Shellfish Investigation: L2 - L4
1/28
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
shellfish
organism that lives in water and has a shell

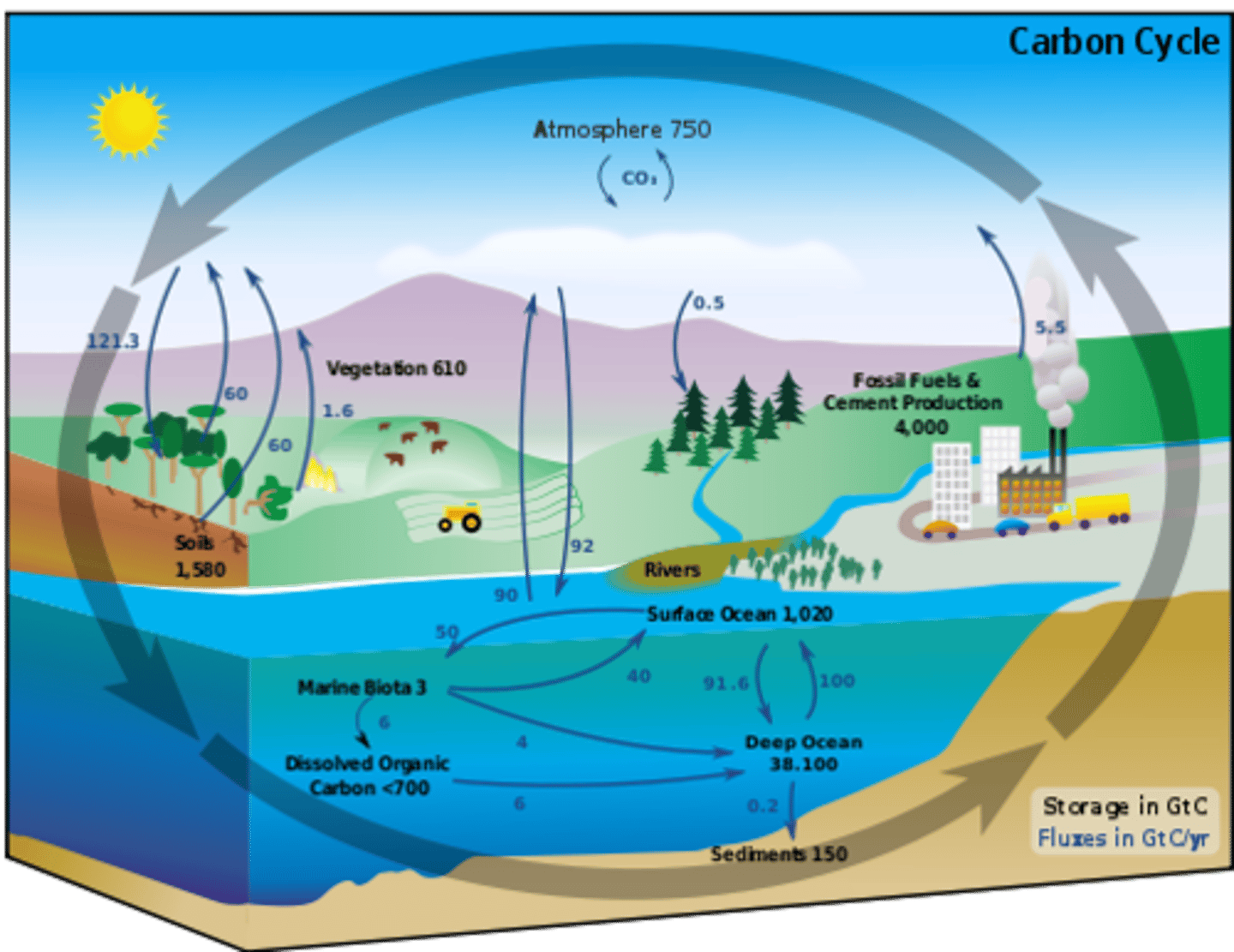
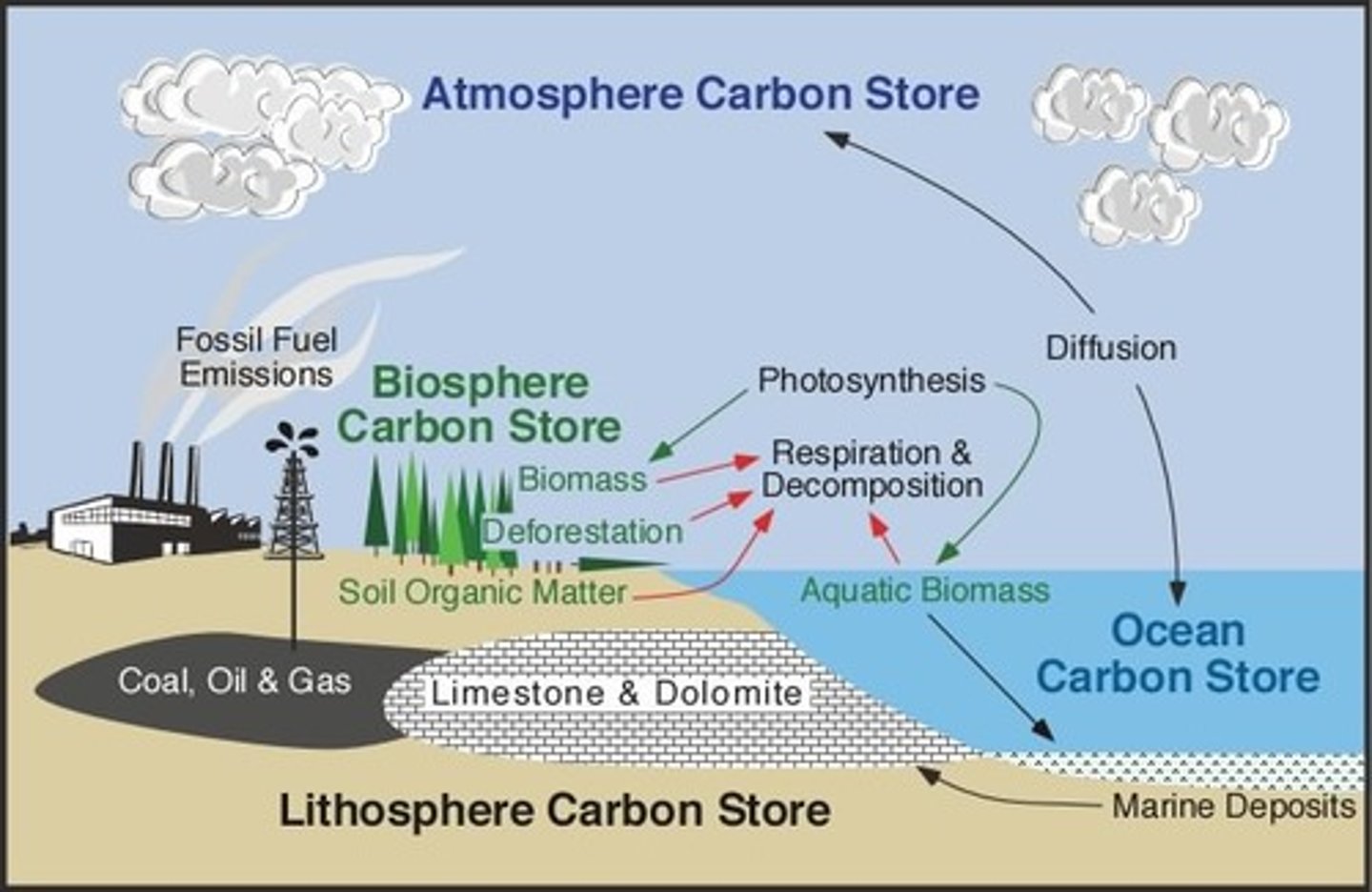
carbon cycle
the movement of carbon from the nonliving environment into living things and back
combustion
