Modelling Chemical Reactions
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
What is an Atomic Number?
Atomic number refers to the number of protons (+ positive) particles found in the nucleus of an atom.
In each neutral atom the number of Protons (+ positive) particles is equal to the number of electrons (- negative) particles found in the electron shells, electrons orbit the nucleus in electron shells.
What is an Atomic Mass number?
Atomic mass is the sum of the relative mass’s of all the protons, neutrons, and electrons in a single atom or molecule.
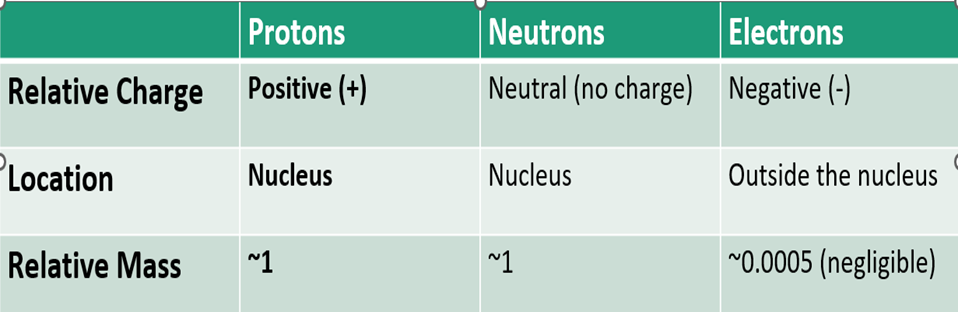
Where are each type of sub-atomic particle located in an atom?
Electrons orbit the nucleus in electron shells.
Neutrons (neutral or no charge) particles are found in the nucleus with the protons.
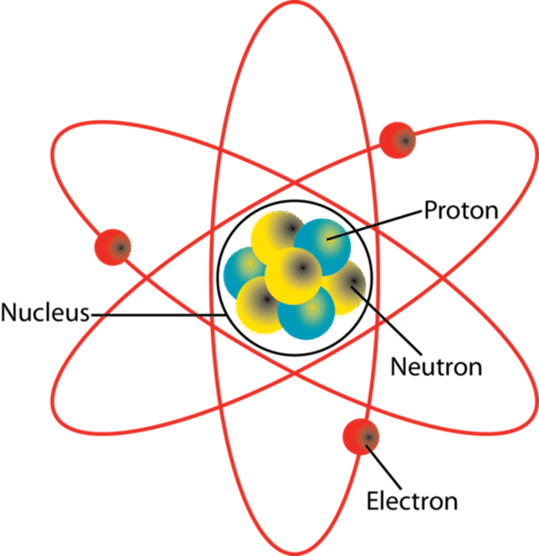
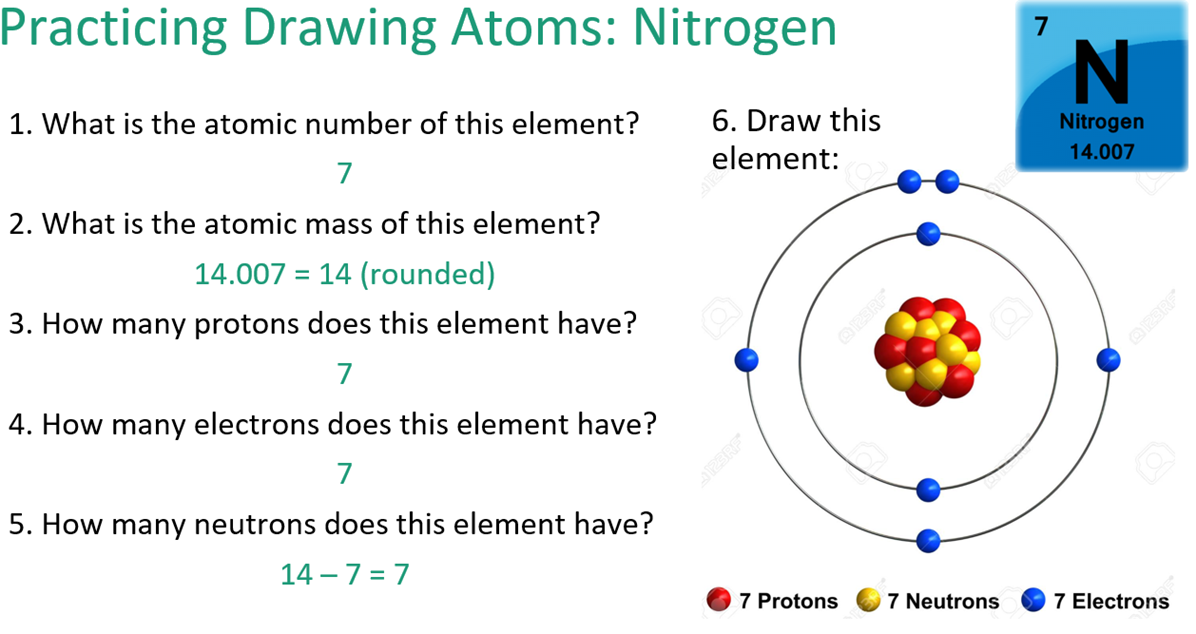
How do you calculate the Number of Protons, Neutrons and electrons in each element?
The Atomic number tells us how many protons are found in the nucleus, the number of protons in a neutral atom is always equal to the number of electrons.
Atomic number = number of protons which is then = to the number of electrons.
Atomic Mass - Protons = Neutrons
What are the Relative Charges, Location and Relative mass of each sub-atomic particle?
What is the Bohr’s Dot Diagram?
A Bohr’s Dot Diagram is a representation of how the electrons form shells around the nucleus. We will look at the first 20 element.
What are the rules for Bohr’s Dot Diagram?
Remember the following rules
1)The 1st Shell hold 2 electrons, 2nd Shell holds 8 electrons, 3rd Shell holds 8 electrons and the 4th shell holds 32 electrons.
2)The outer most shell can only hold 8 electrons as a maximum, except the 1st shell which will only hold 2 electrons.
What is an Isotope?

What is an Ion?
Ions have more or less number of electrons compared to protons of the same element.
