antiviral agents (non-retroviral) - LAST LECTURE EVER
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Viruses- Definition and Classification
Utilize host-cell materials and some host machinery for replication
Effective antivirals inhibit events specific to the virus life cycle
Because the enzymes/ processes each virus uses are often unique to that virus, very few antivirals offer broad coverage (cf antibiotics)
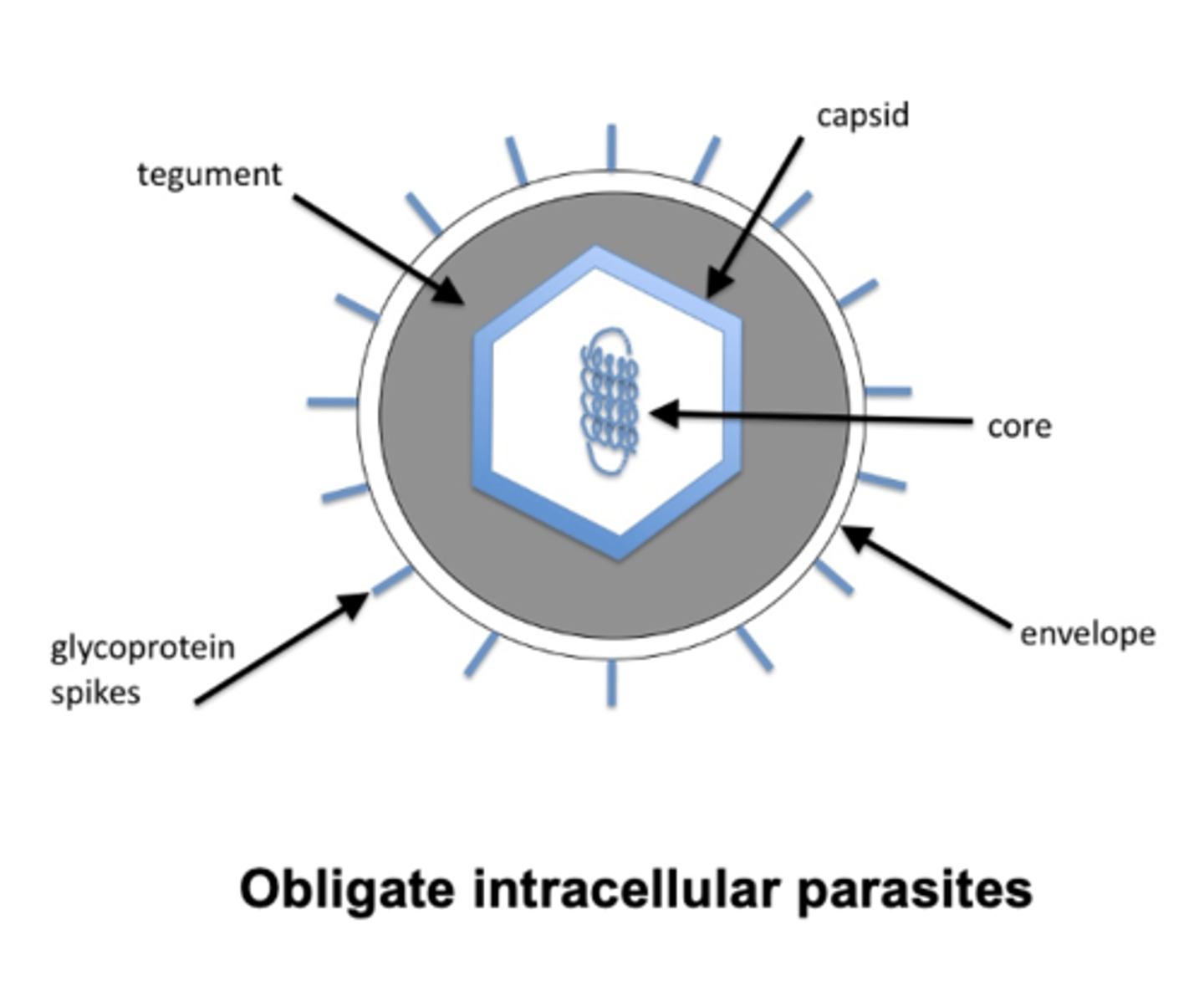
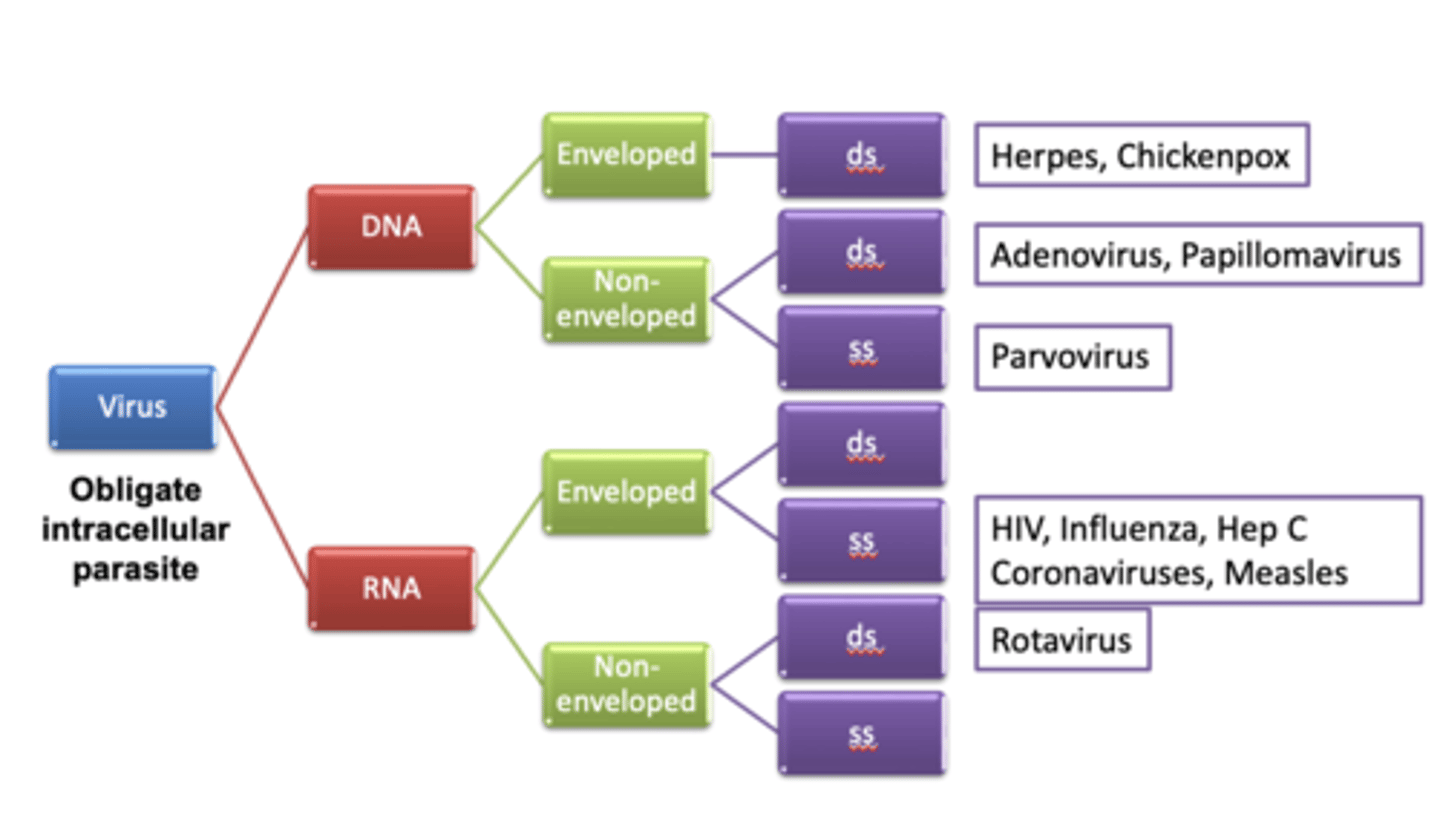
Definition and Classification (2)
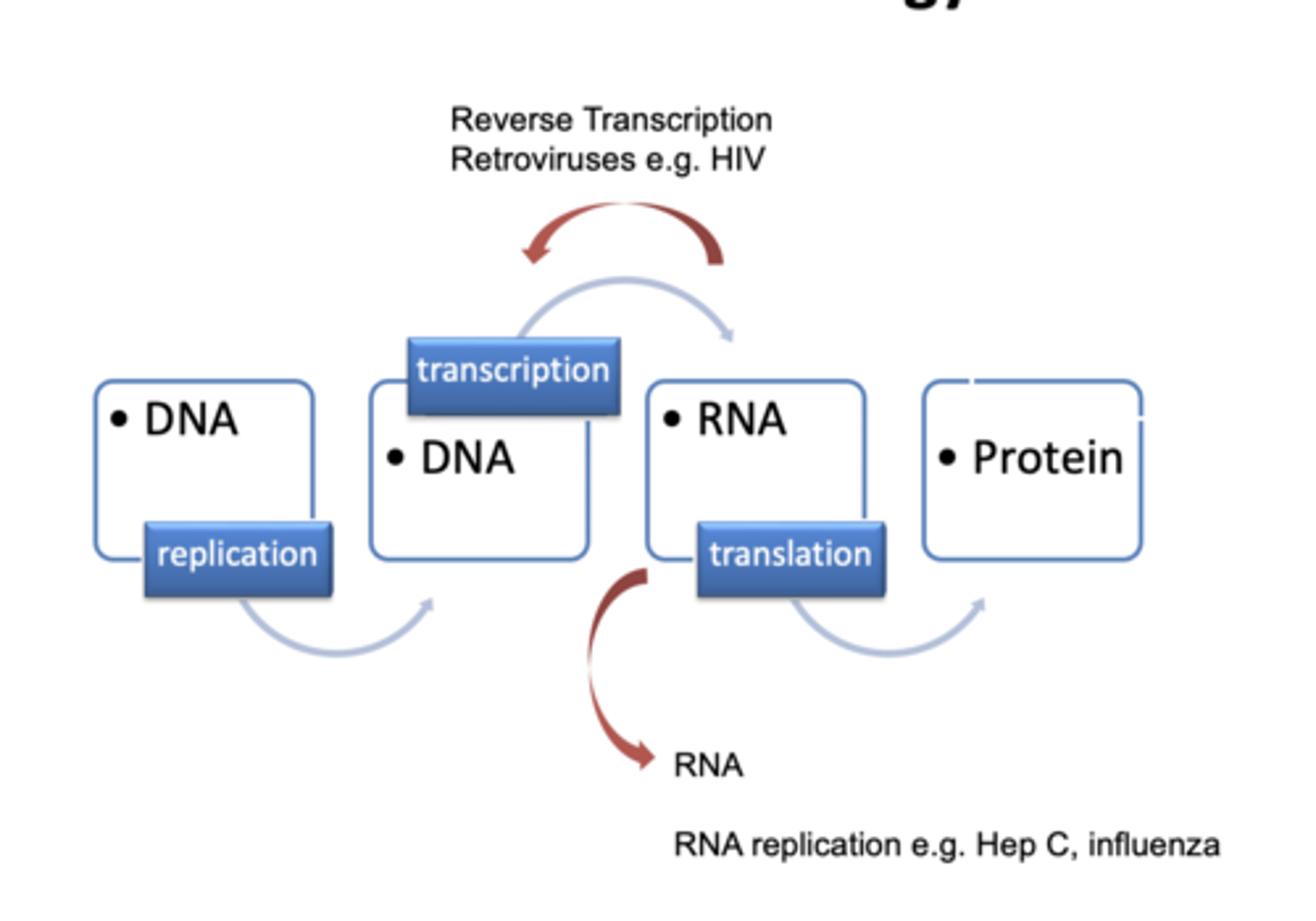
Review of Terminology
DNA Viruses and disease
Poxvirus
-smallpox
Herpesvirus
-Chikenpox/shingles, cytomegalovirus
Adenovirus
-Conjunctivitis, sore throat
Hepadnavirus
-Hepatitis B
Papillomavirus
-warts
Herpesviruses
HSV, VZV, CMV, HHV-6, EBV, KSHV
Establish lifelong infections in the host
Three stage life cycle:
-Primary infection
-Latent infection
-Reactivation
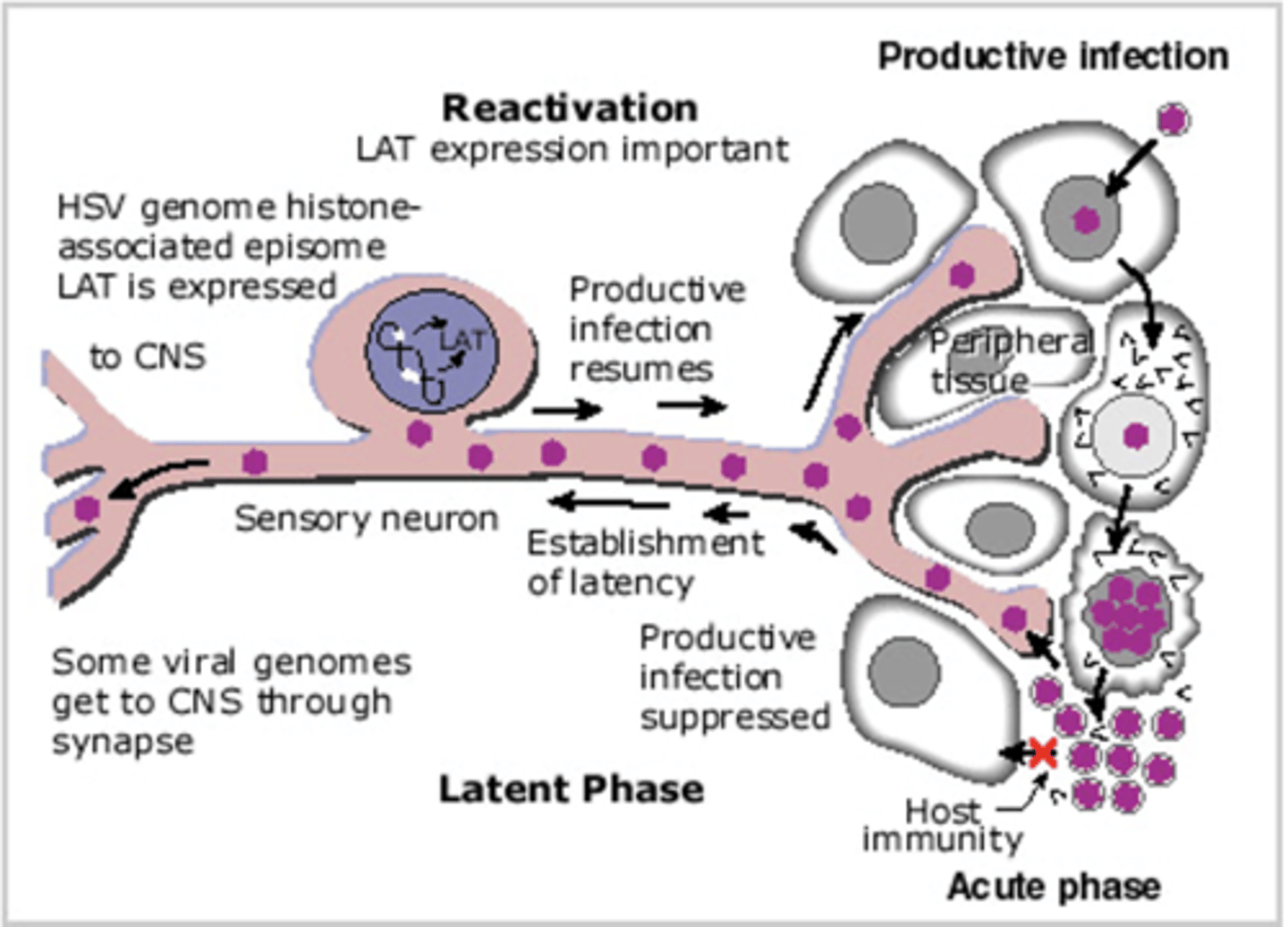
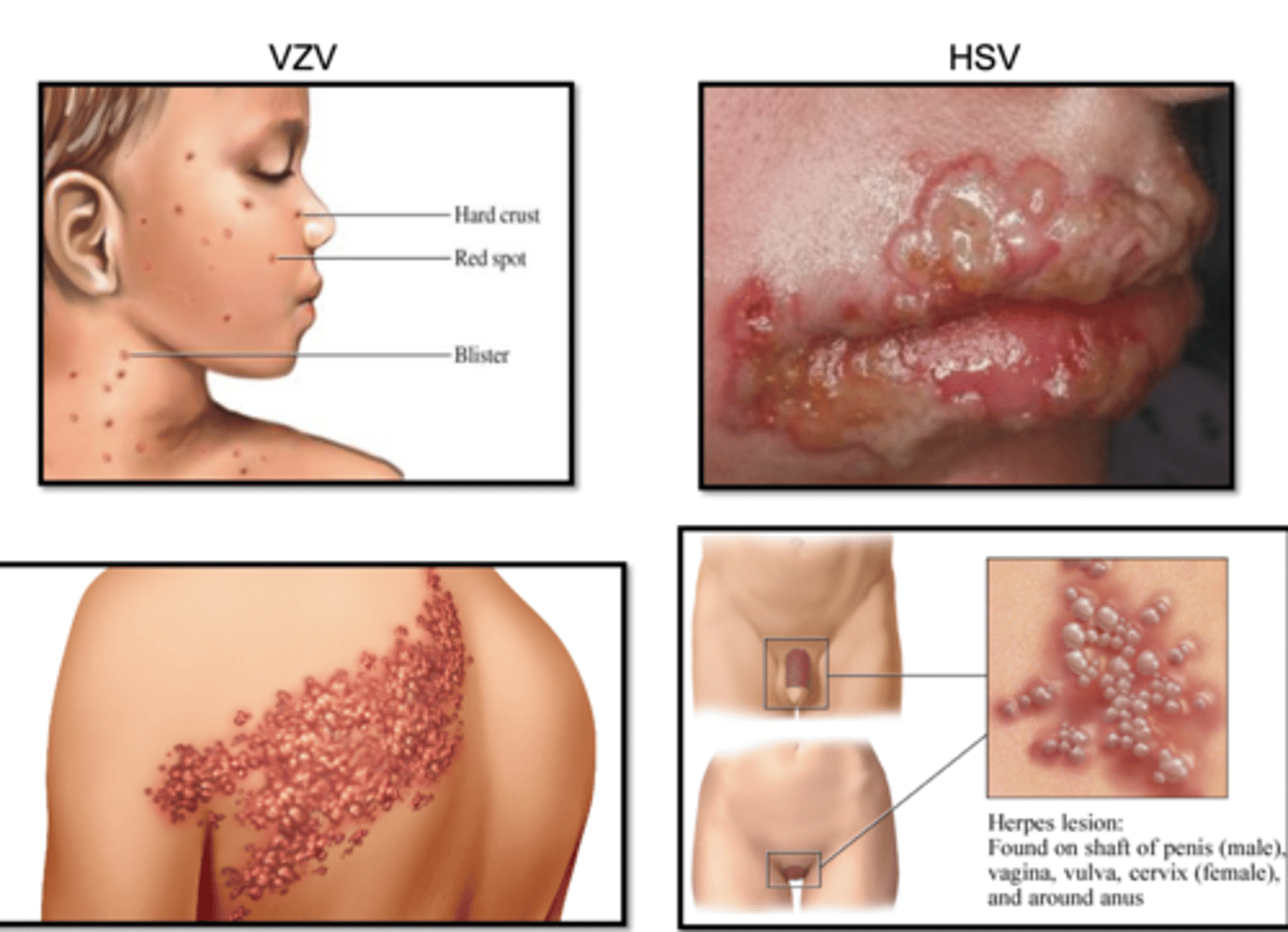
Alpha Herpesviruses
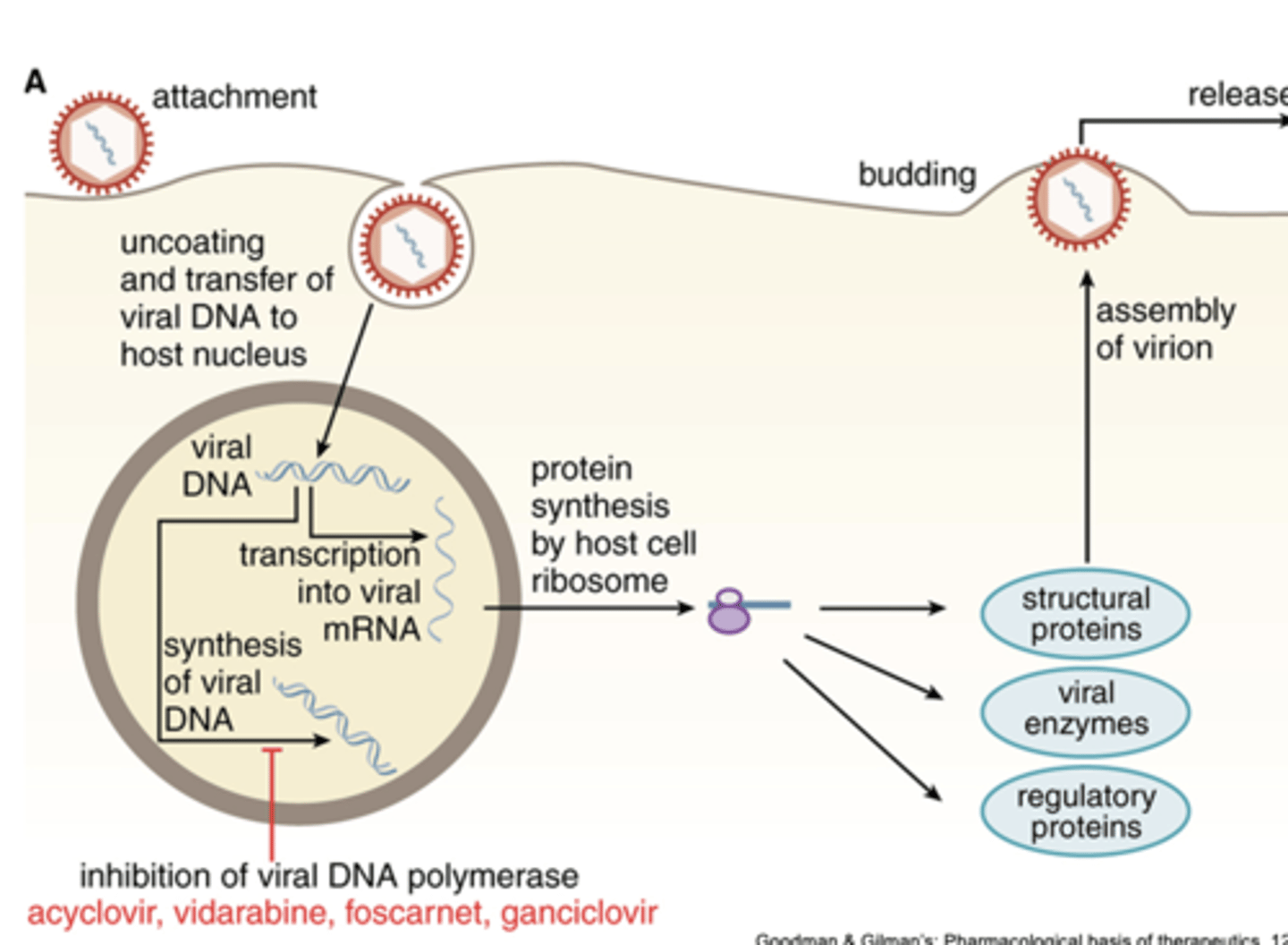
DNA virus entry and replication cycle
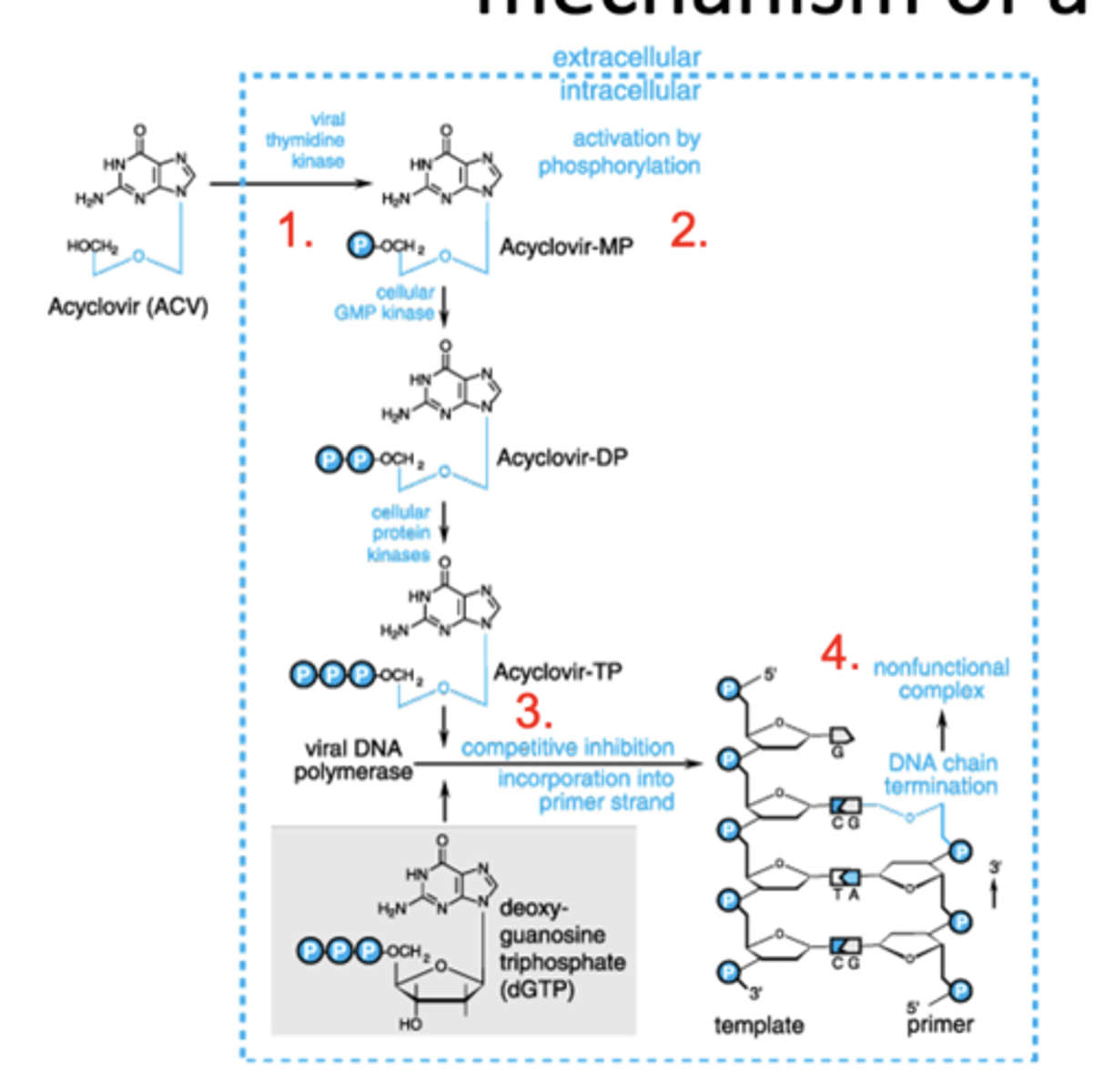
Acyclovir and its prodrug Valacyclovir: mechanism of action
ACV affinity for viral TK is ~200 times greater than cellular kinase
1. Nucleoside analog; monophosphorylated by HSV/VZV thymidine kinase