the chemical reaction that involves burning a fuel and produces carbon dioxide

fossil fuels
a natural fuel such as coal or gas, formed in the geological past from the remains of living organisms.

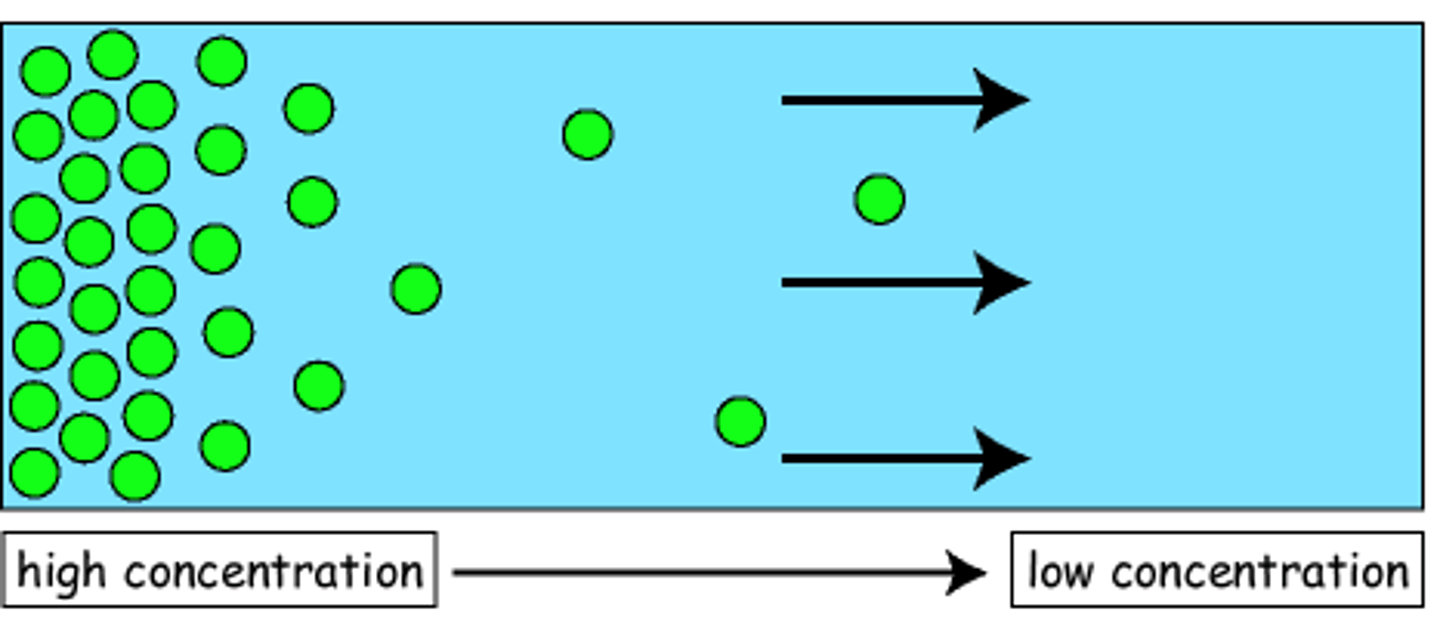
diffusion
Movement of molecules from an area of higher concentration to an area of lower concentration.