As in the atom is either net-positive or net-negative.
How do we know which number in the Periodic Table is the Mass Number?

How do you know when Atoms are stable or unstable?
Either it is stable as it has a full outer shell OR
•When the protons and neutrons are held together strongly; the nucleus in the atom is stable.
•When the protons and neutrons aren’t held together strongly, the nucleus in the atom is unstable.
What do Unstable Isotopes do?
•Unstable isotopes decay – these are called radioactive isotopes (radioisotopes). They release energy via waves called alpha, beta and gamma radiation.
•P.S. you don’t need to know if an isotope is stable or not – you will be told!
What is radioactive half-life?
A half–life is the interval of time required for one-half of the atomic nuclei of the radioactive sample to decay.
What is a use of Radioisotopes?
Naturally occurring radioisotopes can be used to calculate the age of samples from archaeological sites and in determining the age of geological formations. This technique is called radiometric dating.
What is radiocarbon dating?
One of the most useful types of radiometric dating is radiocarbon dating. This uses the carbon-14 isotope, which has a half-life of 5700 years. Carbon is a very common element in living organisms, so the amount of carbon-14 left in a fossil or in an archaeological sample can be used to determine how long ago that organism died.
What is required for a chemical reaction?
1)Energy is required to start a chemical reaction, sometimes only a very small amount of energy (heat for example) is required.
What are some signs that a chemical reaction has occurred?
1)Colour change – The substance changes colour (for example, copper turning green or clear solutions becoming cloudy).
2)Gas production – Bubbles or fizzing appear, showing that a gas is being released.
3)Temperature change – The reaction gives off heat (exothermic) or absorbs heat (endothermic).