2. Phospho ACV is trapped/accumulates in infected cell
3. Phospho ACV competes with endogenous nucleotides to inhibit viral DNA polymerase
4. Phospho ACV lacks 3’ hydroxyl group-acts as a chain terminator in viral DNA synthesis
Acyclovir/Valacyclovir uses
Most useful against primary HSV-1 (oral or facial lesions) and primary HSV-2 (genital)
–Treatment associated with significant reduction in shedding, symptoms and recovery times
Spectrum of activity:
–HSV-1 & HSV-2 recurrence: prophylactic use reduces duration
–VZV-used within 24 h of rash onset reduces lesions & duration
–Shingles (herpes zoster)- reduces pain and healing times
Resistance to therapy seen in strains of the virus expressing mutant thymidine kinase or mutant DNA polymerase
adverse effects of ACV
Principle dose limiting toxicities of IV acyclovir:
–Renal and neurotoxicity
Drug Interactions:
–other nephrotoxic agents increase nephrotoxicity
Cidofovir
Nucleotide analog (ie already phosphorylated) with inhibitory activity against herpes, papilloma, polyoma, pox and adenoviruses
Therapeutic uses:
–IV formulation used for treatment of CMV retinitis in HIV+ patients
-- Increases the time to progression of CMV retinitis
-- Has been used in ACV resistant HSV infections
RNA Viruses and disease
Rubella virus (German measles)
Rhabdovirus (rabies)
