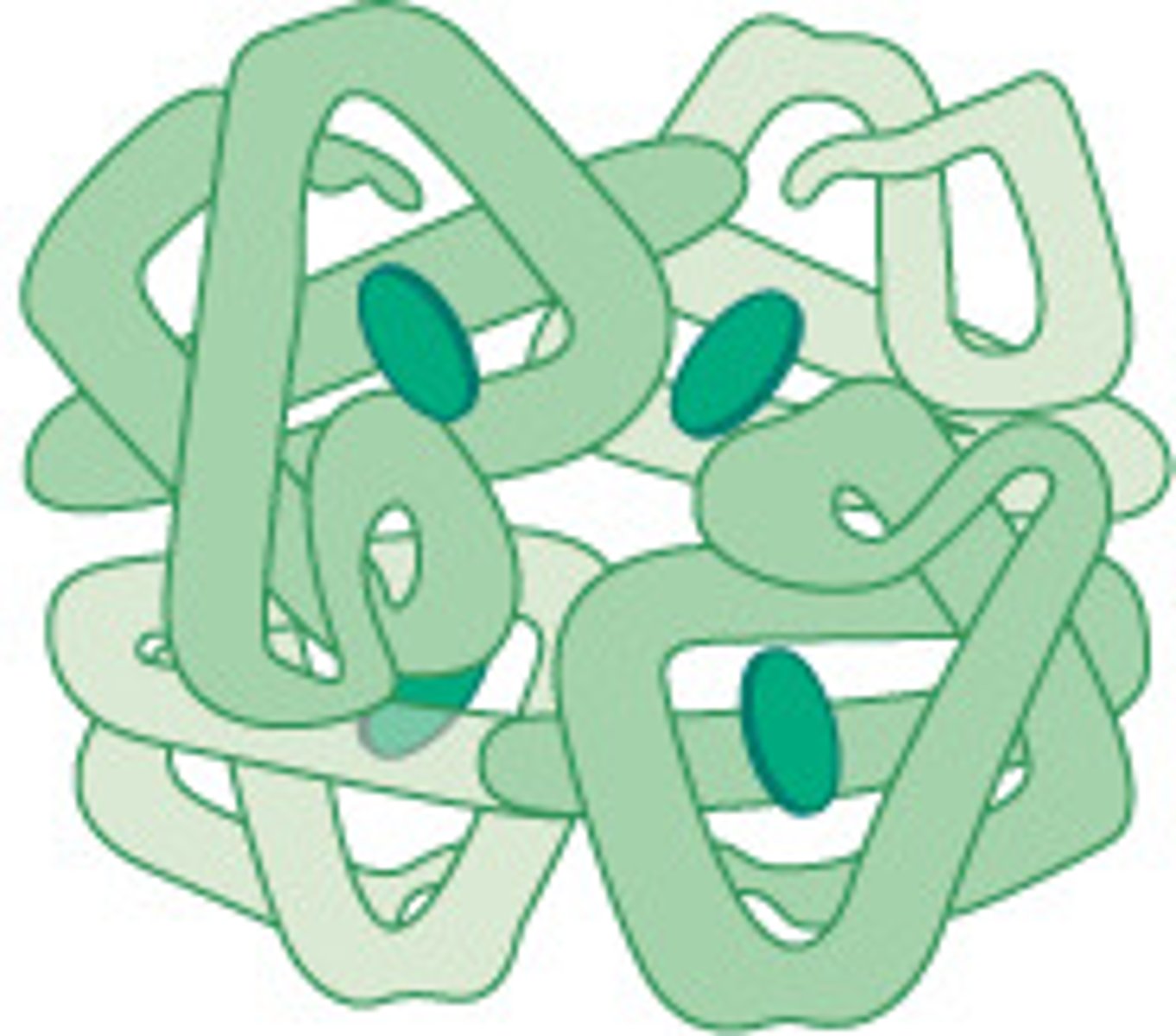
Globular Proteins
Polypeptide chains folded into compact, rounded shapes
Sickle Cell Anemia
Result of misfolding of hemoglobin protein

