Reaction Kinetics
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
what is the gibbs free energy equation

what is ΔG equal to
-change in enthalpy minuc energy req to drive entropy change
what must the value of ΔG be for a reaction to be spontaneous
-ΔG must be negative
can we tell the kinetics of a reaction from delta G
no
what can ror be changed by
-change of temp: more collisions
-reactant conc/ parital pressure
-addition of catalyst
how to calculate RoR
-calculate rate of change
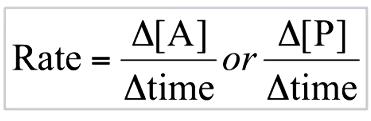
how to find the initial ror from time conc graph
-tangent when t=o then find gradient
how to calc order of reaction from rate constant and rate
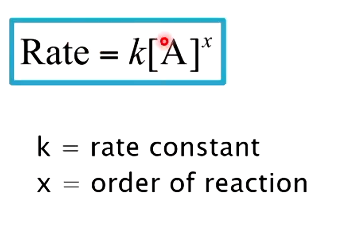
what does it mean if reactions is 0 order
-rate of reaction changes independently of reactant concentration
why are most catalysed reactions zero order
-reaction conc outweight conc of enzyme or catalyst present
-number of binding sited on enzymes is full
-ror depends on catalyst conc
what does it mean if a reaction is first order
-rate is proportional/linearly dependant to the conc of reactant
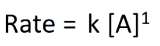
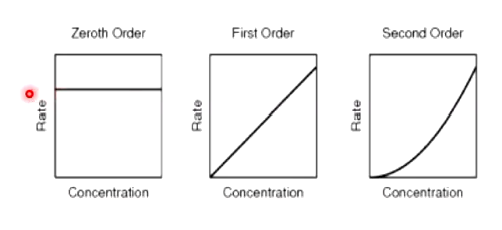


why do we use half life
-very difficult to get accurate measurement of initial rates
-half life is easier to measure
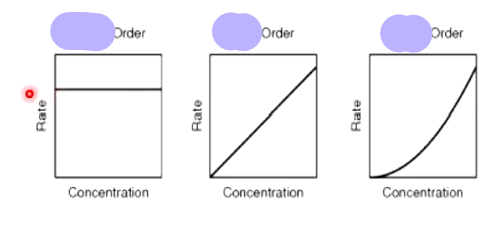
which order has a constant half life
1st order as it has exponential decay