Carbon sinks/reservoirs
Anything that stores carbon atoms in the Carbon Cycle; examples: living things, soils, fossil fuels, oceans and the atmosphere
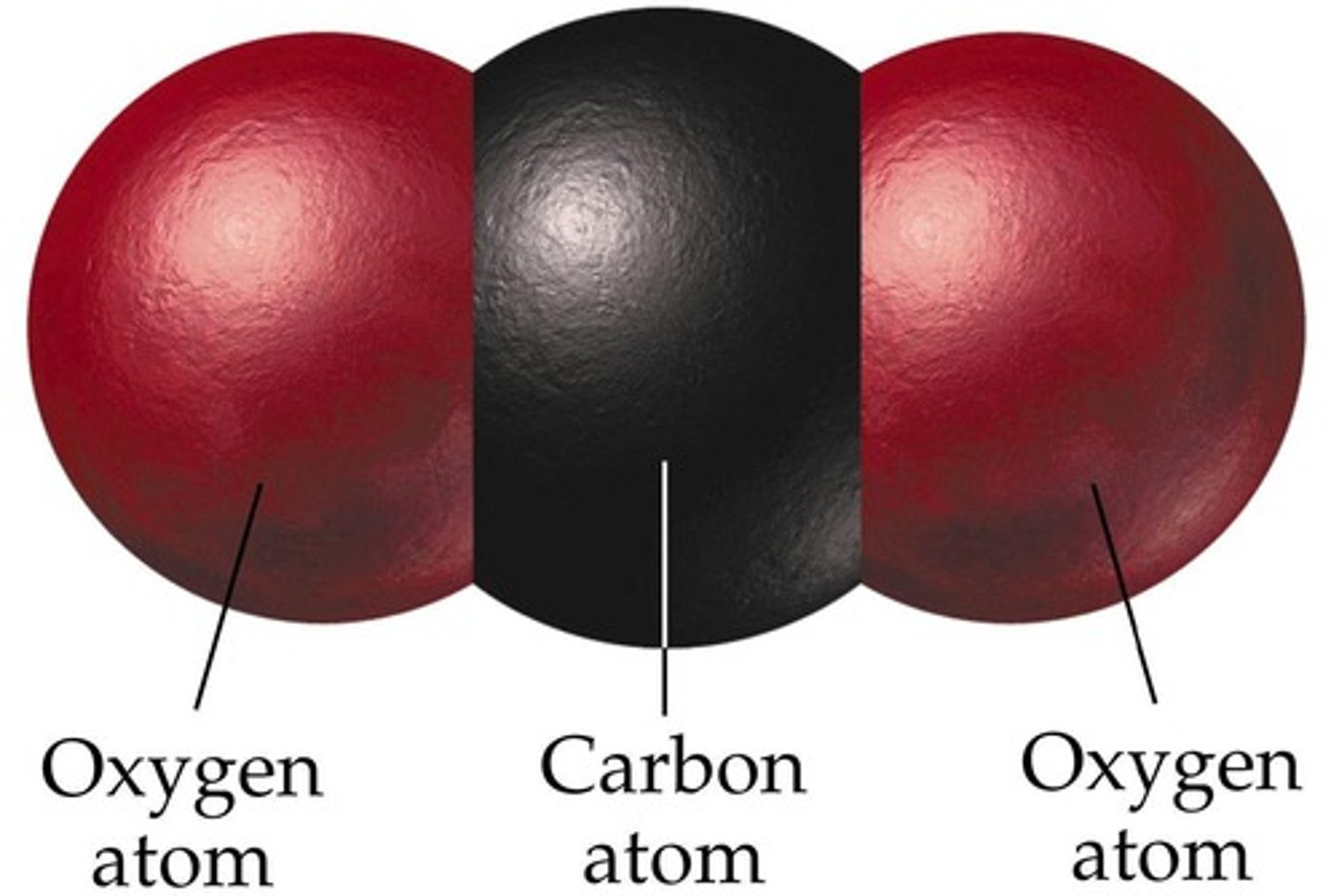
carbon dioxide
CO₂; a colorless, odorless gas
pH indicator
Chemical dye that changes color in response in changes to acidity of solution

pH scale
Measures the strength of acids and bases low ph=strong acid, high ph=strong base; 0-14