4)Formation of a precipitate – A solid forms when two liquids are mixed.
5)Change in odour – A new smell appears as new substances are formed.
6)Light or sound produced – Some reactions produce light (like burning magnesium) or sound (like fireworks).
What is a Reactant?
Reactants are the substances you start with in a chemical reaction. They are the chemicals that react together.
What is a product?
Products are the new substances that are formed after the reaction has taken place.
What is a Ionic Compoud?
Ionic compounds are formed when metals and non-metals react and transfer electrons from one atom to another. This creates positive and negative ions that attract each other.Example: Sodium chloride (table salt) is an ionic compound made from sodium and chlorine.
What is a Covalent compoud?
Covalent compounds are formed when two or more non-metal atoms share electrons instead of transferring them. This sharing creates a strong bond between the atoms.Example: Water (H₂O) is a covalent compound made from hydrogen and oxygen.
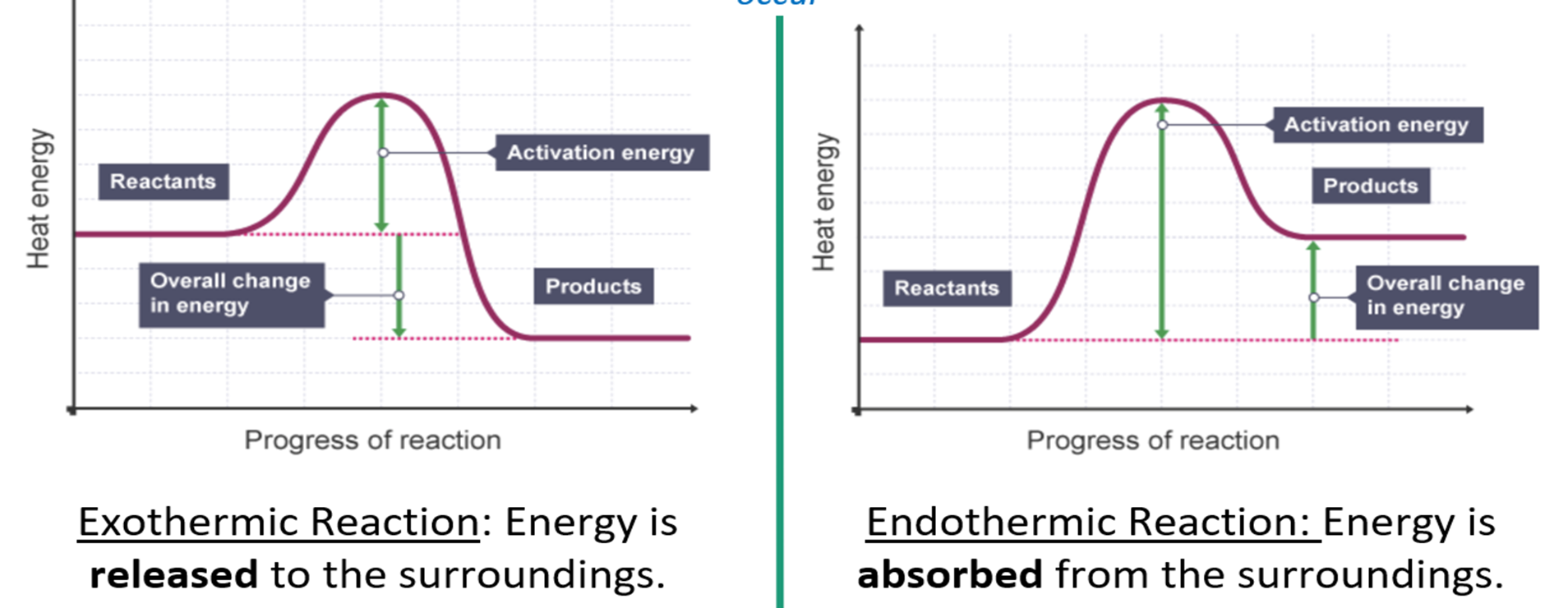
What is an Exothermic Reaction?
