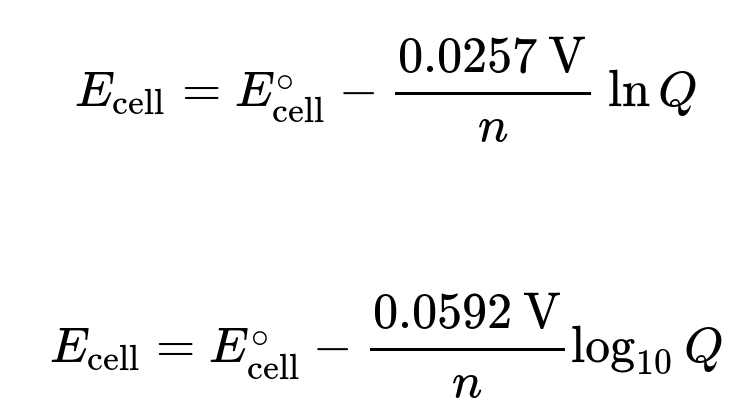Ap Chem Stuff
1/13
Earn XP
Description and Tags
For Pierce o7
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Strong acid
H2SO4, HNO3, HClO4, HI, HBr, HCl | So I Brought No Clean Clothes
Oxidation number
Na+ = 1 and Cl- = -1 | F always -1 | H usually +1 | O is usually -2
Integrated rate law half life
substitue [At] with ½ A0 | x = A0 | 2nd: 1/kx | 0: x/2k
Always Soluble
Always Soluble: Group 1 Ions, Nitrate (NO3-), Acetate (C2H3O2-), Ammonium (NH4+)
Usually Soluble
Halides: Cl-, Br-, F- but important exceptions being Silver (Ag+), Mercury (Hg2+2), and Lead (Pb2+) | Sulfate (SO42-) Except group 2 ions
Mostly Insoluble
Carbonate (CO32-), Phosphate (PO43-), and Hydroxide (OH-). Exceptions being group 1 or NH4+ or Nitrate
Ammonium
NH4+
Nitrate
NO3-
Sulfate
SO42-
Carbonate
CO32-
Phosphate
PO43-
Hydroxide
OH-
Cyanide
CN-
nernst equation
Bottom is at standard condition