Factors Affecting Protein Structure
Changes in DNA sequence, pH, temperature, or salt concentration
Activation Energy
Energy required to start a chemical reaction
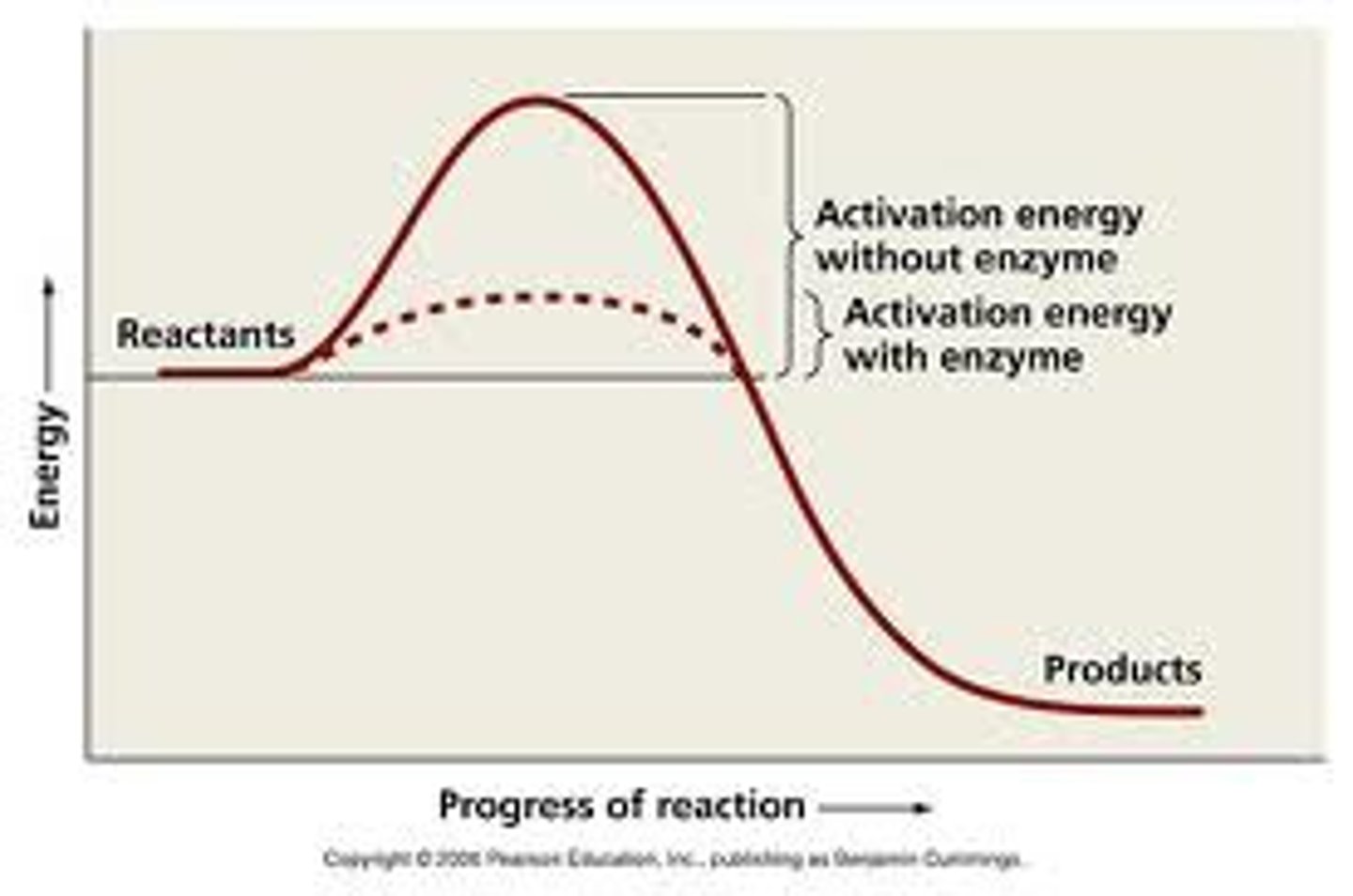
Enzymes
Biological catalysts that speed up reactions without being consumed
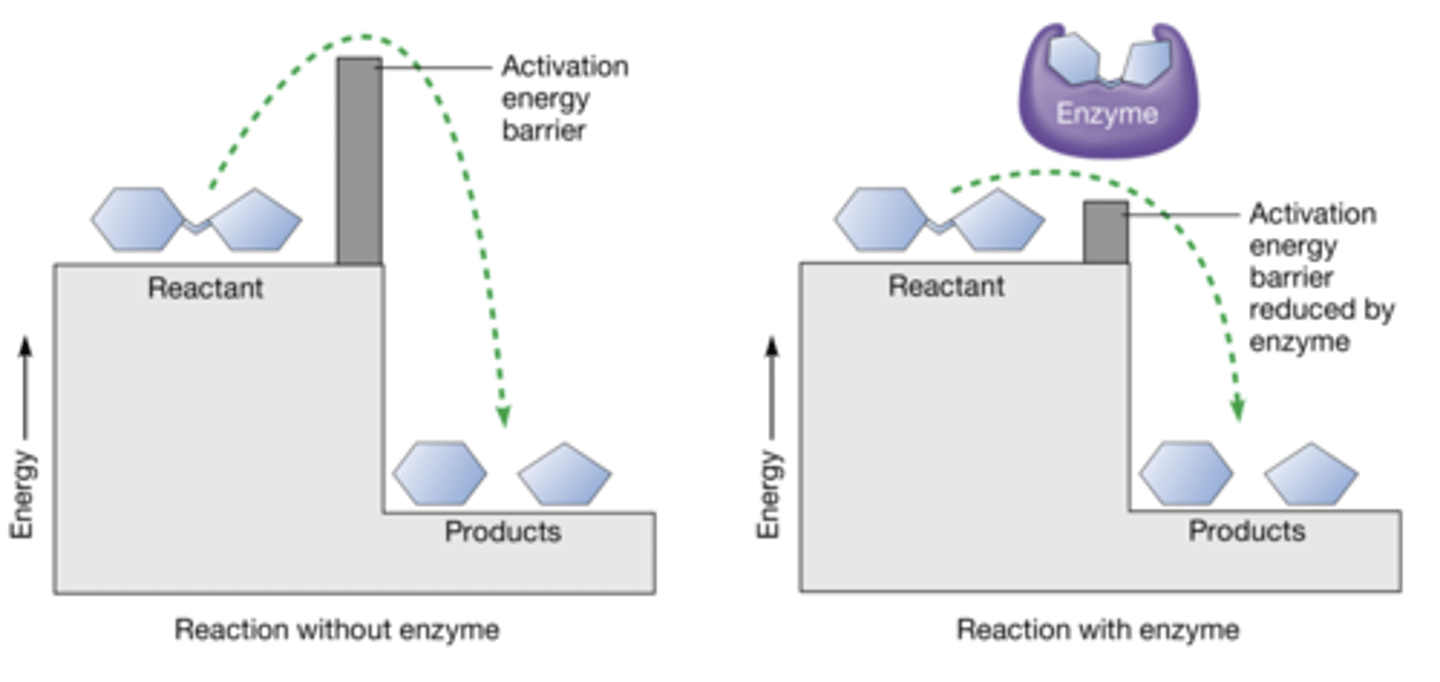
Enzyme Activation Barrier