A chemical reaction where energy is released to the surroundings.
What is an Endothermic reaction?
A chemical reaction where energy is absorbed from the surroundings.
What is activation energy (for exothermic and endothermic reactions)?
Activation Energy: the minimum energy needed by particles when they collide for a reaction to occur.
What happens during all chemical reactions?
Energy is transferred when bonds are broken or are formed.
During all chemical reaction:
• Bonds in the reactants are broken.
• New bonds are made in the products.
Breaking bonds requires energy- endo
Making bonds releases energy- exo
What are the different types of physical states?

What is the law of the Conservation of Mass and what this means for chemical equations?
Matter cannot be created or destroyed during a chemical reaction in a closed system.
This means that chemical equations need to balanced
•The mass of the products, must also be the mass of the reactants
•The atoms (matter) that enter a chemical reactions will also be found in the products; no new atoms are formed, and they do not disappear.
What is a closed system?
A system where nothing can escape the reaction, except energy.
What is a open system?
A system where things can escape the reaction, usually gases.
What is an Acid?
Acids are:
Substances that are corrosive, that is when they are applied to another substance they can chemically react with it and degrade or break the other solid substance down.
Acids have a SOUR taste (not recommended to sample them) and
They donate Hydrogen Ions (positive charged hydrogen atoms) to a solid substance as part of the corrosive reaction.
Examples of strong acids are Sulfuric Acid in car batteries, Hydrochloric Acid in your stomach, Lemon Juice and carbonated soft drinks.
What is a base?
A base is:
a type of substance that can react with acids to neutralise them.
Bases often feel slippery to touch, and many of them can dissolve in water.
When a base does (can) dissolve in water, we call it an alkali.
Alkali (Alkaline) solutions usually have a bitter taste and a pH greater than 7.
They produce hydroxide ions (OH⁻) in water, which is what makes them alkaline.
Strong Bases (pH 10 to 14): Sodium hydroxide, calcium carbonate, industrial cleaning products
Weak Bases (pH 8 to 10): (found in) toothpaste, antacid tablets and baking powder, copper oxide and ammonia, cleaning products like soap.
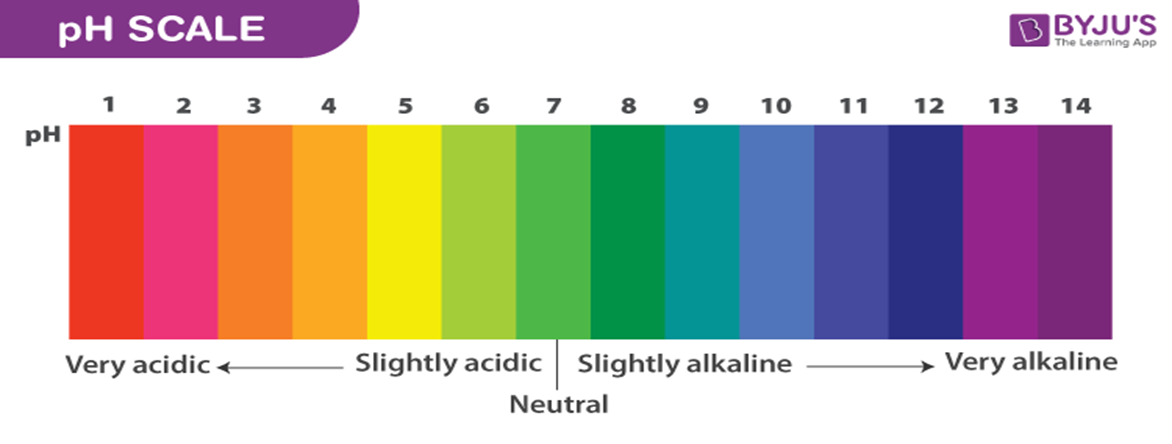
What is the pH Scale?
