Bio 161 W3 -Lipids & Membranes
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
What is the key chemical feature of lipids?
Mostly nonpolar hydrocarbon chains (C–H bonds)
How do lipids interact with water?
They are hydrophobic and avoid water (cluster together) (hydrophobic effect)
What type of interactions hold lipids together?
Hydrophobic interactions + van der Waals forces
—> No strong covalent bonding between lipid molecules in membranes
Lipids come in many shapes/sizes but backbone is….
composed of carbon & hydrogen
Why are lipids NOT considered true polymers?
not built from long chains of repeating, similar monomer subunits
Instead composed of a few distinct parts, like glycerol & fatty acids
Which of the four biological macromolecules are considered "true polymers"?
Polypeptides (proteins), nucleic acids, and polysaccharides
(Polymer) Polypeptides
(Monomer) Amino Acids
(Polymer) Nucleic Acids
(Monomer) Nucleotides
(Polymer) Carbohydrates/Polysaccharides
(Monomer) Monosaccharides
Which elements are always found in polypeptides and nucleic acids, but are often missing in many common lipids like triglycerides?
Nitrogen (found in both) and Phosphorus (found in nucleic acids)
Important lipids found in the cell
Triglycerides
Fats
Phospholipids
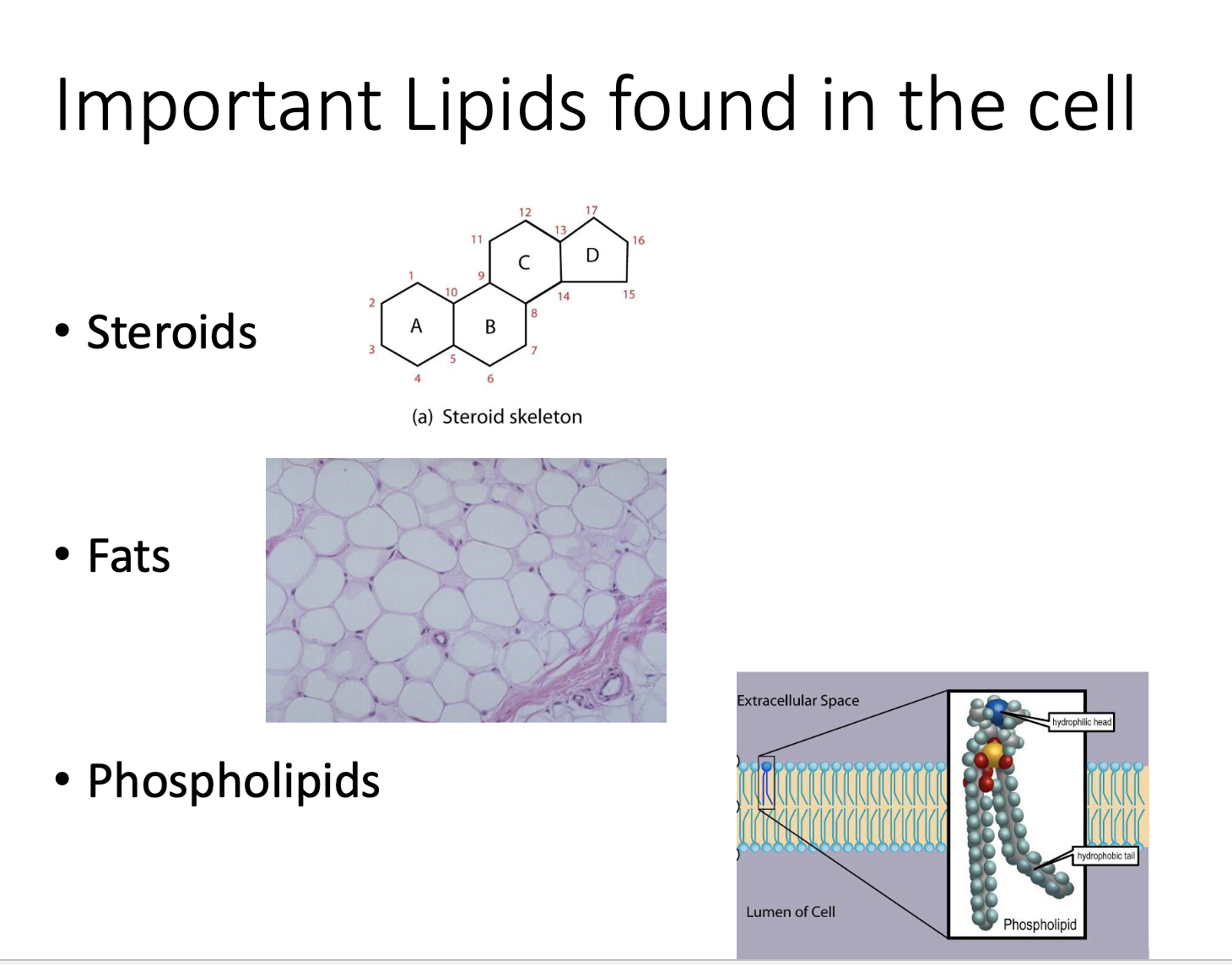
Lipid Type: STERIODS
Lipids with 4 fused carbon rings
small modifications along backbone
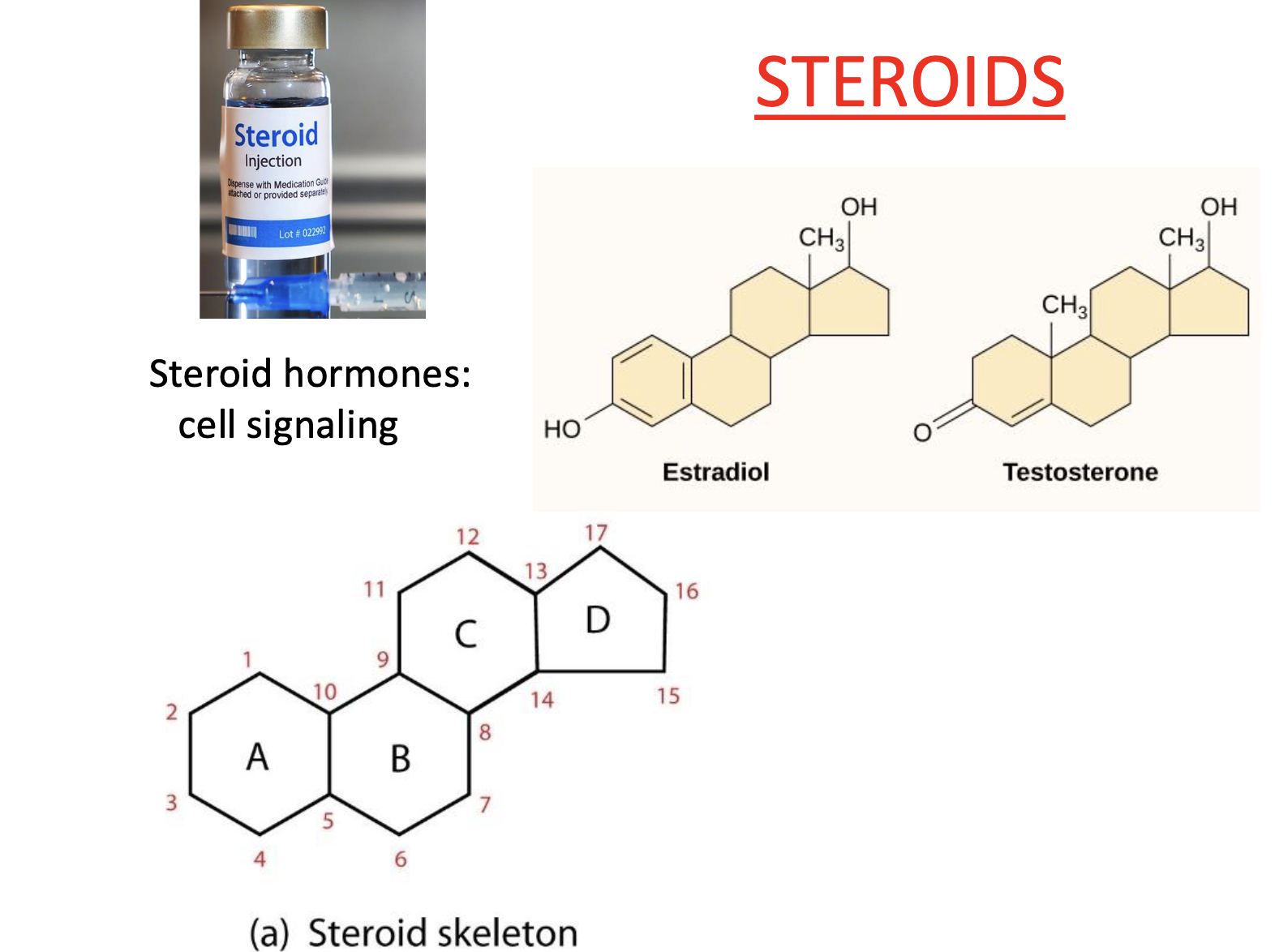
Steroid hormones
cell signaling
—> many hormones are steroids, BUT not all steroids are hormones & not all hormones are steroids
Type of Steroid: Cholesterol
