Chemistry - periodic table P2
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Periodic table:
It arranges the elements in order of atomic number
elements with similar properties line up in vertical columns (called groups)
Horizontal rows across the periodic table is called periods
Most elements are solids (state) at room temperature
The only elements that are in liquid temperatures Mercury and Bromine
There is an imaginary diagonal stepped line that divides the metals from non-metals
What are metals/ non metals like?
Metals are good conductors of heat and electricity
Metals are shiny (when polished) malleable (can be hammered into shapes) and ductile (can be drawn out into wires)
Most metals are hard, dense, and have high melting points
Iron, Cobalt, Nickle are the magnetic metals
Non metals are poor conductors of heat and electricity
Non metals are usually soft and brittle
Most non - metals have low melting / boiling points so many are gases (state room temperature)
Metal oxide are alkaline non-metal oxides are acidic
How does the periodic table organise the elements?
periodic table organise elements by atomic number, chemical properties and electronic structure
What did scientists observe about about chemicals?
They observed that the chemicals have a difference in reactivity
Which gases are completely unreactive with everything?
Noble gases (group 8)
Give an example of a metal which has a vigorous reaction with water. Which group is it in?
Potassium, Sodium and Lithium (Group 1)
Why were elements ordered by atomic mass?
Protons weren’t discovered yet
In which year did Mendeleev form the first table of elements? And why was it accepted by the scientific community?
1867
He correctly predicted the properties of gallium
What was the mystery element with atomic number 68 which was discovered in 1867?
Gallium
Why was this a turning point in the world of chemistry?
It allowed you to predict properties of unknown elements
What were the remaining problems?
Noble gases weren’t discovered and the ordering of atomic weight
Electronic structure
Elements in group 1 have 1 electron in their outer shell, in group 2 they have 2 electron in their outer shell and so on.
Elements in the same group of the periodic table react in a similar way because their atoms have the same number of electrons in the highest occupied energy level
why do elements react?
Because of their electronic structure
what does periodic mean in the term periodic table?
There is an order of strings repeating
(properties of elements repeat in a regular pattern when they are arranged in order of increasing atomic number)
what is a group what is a period
vertical groups
horizontal periods
How do the electronic structures of the atoms of a metallic element change when they react?
When metallic elements react they lose 1 or more electrons form their outer shell to form positive ions
Explain why the noble gases are so unreactive (group 8)
They already have full outer electron shells
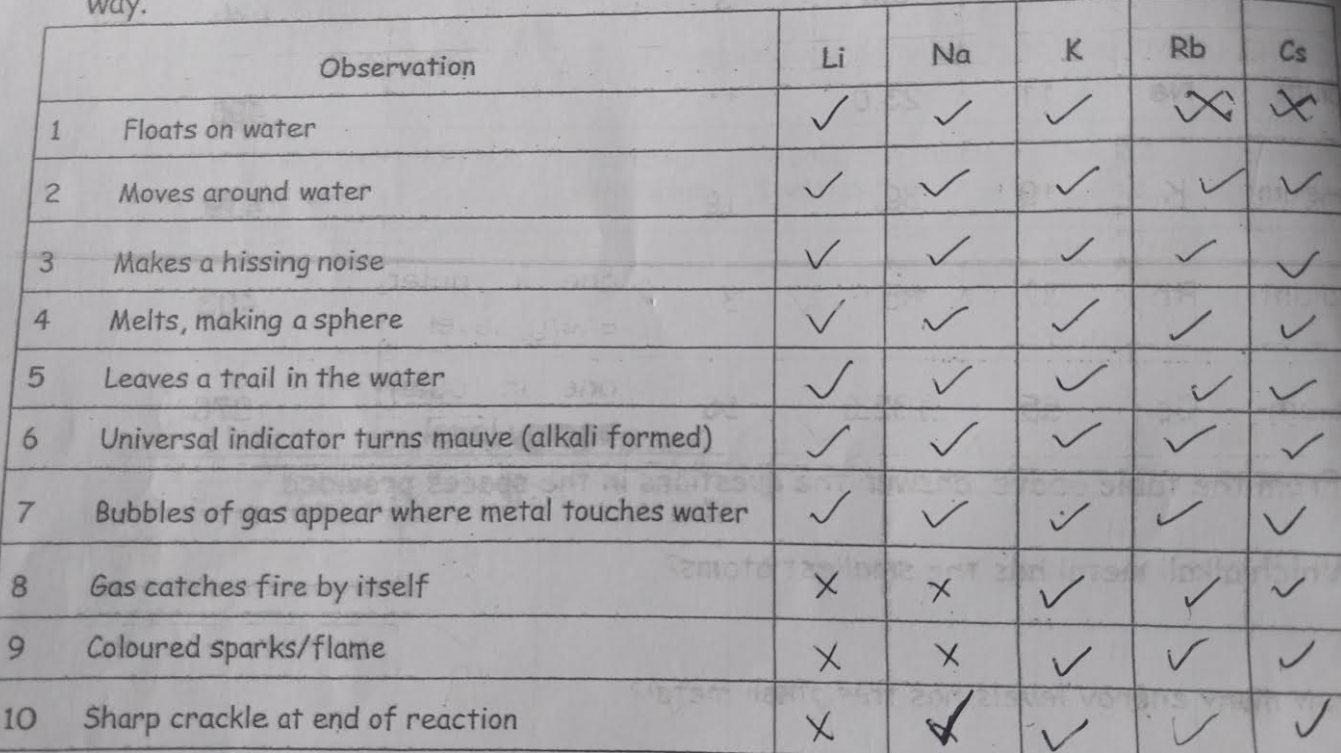
Experiment 1 - appearance
Which group has the shiny appearance?