acid
Substance that creates hydrogen ions (H⁺) in solution; pH below 7
base
Substance that creates hydroxide ions (OH⁻) in solution or accepts hydrogen ions; pH above 7
neutral
A substance that is neither acidic or basic; pH of 7.
equilibrium
the concentration of particles is equal across space
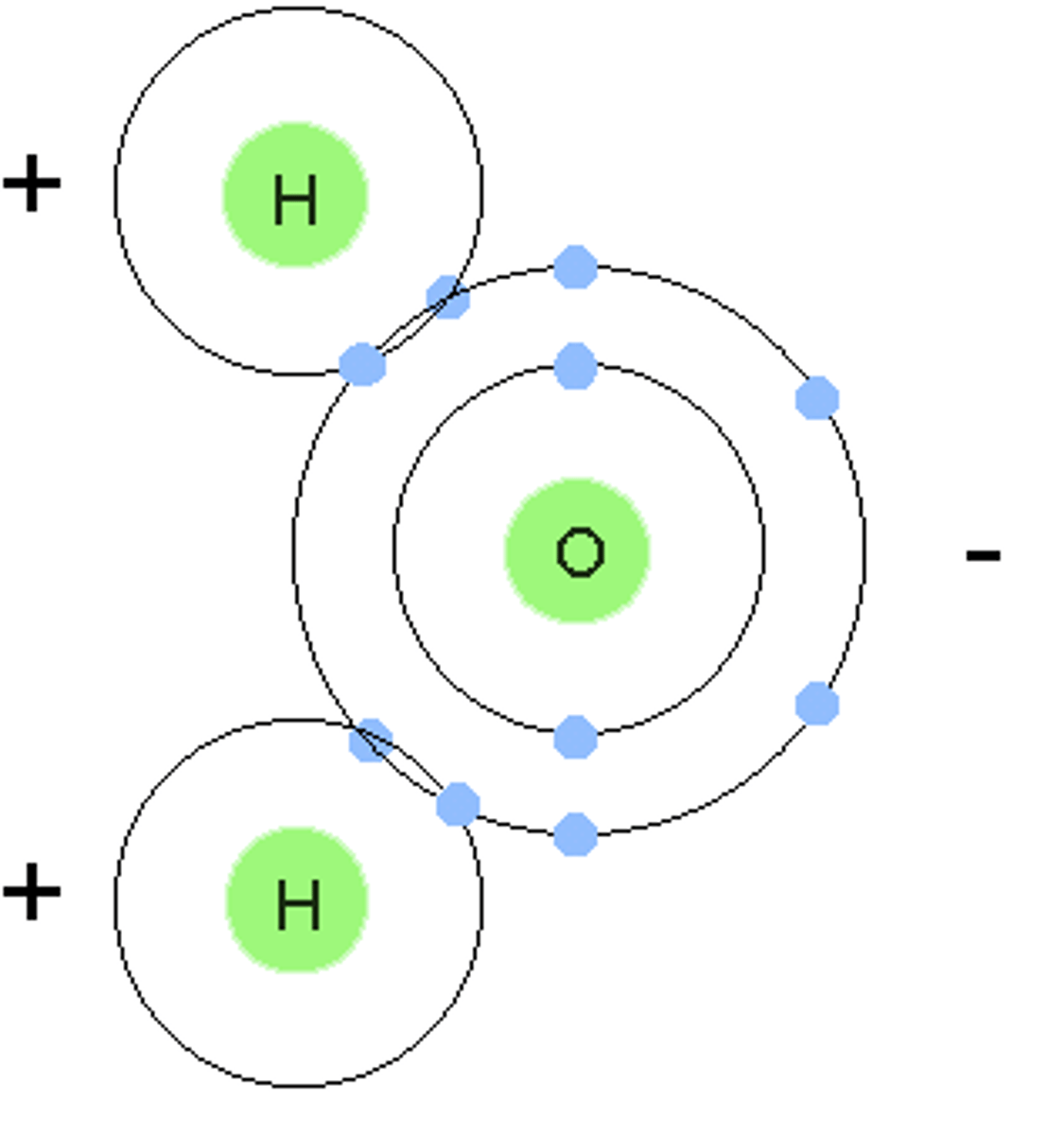
polar molecule
a molecule in which one end is more negatively charged than the other; example: water