unique to eukaryotes
still has that same 4 ring carbon structure
Cholesterol can be carried in blood…
inside of larger particles
WE NEED CHOLESTEROL!! —> vital for life b/c of fluidity & structure it allows membranes
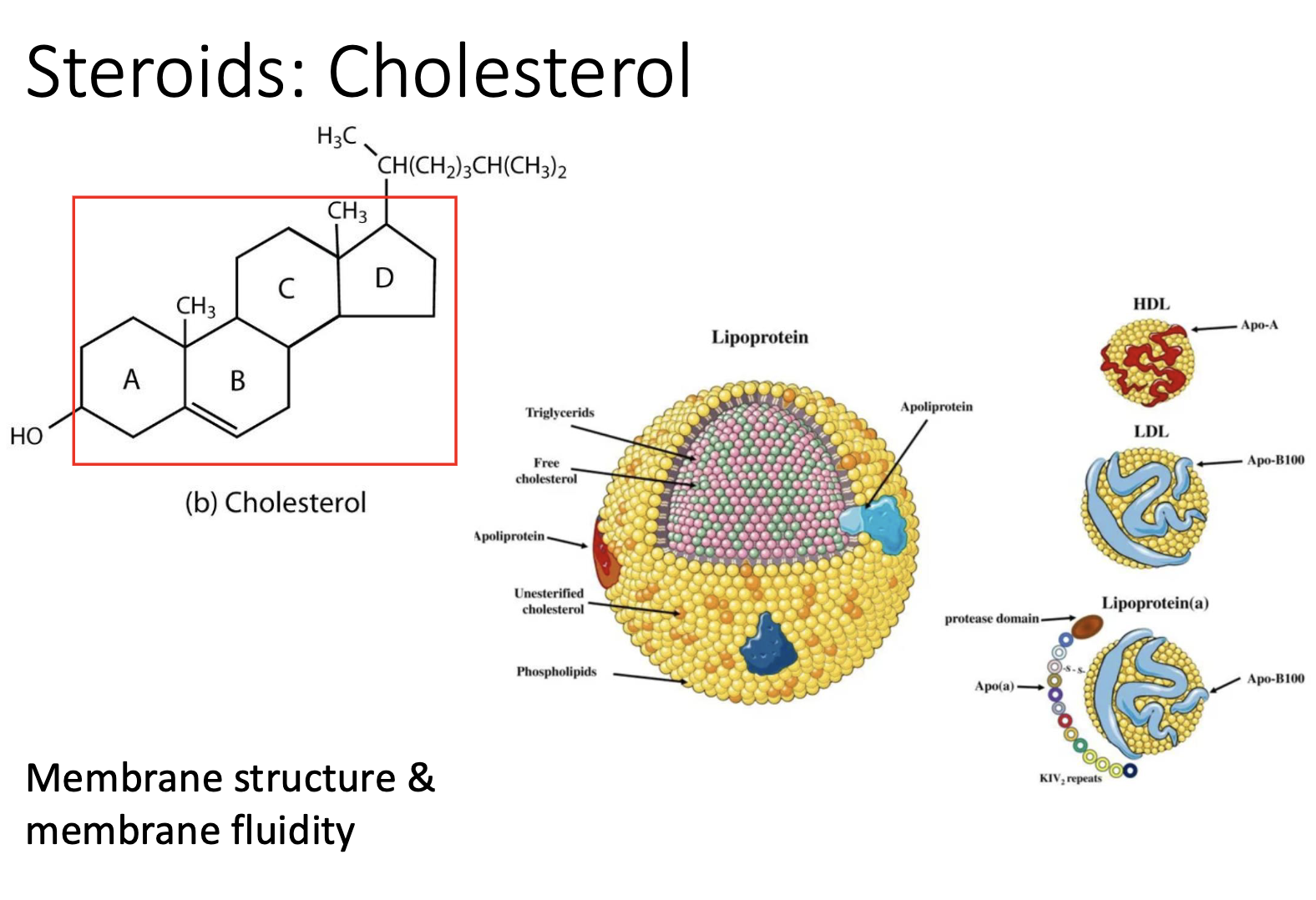
Why cholesterol vital for life?
It allows membranes fluidity & membrane structure
The Fatty acids….
Some of the most important lipids that give rise to life!