Picornavirus (polio, meningitis, colds, hepatitis A)
Arenavirus (meningitis, Lassa fever)
Flavivirus (West Nile, yellow fever, hepatitis C)
Orthomyxovirus (influenza)
Paramyxovirus (measles, mumps, RSV)
Coronavirus (colds, SARS, COVID-19)
Retrovirus (HIV)
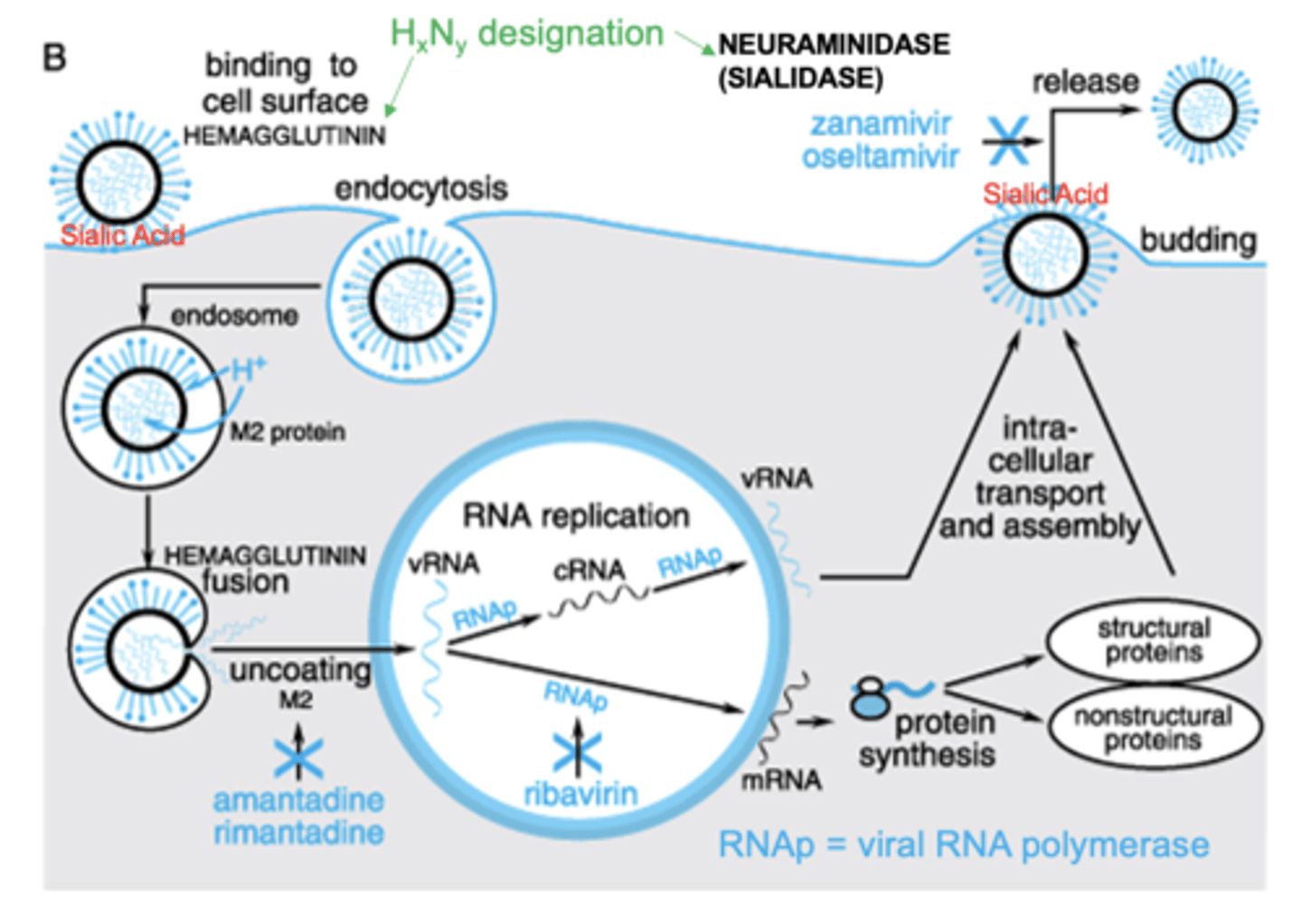
Replication of RNA Viruses (e.g. influenza)
Anti-Influenza Agents
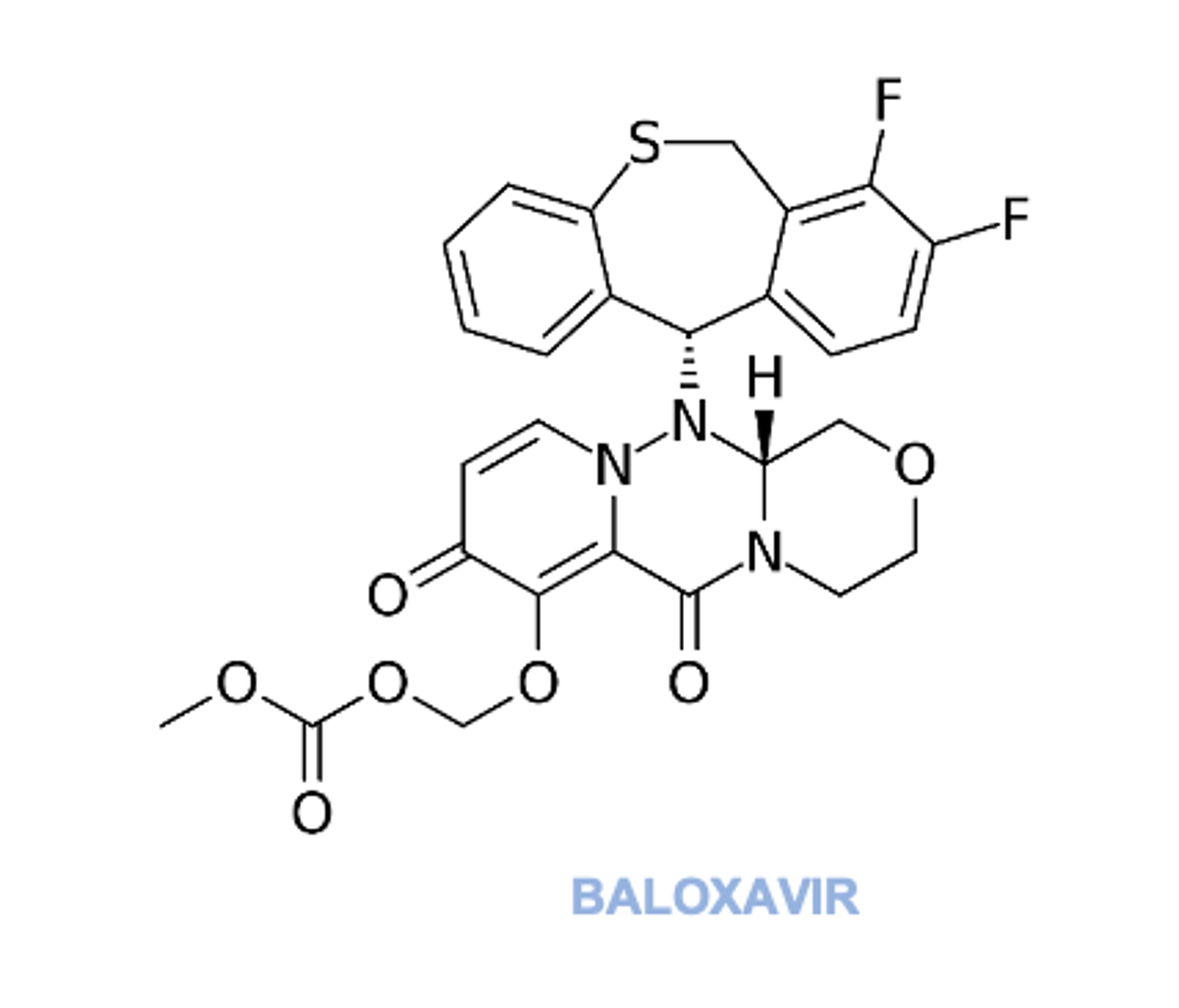
Baloxavir (2018) inhibits influenza A and B RNA polymerase complex, consequently, prevents synthesis of viral proteins
Use within 48 hr of symptom onset
Shortens duration of symptoms
May be used prophylactically
Anti-Influenza Agents (2)
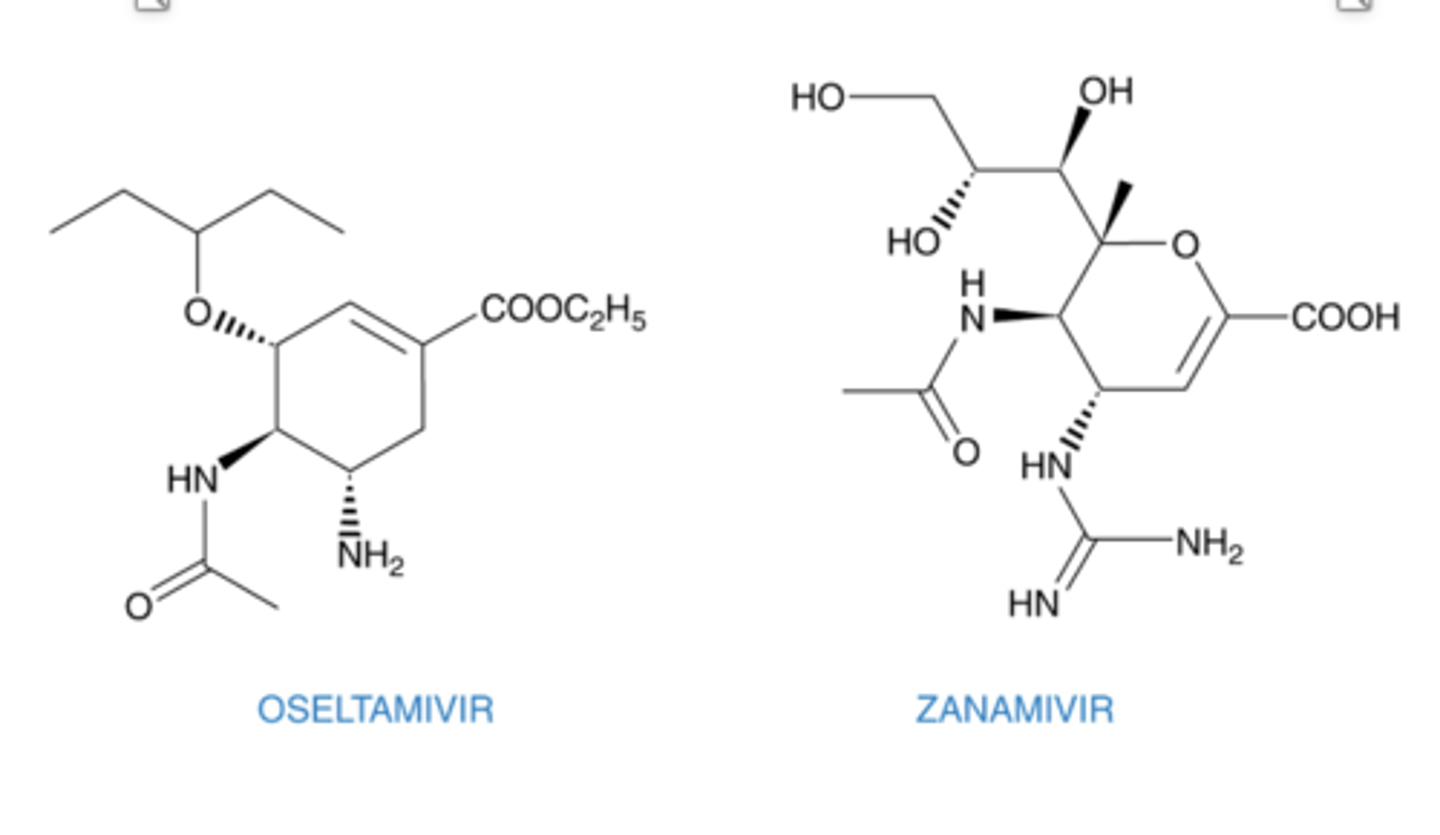
Oseltamivir and zanamivir are neuraminidase inhibitors that inhibit virus release from infected cells
Neuraminidase Inhibitors
block Influenza release
Mimic Sialic Acid and thus inhibit neuraminidase/sialidase
Block virus release and consequently new infection
Zanamivir & Oseltamivir
–Reduce duration by 1-2 d
Zanamivir
–Influenza A & B
Oseltamivir
–Prophylactic use
Anti-hepatitis C Drugs
Don’t memorize these names! Just know that combination drugs targeting two HepC proteins required for replication are therapeutic.
Sofosbuvir - Ledipasvir (Harvoni) - RNA pol and RNA replication complex inhibitors; Hep C genotype 1
Elbasvir - Grazoprevir (Zepatier) - RNA replication complex and protease inhibitor; Hep C genotypes 1 and 4; sometimes with Ribavirin