Energy barrier enzymes help overcome for reactions to proceed
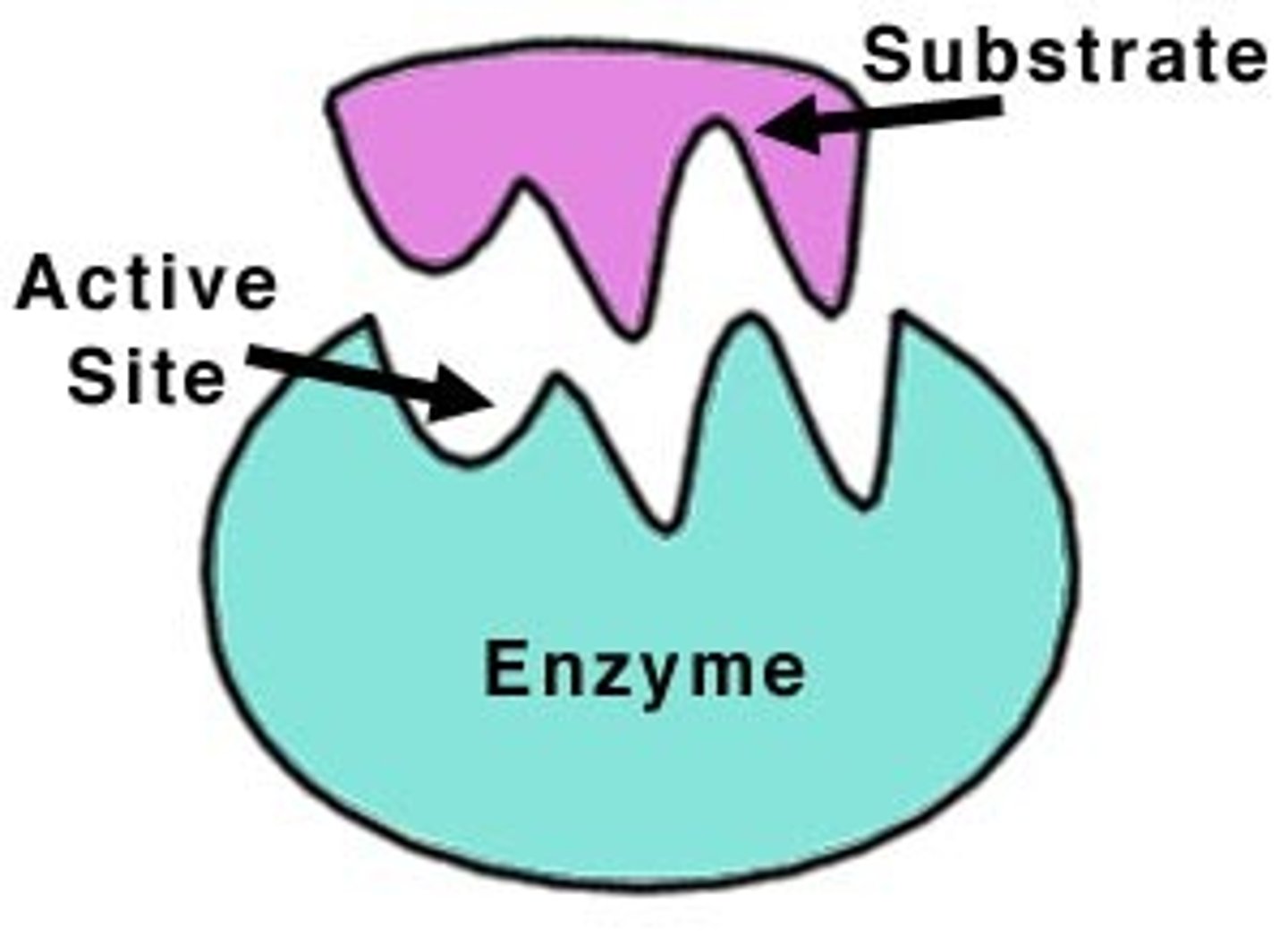
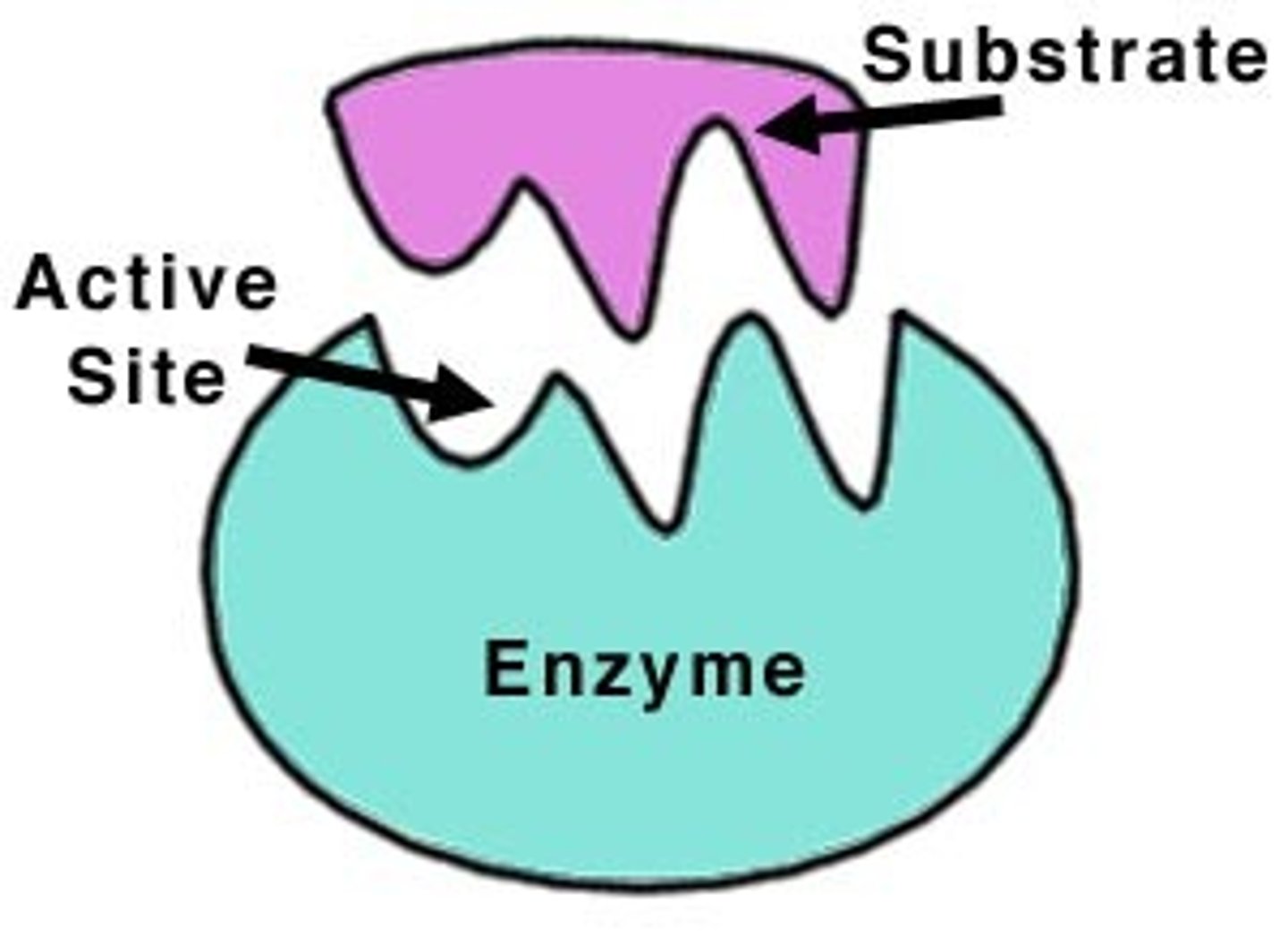
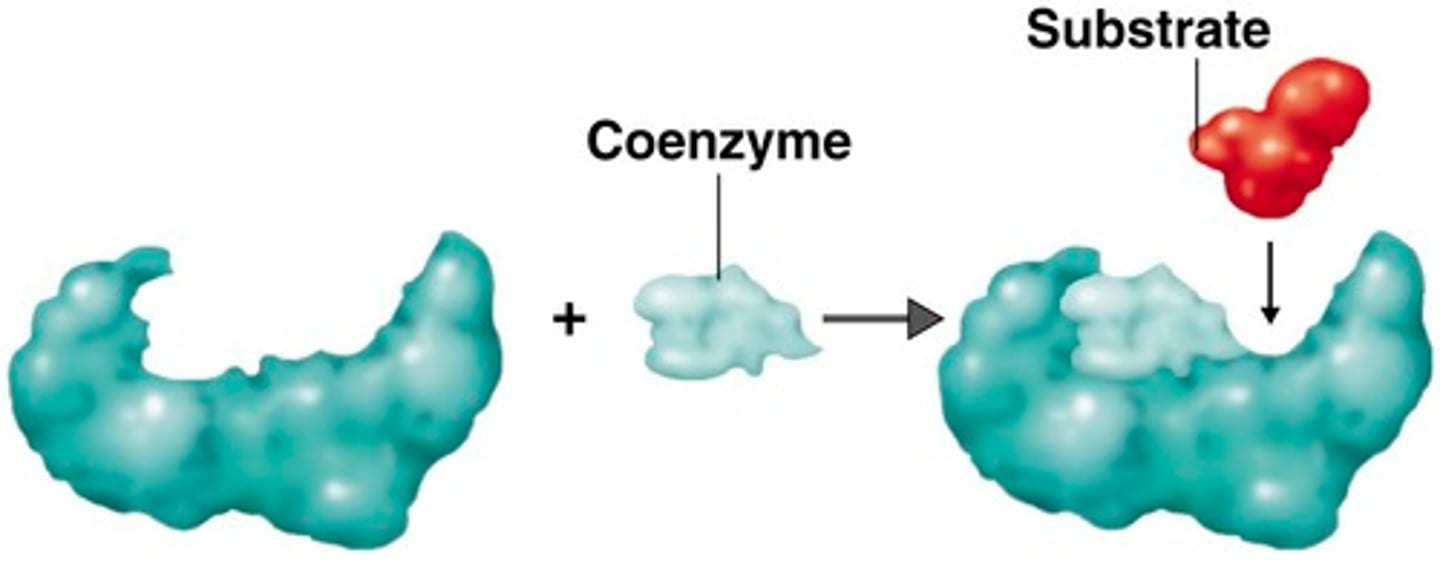