The pH scale is a way of measuring how acidic or basic (alkaline) a substance is. It uses numbers from 0 to 14.
How do we use the pH scale?
·pH ranges from 0 to 7 → the substance is an ACID (e.g., lemon juice, vinegar).
·pH exactly 7 → the substance is NEUTRAL (e.g., pure water).
·pH ranges from 7 to 14 → the substance is a BASE (alkali) (e.g., soap, baking soda).
What is special about the pH Scale?
•The scale is logarithmic, which means that each step is 10 times stronger than the last. For example, a substance with a pH of 3 is 10 times more acidic than one with a pH of 4.
What is an indicator (for pH)?
An indicator is a special chemical that changes colour depending on whether a substance is an acid or a base (alkali). Scientists use indicators to find out the pH of a solution.
What are 3 ways to measure pH?
Universal indicator, Litmus Paper and pH Meter.
What is Universal Indicator?
One of the most common indicators is universal indicator. This indicator can show many different colours across the pH scale, not just “acid” or “base.”
·In a strong acid (pH 1–2), it turns red.
·In a weak acid (pH 3–6), it turns orange or yellow.
·At neutral (pH 7), it is green.
·In a weak base (pH 8–10), it turns blue.
·In a strong base (pH 12–14), it turns purple.
What is Litmus Paper?
Litmus indicator solution turns red in acidic solutions and blue in alkaline solutions. It turns purple in neutral solutions. It is a piece of paper you place in the solution and it will turn colour.
What is pH Meter?
A pH meter is a scientific instrument that measures the hydrogen-ion activity in water-based solutions, indicating its acidity or alkalinity expressed as pH.
What is important to remember about the color that indicators turn in Acids and Bases?
1) Acids are substances that release hydrogen ions. So acidic solutions have more hydrogen ions than hydroxide ions. Red Indicator
2) Bases are substances that release hydroxide ions. So basic solutions have more hydroxide ions than hydrogen ions. Purple Indicator
3) Neutral substances have equal number of hydroxide ions and hydrogen ions. Green indicator.
What is a Neutralisation reaction?
A neutralisation reaction happens when an acid reacts with a base (alkali) and they cancel each other out, achieving a pH of 7.
What happens during a Neutralisation reaction?
In this reaction:
·The acid provides hydrogen ions (H⁺).
·The base provides hydroxide ions (OH⁻).
·These combine to form water (H₂O), which is neutral.
The other product formed is usually a salt (a type of compound made from the acid and base).
General word equation:
Acid + Base → Salt + Water
What are two important neutralisation reactions?
1) Hydrochloric acid + Sodium hydroxide → Sodium chloride (table salt) + Water
2) Sulfuric acid + Potassium hydroxide → Potassium sulfate + Water
What is a neutral solution?
Solutions that are neither acidic nor alkaline are neutral solution.
How is an acid usually named in a neutralisation reaction?
·ACIDS – End in ‘acid’
How is a base named in a neutralisation reaction?
·BASES – Start with a metal, and end in ‘hydroxide’, ‘oxide’ or ‘carbonate’
How is salt named in a neutralisation reaction?
·Start with a metal, and End in ‘nitrate’, ‘sulfate’ OR ‘chloride’
What is a metal carbonate?
A metal carbonate is a compound containing a metal, carbon and oxygen (see notes above).
What happens when an acid reacts strongly with Metal Carbonates?
Acids react with metal carbonates to form Salts, Carbon dioxide (gas bubbles) and water (H2O).
What are two examples of a metal carbonate reacting with an acid?
Examples
·Hydrochloric acid + Calcium carbonate → Calcium chloride + Water + Carbon dioxide
·Sulfuric acid + Sodium carbonate → Sodium sulfate + Water + Carbon dioxide
What is a Molecule?
2 or more atoms bonded together.
What is an Atom?
Particles made up of electrons, protons and neutrons.
What is a compoud?
Anything with multiple types of atoms/elements.
What is an element?
Molecules with only 1 type of atom.