Metals - silver - shiny, copper - bronze, Magnesium - shiny
Which group has a dull appearance?
Non - metals - Hydrogen, oxygen, chlorine - clear sulfur, carbon - yellow
Experiment 2 - conduction of heat
This is strips of different metals: brass copper iron and aluminium dipped into hot water and must state which one heats the quickest in order
Copper, Aluminium, Brass, Steel
Experiment 3 - Conduction of electricity
Simple electrical circuit. Make sure bulb lights up
which ones conduct electricity?
copper zinc steel
which ones don’t conduct electricity?
straw wood
Experiment 4 - Sound
Use a small hammer and ht the 2 materials (cans)
Which ones are sonorous?
metals are sonorous
Experiment - Malleable or brittle
Provided some metals and non metals
which ones can you bend the material without breaking?
Copper, Tin, Zinc, Straw - malleable
Wood, Pasta - Brittle
Experiment 6 - Density
density = mass / volume
based in the different blocks work out the mass and volume and divide them to get the density
Experiment 7 - Melting points
Do metals or non metals have the higher melting and boiling points?
Metals generally have significantly higher melting and boiling points than non - metals
Alkali metals
element colour hard? does it tarnish? Electrical conductivity m.p. b.p. density
lithium no/ silver no yes yes 181 1347 0.53
sodium no/ silver no yes yes 98 883 0.97
potassium no/ silver no yes yes 64 774 0.86
rubidium no/ silver no yes yes 39 686 1.53
caesium no/ silver no yes yes 28 671 1.87
What colour are alkali metals?
Silver
Are alkali metals hard or soft?
soft
what happens as you descend the group ?
As you descend the group the melting / boiling points of the metals decrease
What are the densities of alkali metals like?
The alkali densities of lithium sodium and potassium are all below one, therefore they float on water
Reaction of alkali metals
As you go down the group in the periodic table the number of complete energy shells increases. This means the atoms become larger. In group 1 the atoms want to lose an electron when they react. Larger atoms lose electrons more easily than smaller ones because
outermost electron further from nucleus
inner shells of electron shields the outermost electron from nuclear charge
greater nuclear charge of larger atoms have less electrostatic attraction towards outer most electron due to the size and shielding
words / symbols
words: caesium + water → caesium hydroxide + hydrogen
symbols: 2Cs + 2H2O → 2CsOH + H2
words: rubidium + oxygen → rubidium oxide
symbols: 4Rb + O2 → 2Rb2O
words: sodium + chlorine → sodium chloride
2Na + Cl2 → 2NaCl
Forming Sodium Ion
There is an electrostatic attraction between:
the positively charged nucleus (protons)
and the negatively charged electrons.
When sodium reacts, it loses its outermost electron.
The outer electron gains enough energy to leave the atom.
Electrons that are further from the nucleus are easier to remove.
Inner electron shells protect (shield) the outer electron from the nucleus’ pull.
After losing one electron, sodium becomes a sodium ion (Na⁺) with electron arrangement 2,8.
Properties of alkali metals
lithium - deep red flame
sodium - deep yellow flame
potassium - lilac flame
rubidium - catches fire as soon as exposed to air and burns fiercly
caesium - melts in warm room can explode on exposure to air
Halogens/ why do they become less reactive as you go down the group?
Become less reactive as you go down the group - As its easier to gain electrons because numbers are positive and with less shells they are closer making it easier to attract. Atomic radius increases or more energy levels / shells. Attraction between nucleus and outer electron reduces due to inc. in shielding.
Table:
halogens
Fluorine
chlorine
bromine
Iodine
What do diatomic molecules mean? And name the 7 diatomic molecules
they travel in 2 pairs
Hydrogen, oxygen fluorine nitrogen bromine chloride iodine
(H,O,F,Br,I,N,Cl)
Experiment (REACTIONS) - S = Solution W = Water
Chlroine S + Potassium Chlorine S = X
Bromine W + Potassium Chlorine S = X
Iodine S + Potassium Chlorine S = X
Chlorine S + Potassium Bromide S = Brown/ orange soln
Bromine W + Potassium Bromide S = X
Iodine S + Potassium Bromide S = X
Chlorine S + Potassium Iodine S = Brown/ orange soln
Bromine W + Potassium Iodine S = Brown/ orange soln
Iodine S + Potassium Iodine S = X
words / symbols
Potassium Iodine + Chlorine (gas)→ Potassium chloride + iodine
2Kl + Cl2 → 2KCl + I2
Potassium iodine + Bromine → Potassium bromide + iodine
2KI + Br2 → 2KBr + I2
Potassium bromide + Chlorine → Potassium chloride + Bromine
2KBr + Cl2 → 2KCl + Br2
Uses of noble gases
Helium - It is used for balloons and airships used for medical applications like MRI, respiratory therapy
Neon - Signage and lighting, lasers, electrical equipment
Argon - welding, manufacturing industry
Krypton - Fluoride lasers,- for medical issues, Insulation and other applications, energy efficient windows