Sofosbuvir – Velpatasvir (Epclusa) – RNA pol and RNA replication complex inhibitors; all major Hep C genotypes (1-6)
Lecaprevir - Pibrentasvir (Mavyret) - RNA replication complex inhibitor plus protease inhibitor; all Hep C genotypes (less evidence for genotype 3?), 8-week cure in cases
Clinical Uses
Can be curative but also costly.
Adverse Effects
Vary by drug combination; Hep B reactivation.
Anti-SARS/CoV-2 drugs
WILL NOT TEST US ON
FDA approved: Remdesivir – prodrug, nucleotide analog; inhibits RNA-dependent RNA polymerase
Nirmatrelvir-Ritonavir combination – Nirmaltrevir inhibits the SARS/CoV-2 protease that is required to cleave two SARS polyproteins for new virion packaging. Nirmaltrevir is metabolized by CYP3A4. Ritonavir (an HIV protease inhibitor) is used here only to inhibit CYP3A4 and hence boost Nirmatrelvir to required therapeutic conc.
(There are other drugs with emergency use authorization, for updated list see CDC.gov)
HIV Chemotherapy
Goal- Suppress virus replication as much as possible for as long as possible
Reservoirs of long-lived quiescent T-cells harboring latent (non-replicating) HIV DNA integrated into the host chromosomes are not targeted by anti-retrovirals and so cure remains elusive (rare reports to the contrary)