non-polar molecule
a molecule with no polar bonds or charges; example: oil
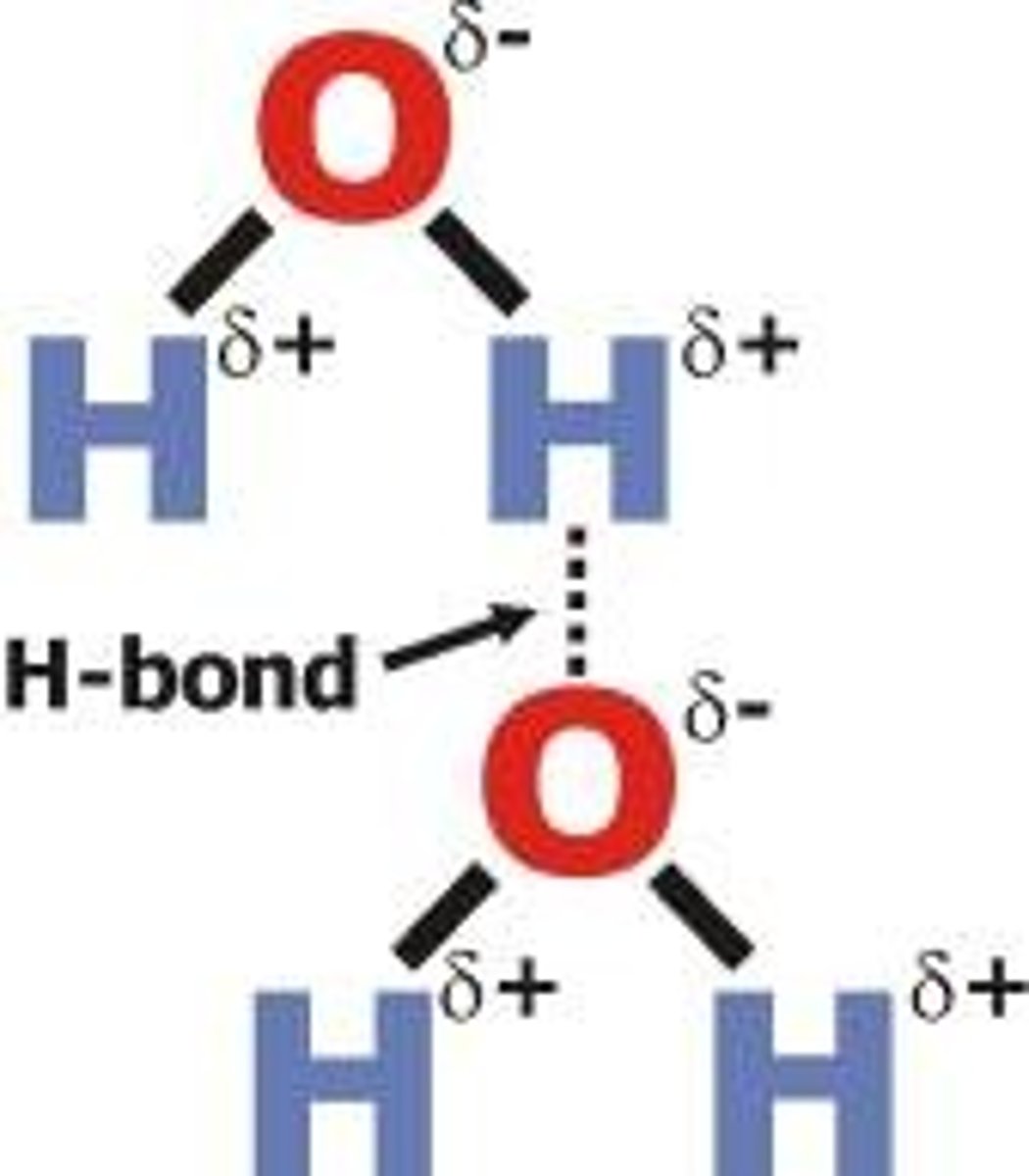
hydrogen bond
weak attraction between a hydrogen atom and another atom on a different molecule; example: temporary bond between two water molecules
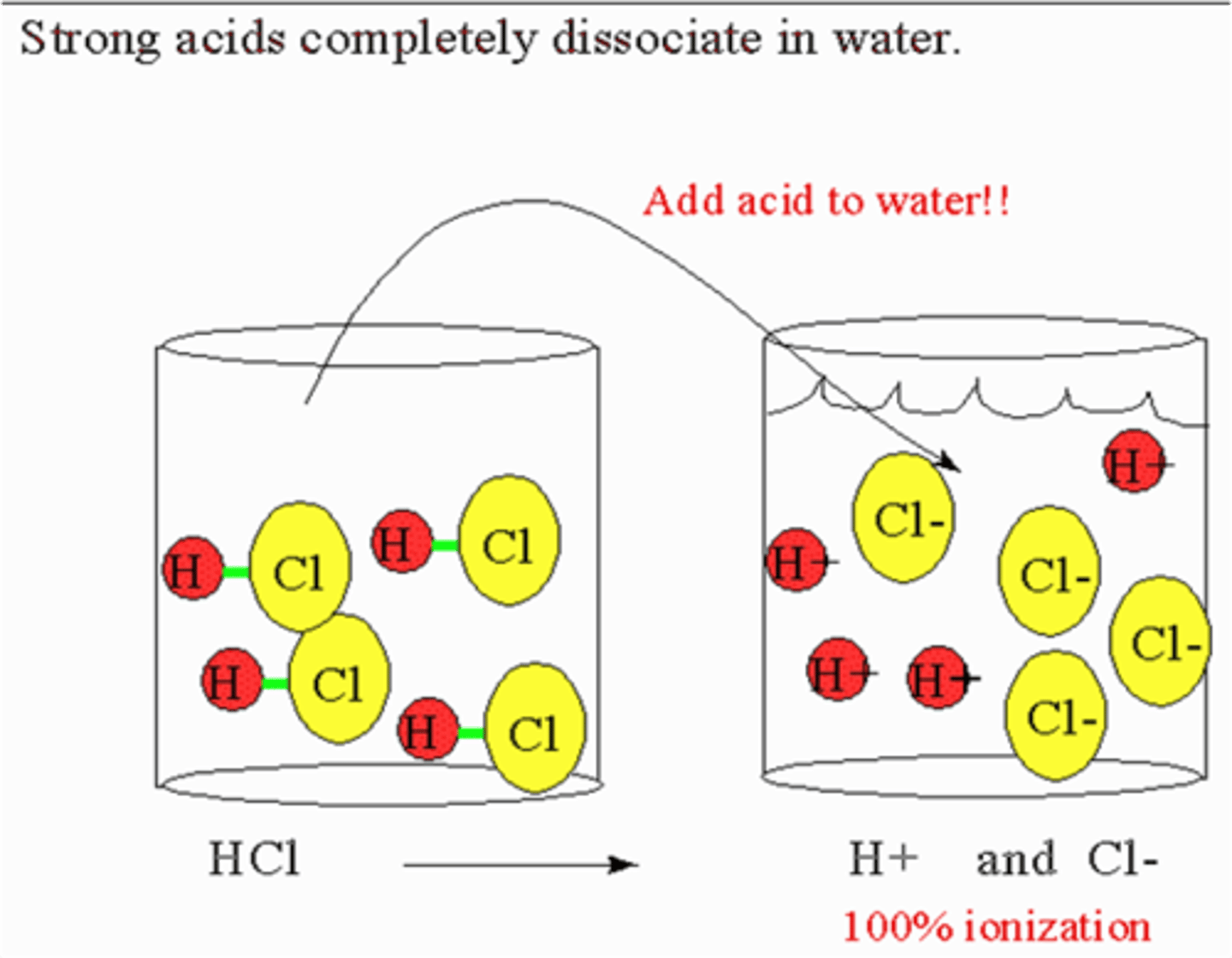
strong acid
an acid that produces a large amount of H⁺ ions
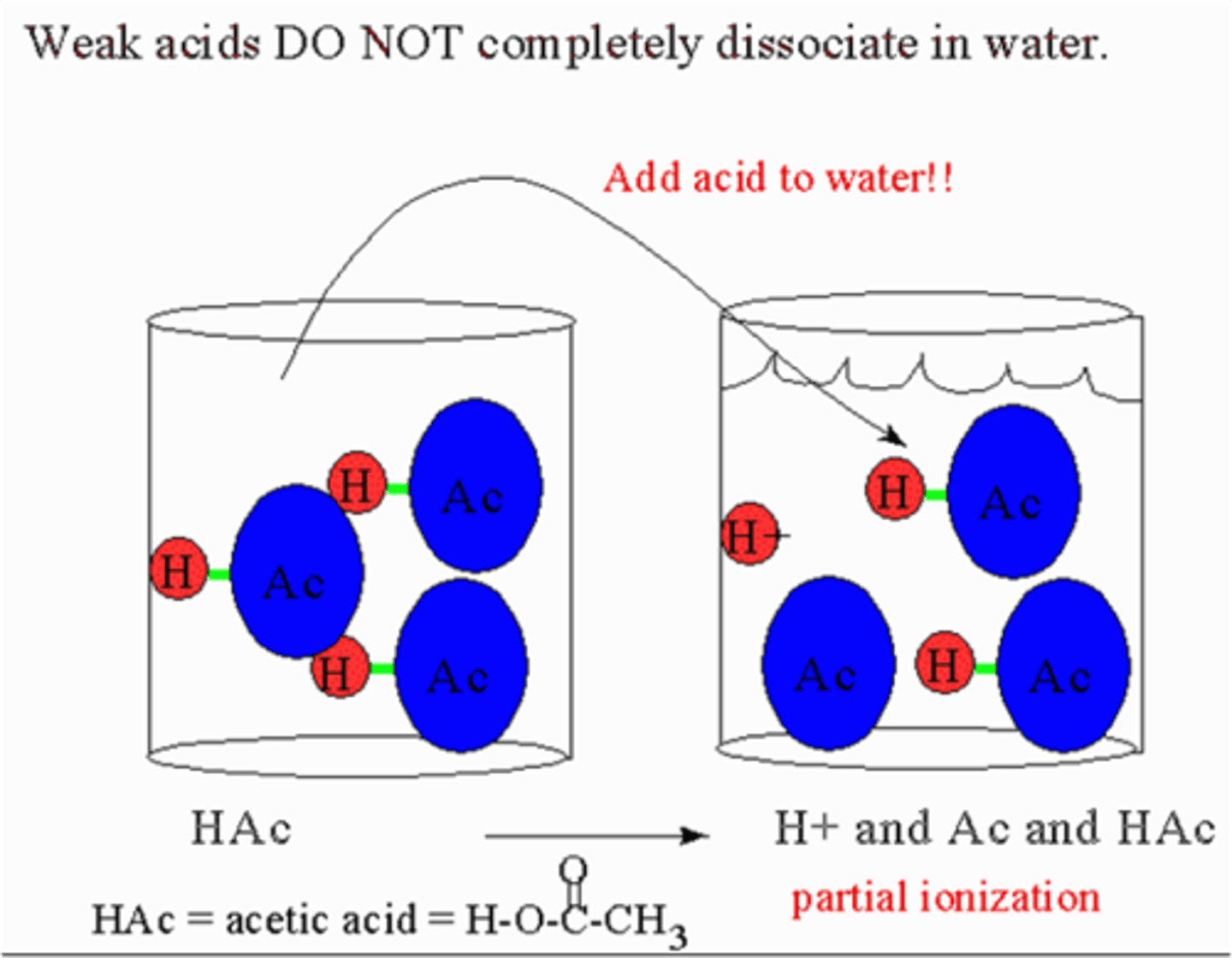