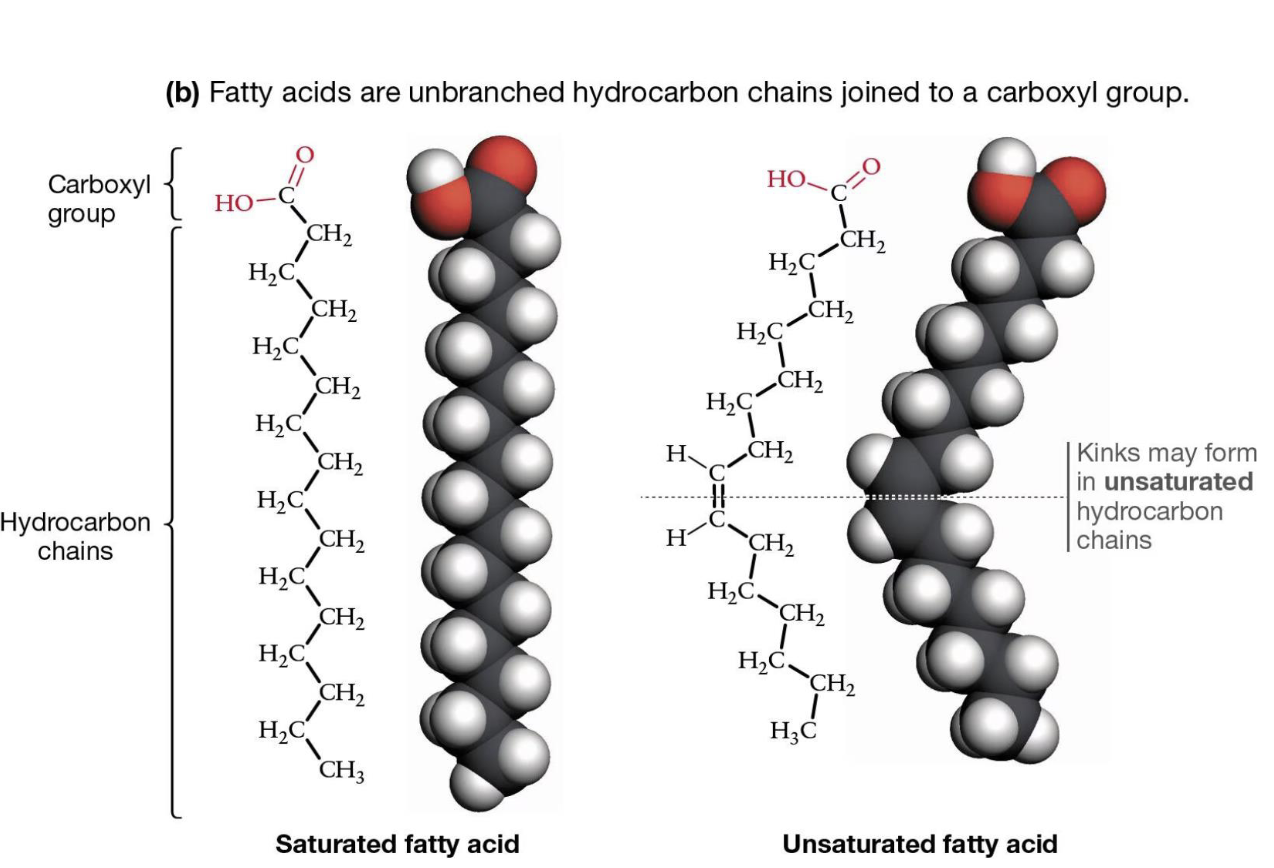
Fatty acids are
unbranched hydrocarbon chains joined to a carboxyl group
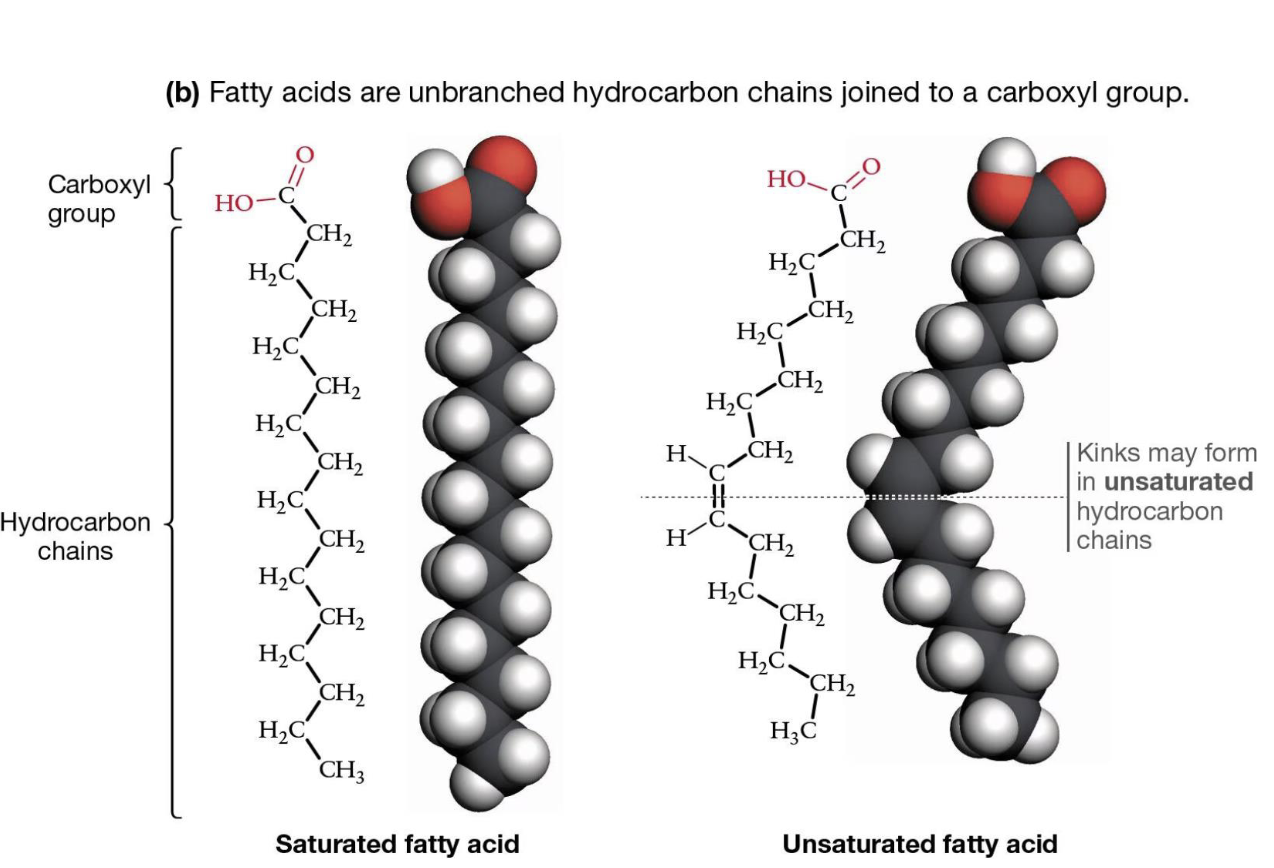
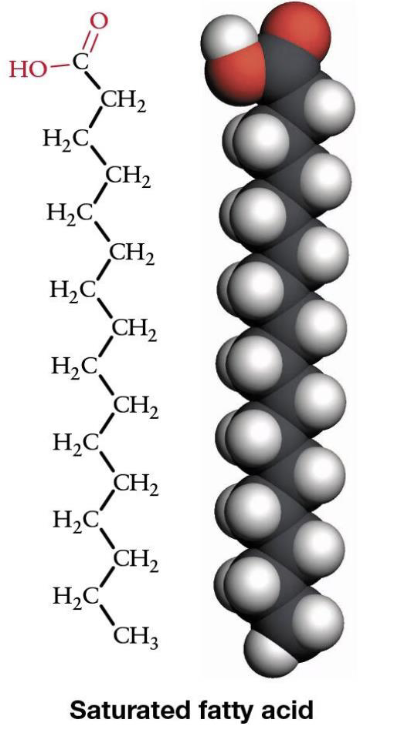
What is a saturated fatty acid?
No double bonds, straight chains, tightly packed
—> carbons FULLY bound to hydrogens
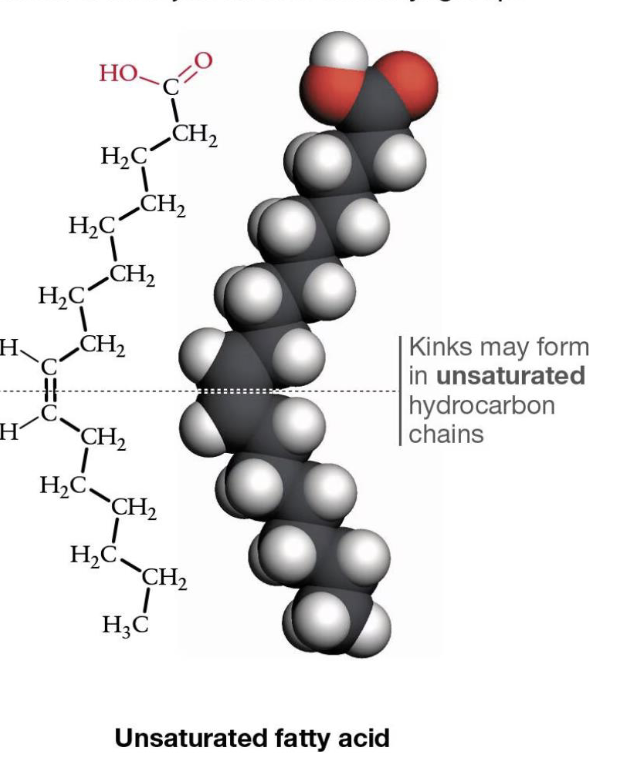
What is an unsaturated fatty acid?