antiretrovirals can prevent virus transmission to fetus in HIV+ pregnant women
Drug resistance is a significant problem-
–HIV has a high mutation rate
–Monotherapy promotes emergence of resistant viruses
HIV structure
Small RNA genome
Surrounded by lipid bilayer that is derived from host cell, plus viral proteins essential for attachment and fusion to target cells
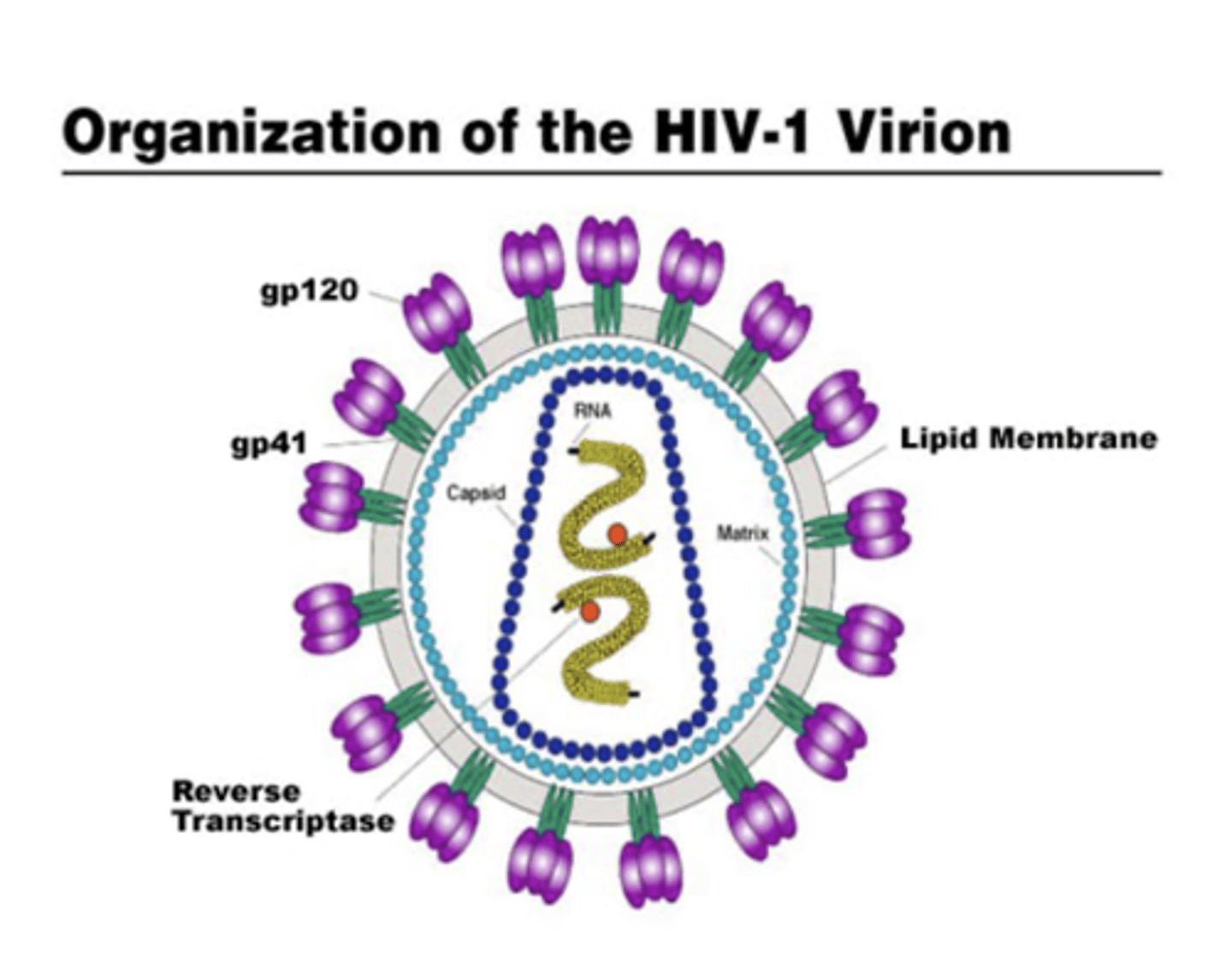
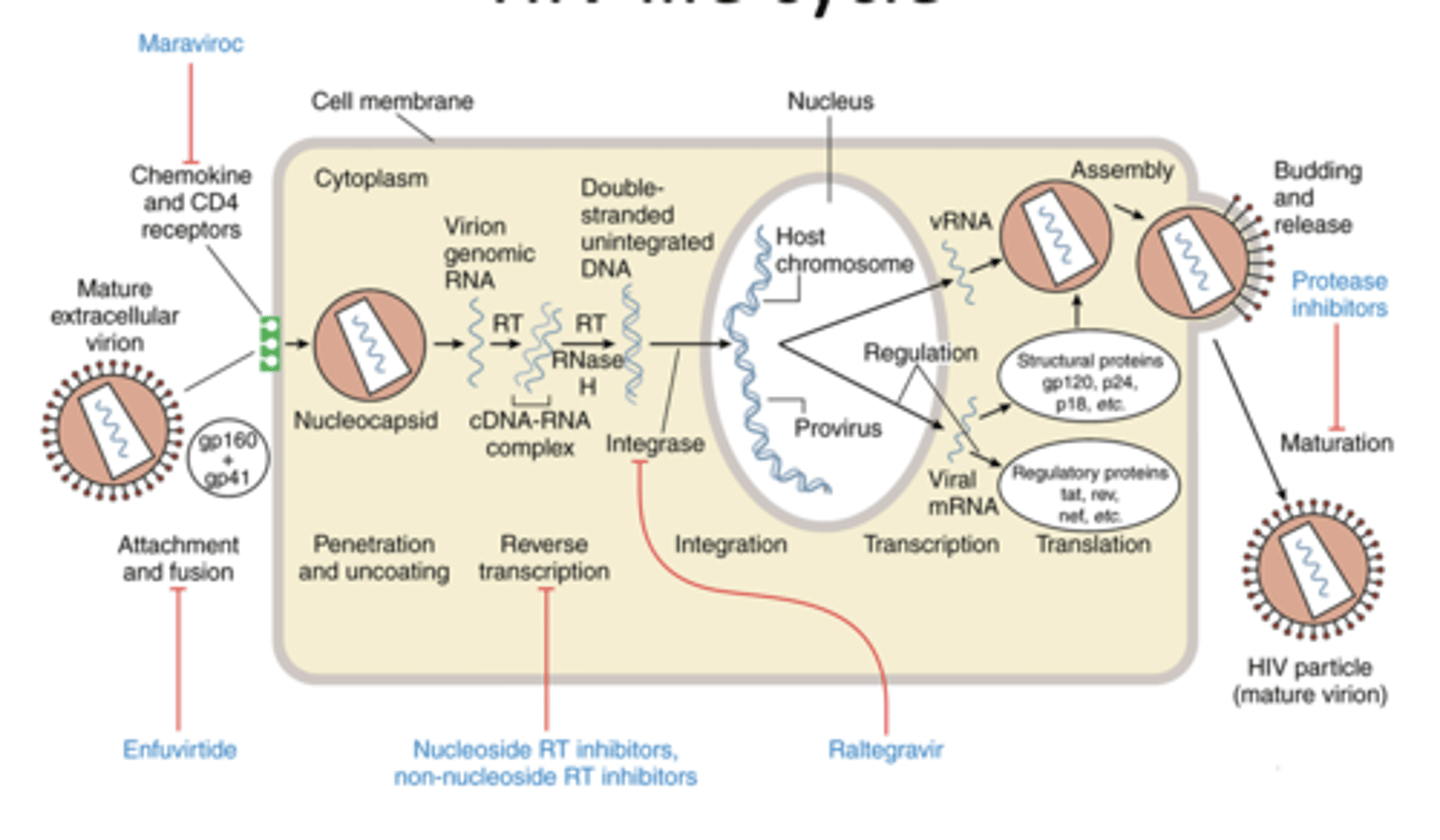
HIV life cycle
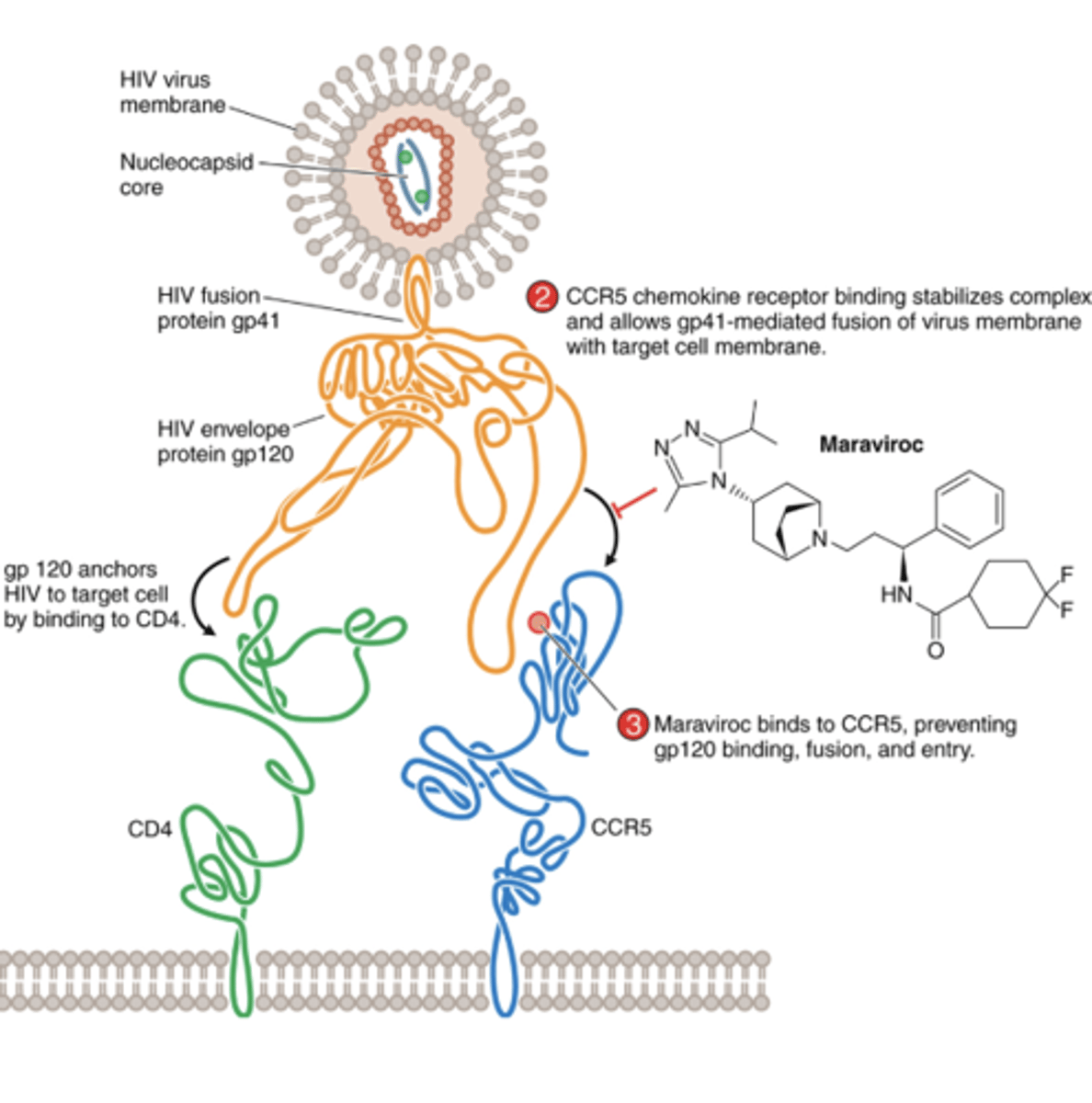
HIV Entry Inhibitors (Maraviroc)
Blocks binding of the HIV outer envelope protein gp120 to the CCR5 chemokine receptor (chemokine receptor antagonist)
Is only active against CCR5-tropic strains of HIV
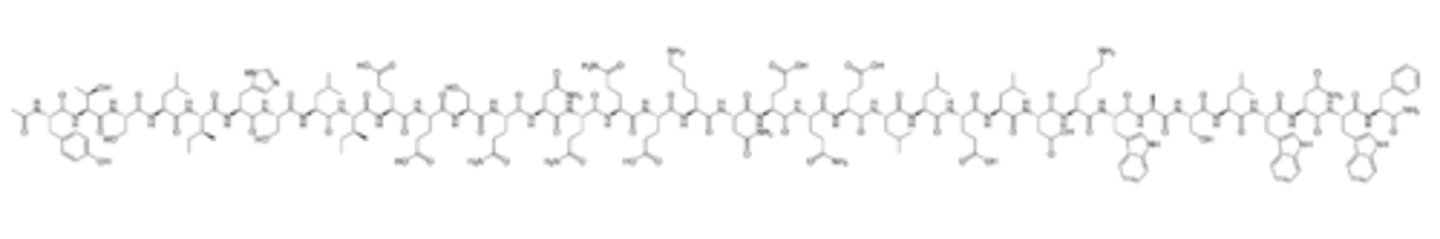
HIV Entry (fusion) Inhibitors (Enfuvirtide)
Peptide drug that binds to the HIV envelope protein gp41
Inhibits gp41-mediated fusion of the virus envelope with the cell membrane
enfuvirtide: others
pharmacokinetics:
- only approved parenteral HIV drug
- route of elimination not determined
adverse effects:
- cyst development at injection site
drug interactions:
- none known
resistance:
- specific mutations to the gp41 domain
therapeutic use:
- approved only for use in treatment - experienced adults who have HIV replication despite ongoing antiretroviral treatment and who can self-inject
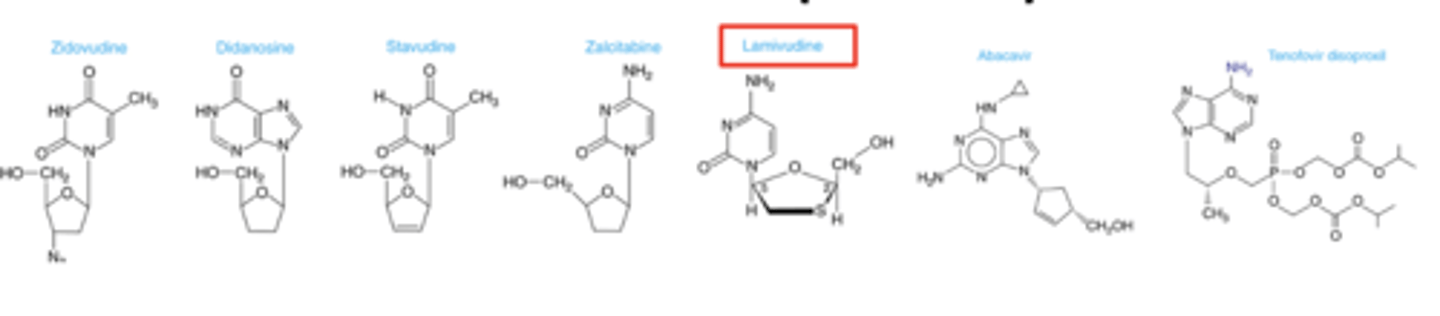