weak acid
an acid that produces a small amount of H⁺ ions
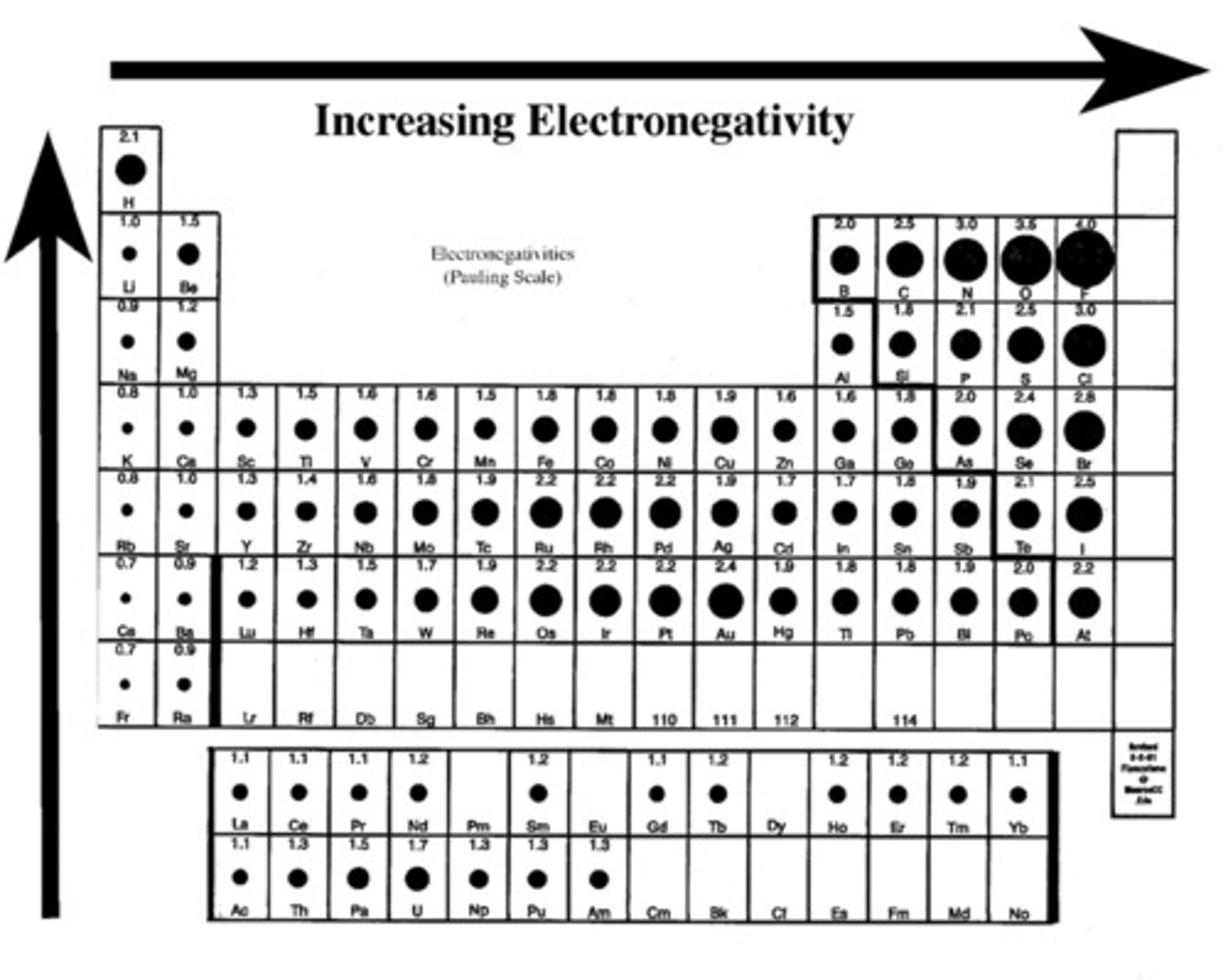
electronegativity
Measure of the ability of an atom to attract electrons in a chemical bond; higher electronegativity will have a stronger pull on the electrons
solubility
The amount of a substance that dissolves in a given quantity of solvent to make a saturated solution at constant temperature and pressure

examples of acids