Has double bonds (usually cis), causes kinks
—> one or more double carbon bonds, not fully saturated w/hydrogens
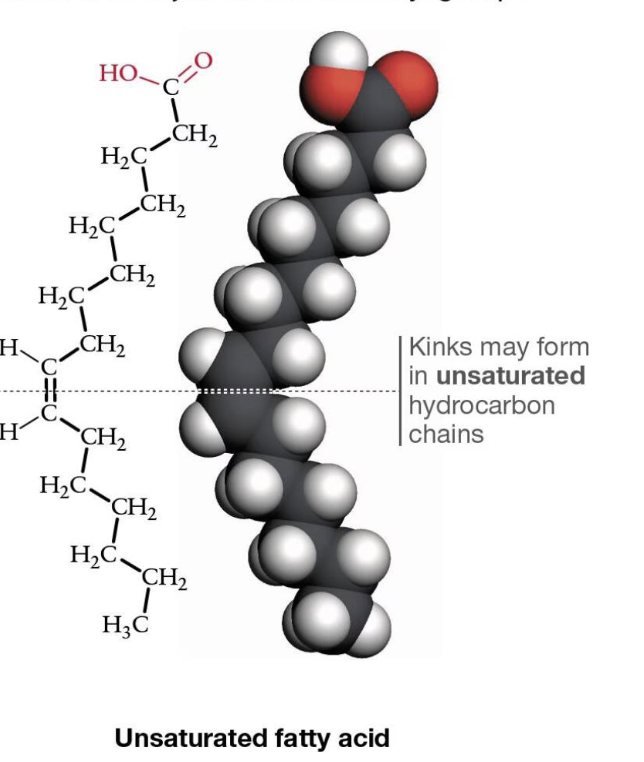
What do double carbon bonds cause in unsaturated fatty acids?