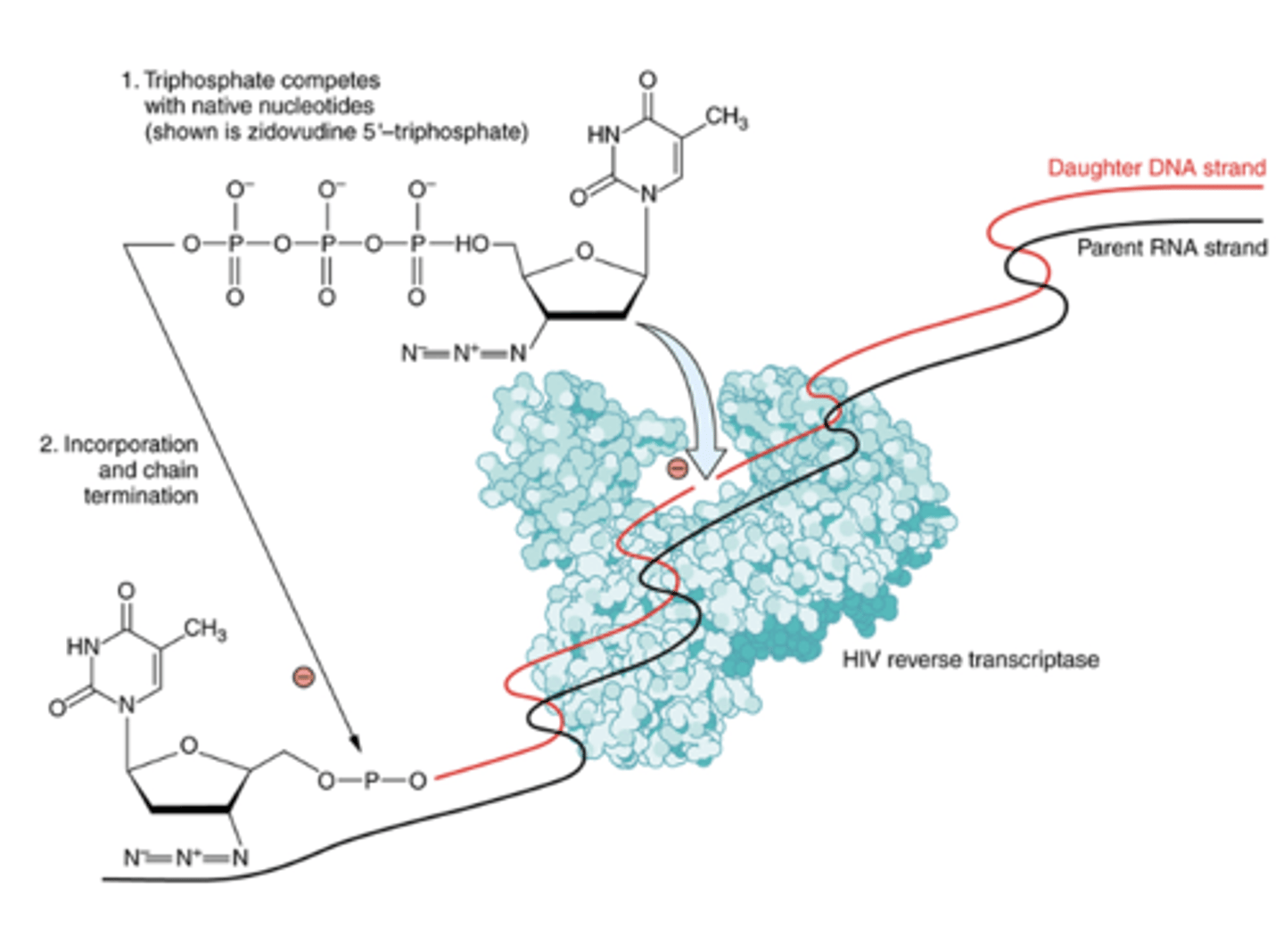
Nucleoside and Nucleotide RT Inhibitors (NRTIs)
Reverse Transcriptase (RT)-- (RNA-dependent DNA polymerase)-copies viral RNA into proviral DNA for integration into host cell chromosome
Drugs prevent infection of susceptible cells but does not eradicate integrated proviral DNA ie not curative
NRTI mechanism of action
NRTI’s competitively inhibit nucleotide binding to reverse transcriptase
- compete with endogenous nucleotides for binding to the active site of RT
- Lack the 3’ hydroxyl group and terminate the DNA chain (recall similar mechanism for acyclovir).
- Nucleoside analogs must be phosphorylated for activity
- Selectively inhibit HIV RT over human DNA polymerase
- Some inhibit a mitochondrial DNA polymerase (causes major adverse effect: myopathy)
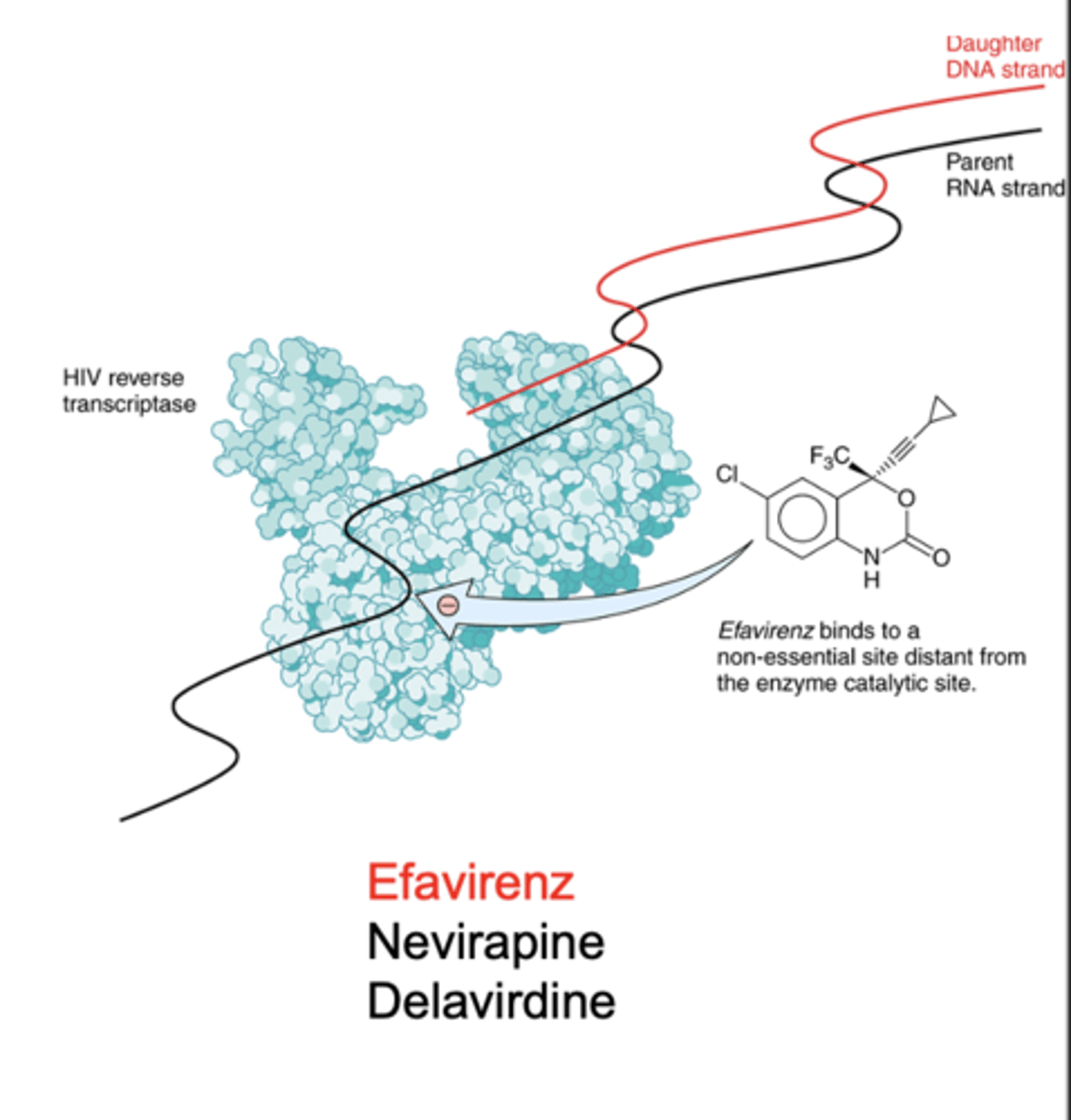
Non-Nucleoside RT Inhibitors (NNRTIs)
Bind hydrophobic pocket distinct from the active site in HIV reverse transcriptase
This pocket is non-essential for enzyme function
Induces a conformational change that reduces enzyme activity
- Non-competitive with nucleotides
- Do not require intracellular phosphorylation
- No activity against DNA polymerase
Efavirenz
Nevirapine
Delavirdine
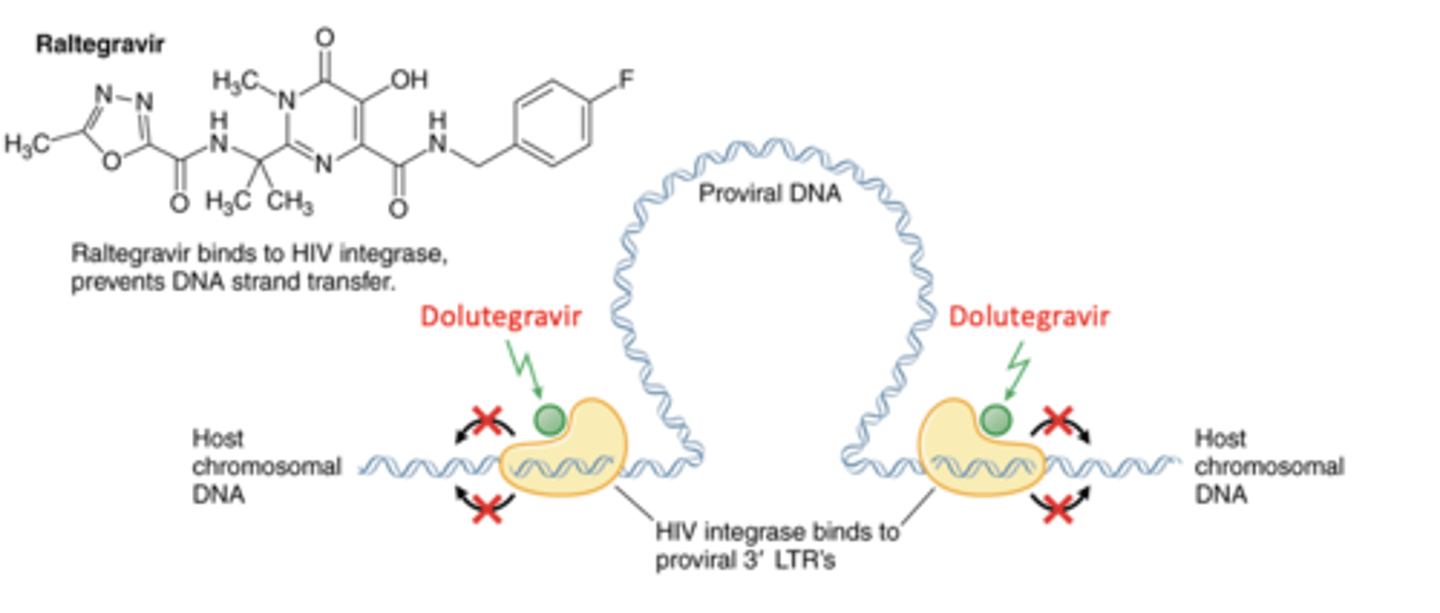
HIV Integrase Inhibitors (cabotegravir, dolutegravir)
Prevents virus integration into host chromosome