HCl; H₂SO₄; HNO₃
examples of bases
NaOH; Ca(OH)₂; KOH
reactants
the chemicals present before the reaction (left of the arrow)
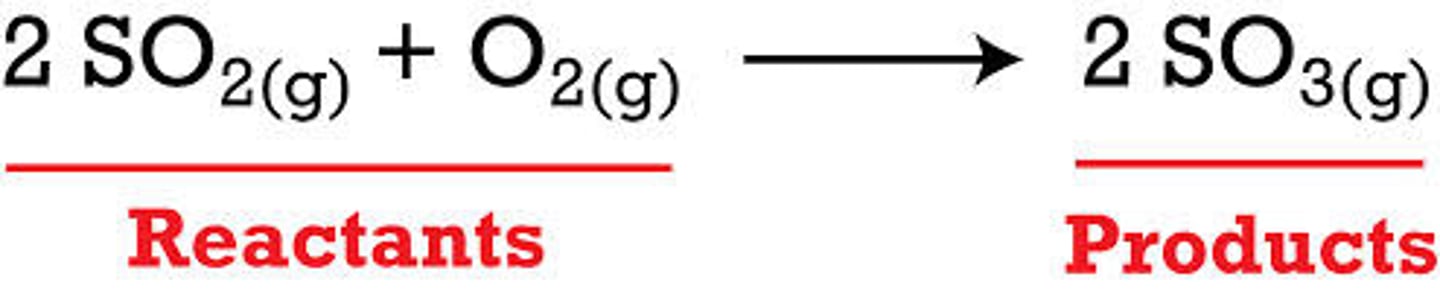
products
the chemicals present after the reaction (right of the arrow)
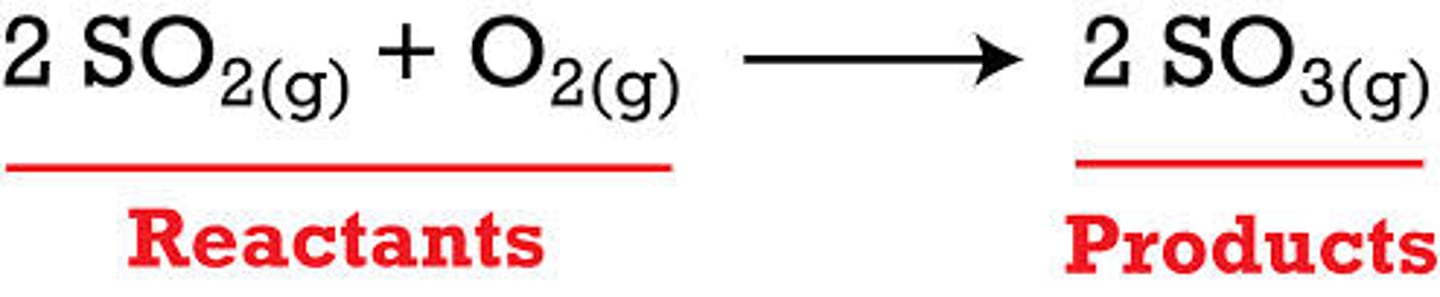
ocean acidification
The process by which the pH of the ocean is lowered (made more acidic) when excess CO₂ dissolves in seawater and forms carbonic acid (H₂CO₃)