Enzyme Active Site
Region where substrate binds and reactions occur
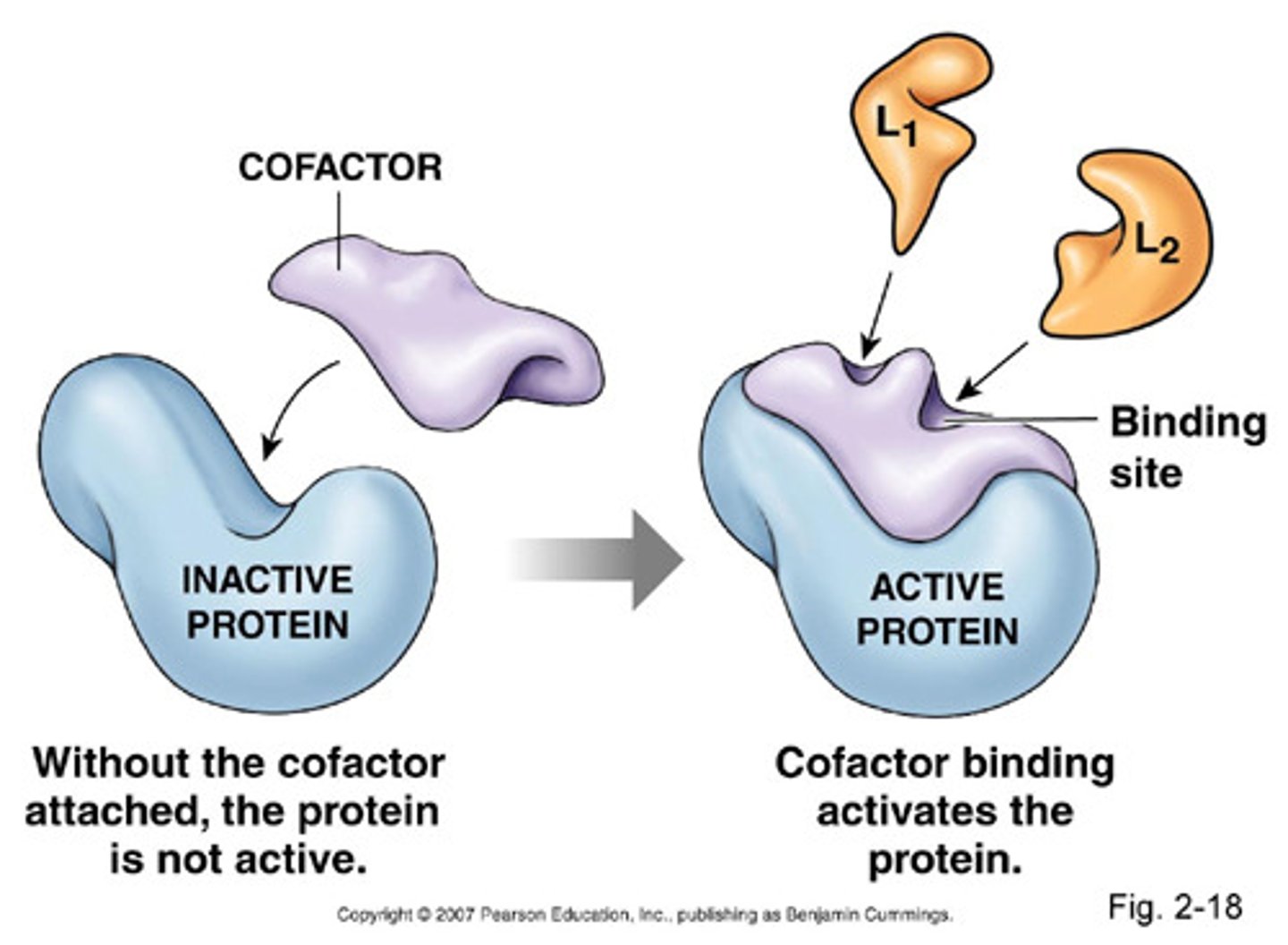
Cofactors
Inorganic helpers like iron, zinc, and copper for enzyme function