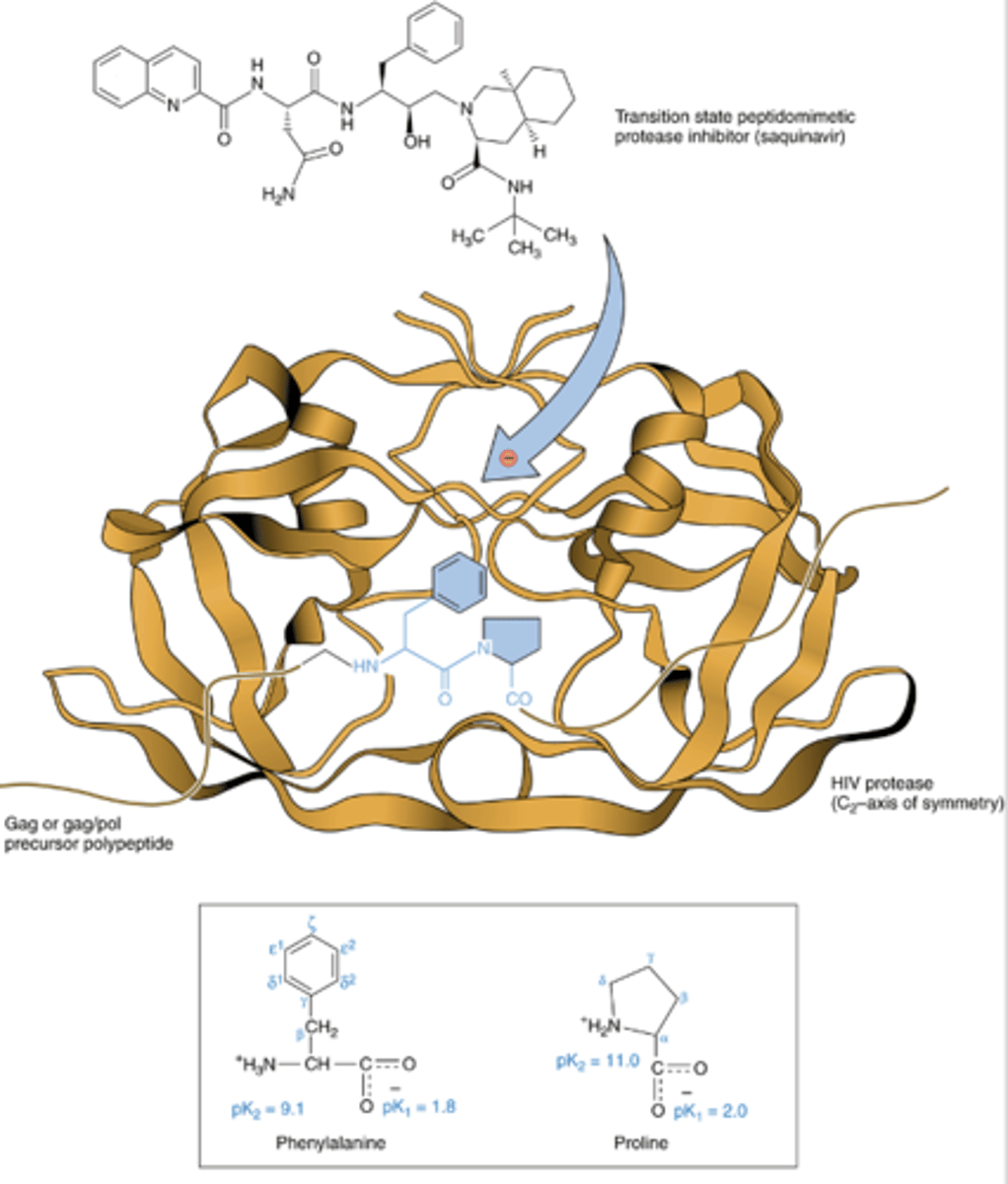
HIV Protease Inhibitors (2)
Competitive inhibitors of HIV protease
-- Saquinavir; ritonavir; lopinavir; indinavir; nelfinavir; tipranavir
Prevent proteolytic cleavage of HIV gag and pol precursor polyproteins
- (gag) Essential structural proteins
- (pol) Essential enzymatic proteins
Prevents maturation of virus to infectious form
HIV Protease Inhibitors: others
pharmacokinetics:
- variable oral bioavailability, hepatic metabolism, plasma protein binding
adverse effects:
- most commonly associated with GI disturbances
- fatal hepatotoxicity has been reported with some analogs
- crystalluria and nephrolithiasis stemming from poor drug solubility
drug interactions:
- substrates and inhibitors of CYP 3A4 - many interactions
resistance:
- primary resistance in the enzymatic active site
- secondary mutations distant to active site leading to reduced proteolytic efficiency
HAART: Highly Active Anti-Retroviral Therapy
Monotherapy is problematic and never used
-- Considerable toxicity for only modest, transient decrease in viral load
-- Resistance develops due to very frequent mutation of viral genome
Combination therapy for HIV: typically 3 drugs, sometimes more. Reverse transcriptase + Protease + Integrase + other inhibitors: different mechanisms of action and of resistance; likelihood of simultaneous resistance to all is very low.
Recently: two drug regimens (e.g. dolutegravir/lamivudine, integrase inhibitor/NRTI) were approved for first line treatment.
Other two drug regimens approved for maintenance, some with dosing every other month.
Advantages of combination therapy:
- More effective at lower individual drug doses
- Reduced toxicity
- Resistance much less likely
- Long term reduction in viral load to undetectable: HIV infection now manageable long term, albeit not curable
Anti-retroviral therapy during pregnancy
-- Can reduce the risk of perinatal transmission to ~1% or less
-- Preferred combinations of three drugs should be started as early in pregnancy as possible to achieve zero detectable virus
PrEP: pre-exposure prophylaxis
Original drug: combination of tenofovir and emtricitabine (both reverse transcriptase inhibitors) prescribed to reduce (>90%) risk of infection; requires daily dosing (oral), regular HIV testing
New extended release cabotegravir (integrase inhibitor); dosing every two months (IM). Testing required before each cabotegravir injection.
Patient compliance is essential for maximum benefit
Patient MUST be HIV negative: these agents are not sufficient to treat ongoing HIV infection. Testing required!
For maximum benefit combine drugs with behaviors that reduce risk of infection