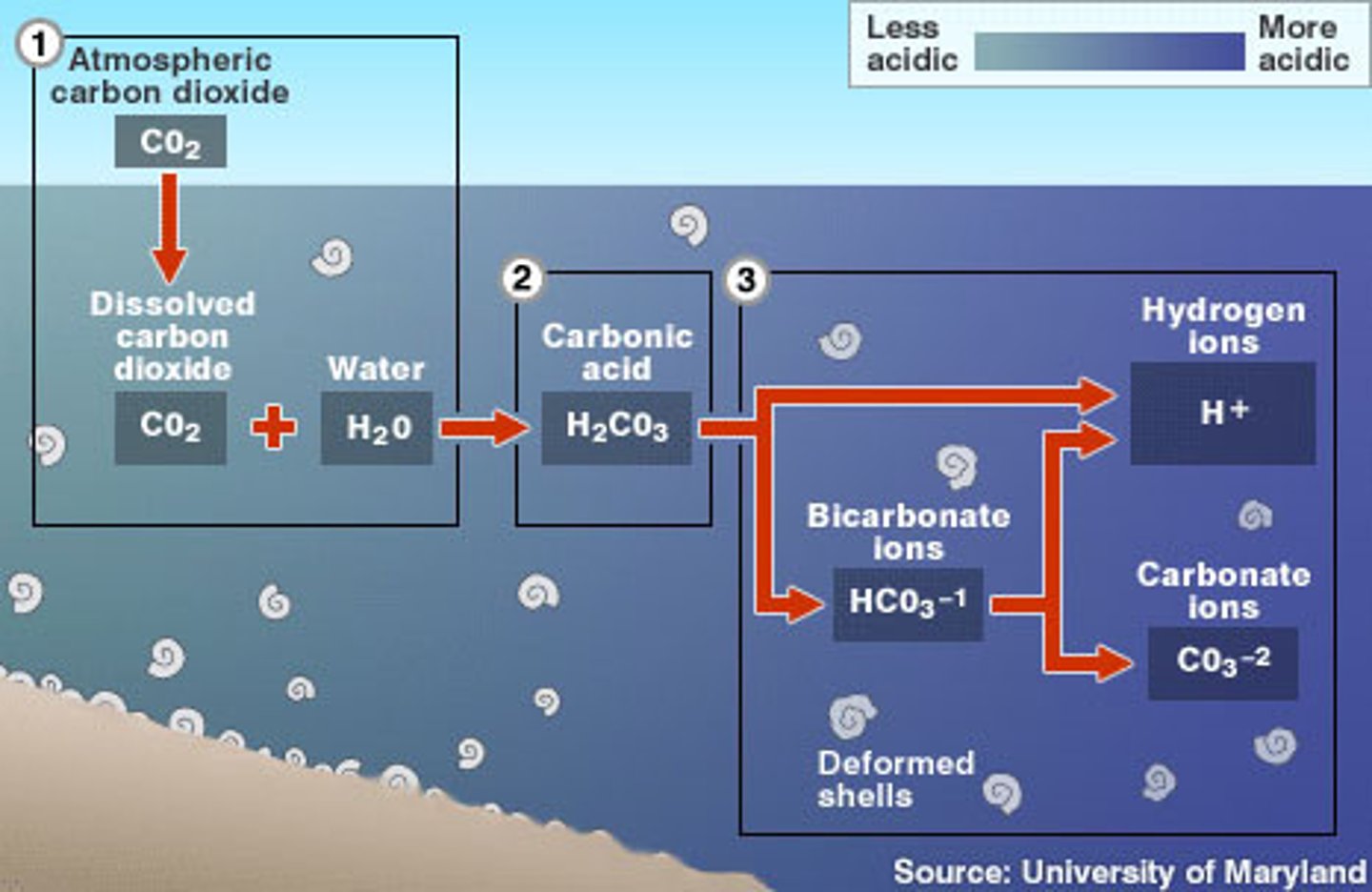
first chemical equation in the ocean acidification process
CO₂ + H₂O ⇌ H₂CO₃
second chemical equation in the ocean acidification process
H₂CO₃ + H₂O ⇌ HCO₃⁻ + H₃O⁺
third chemical equation in the ocean acidification process
HCO₃⁻ + H₂O ⇌ CO₃²⁻ + H₃O⁺
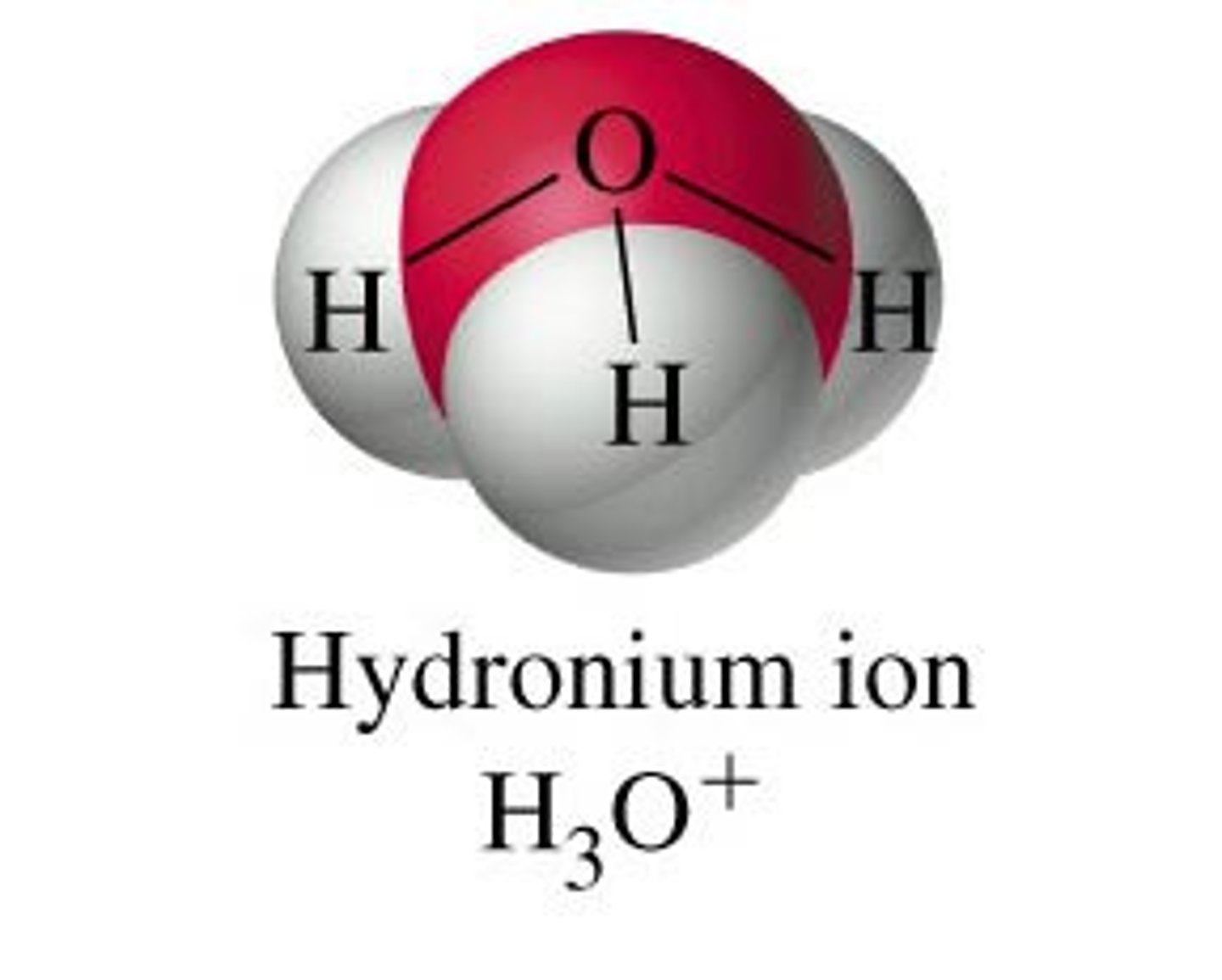
molecule that causes the pH of the oceans to decrease
H₃O⁺ (hydronium)