Coenzymes
Organic molecules aiding enzyme function
Inhibitor
Chemicals hindering enzyme activity

Competitive Inhibitors
Block substrates from binding to the enzyme's active site

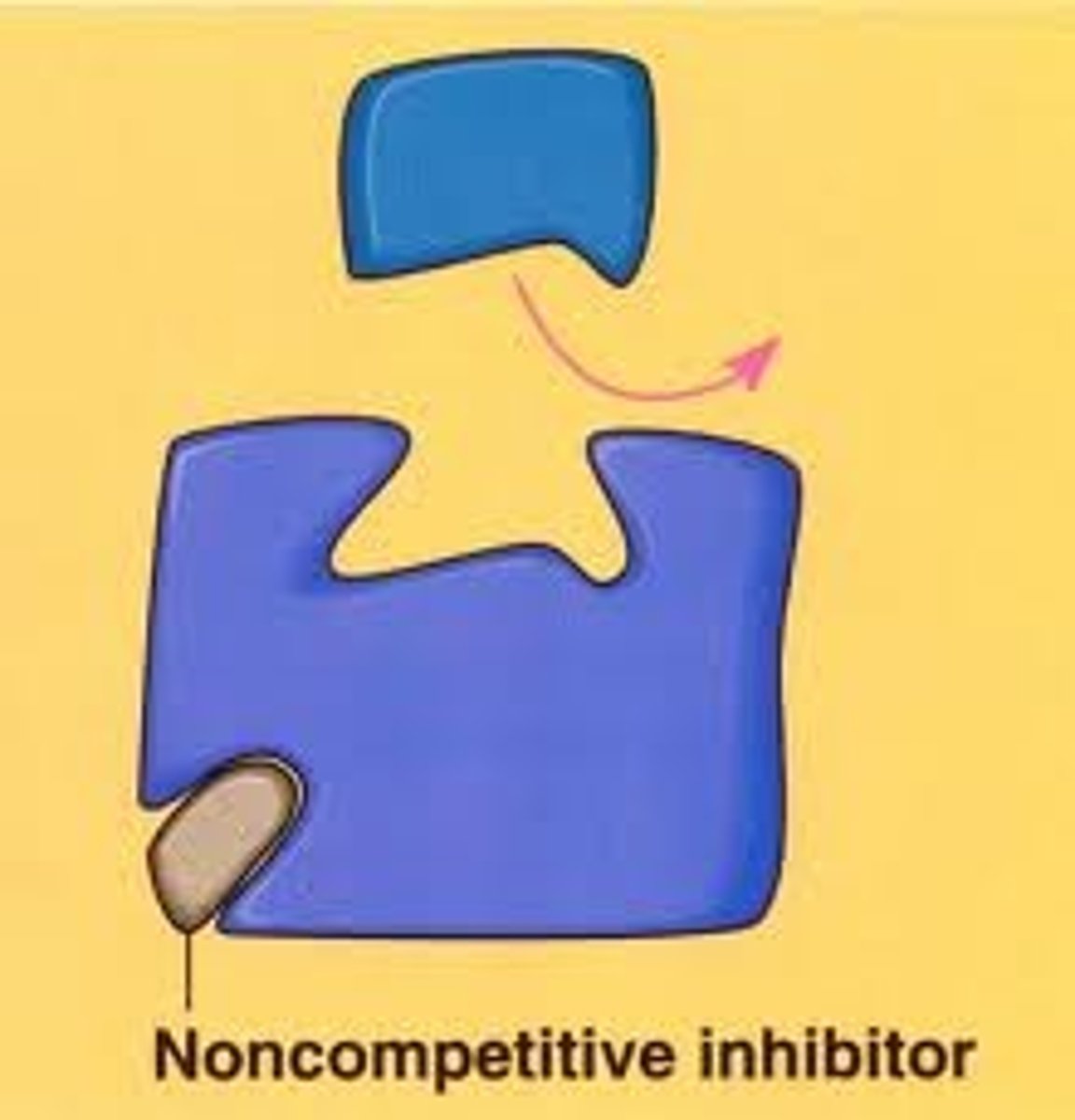
Non-Competitive Inhibitors
Bind elsewhere on the enzyme, altering its shape
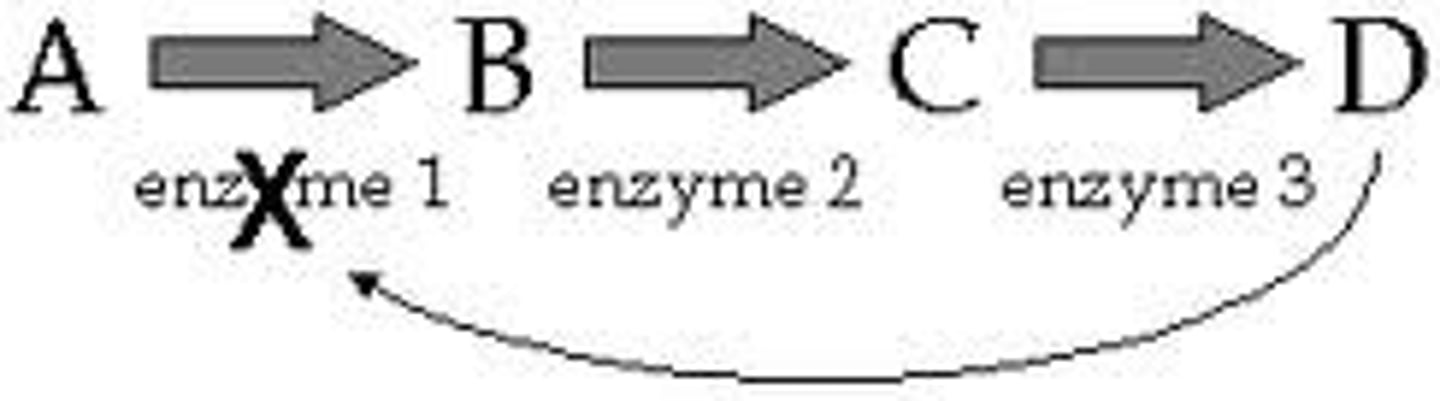
Feedback Inhibition
Product of a pathway inhibits an enzyme to regulate the pathway
Enzyme Inhibitor Example
Aspirin inhibits prostaglandins to reduce pain and inflammation