kinks!
Saturation level of Fatty Acids…
determines fluidity
Effect of unsaturation on membranes?
Increases membrane fluidity
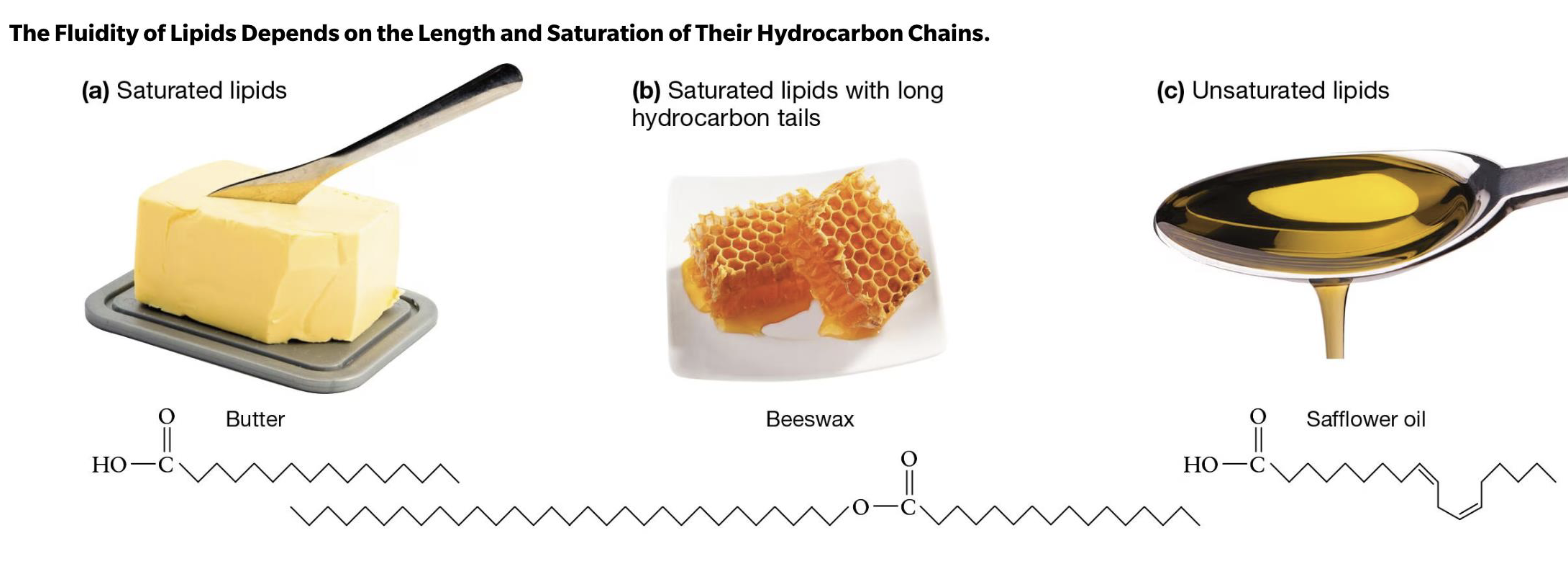
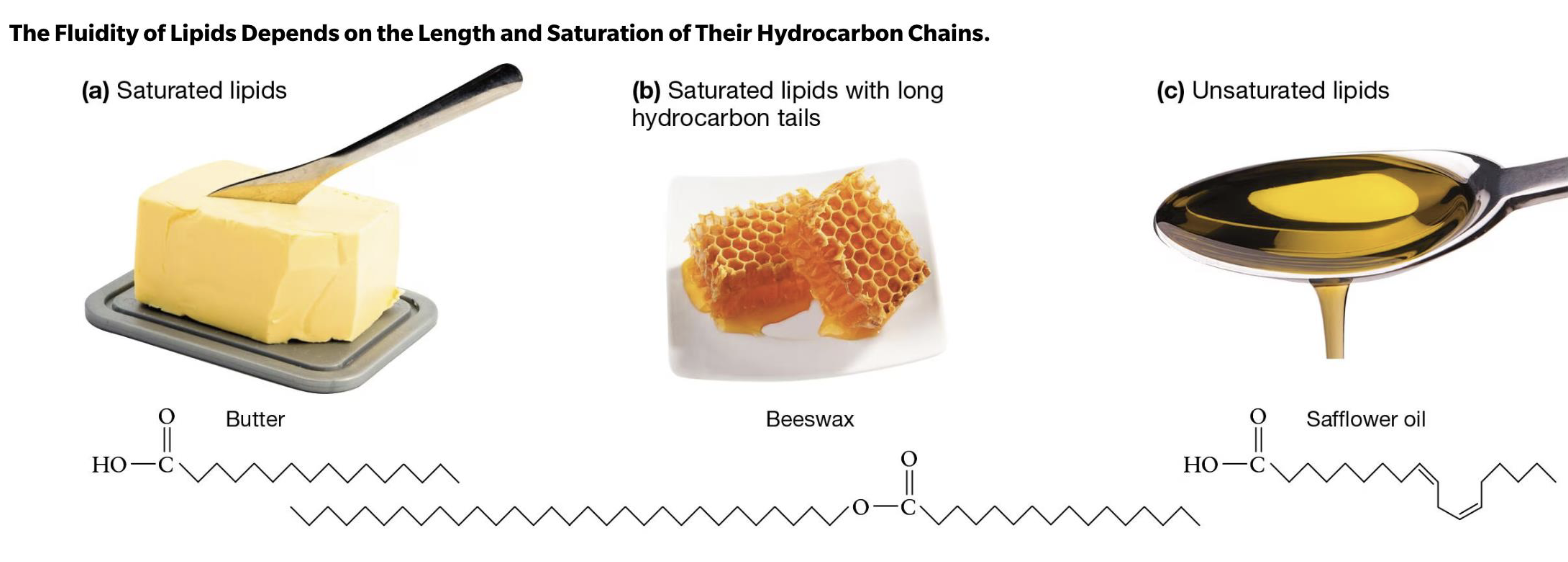
Saturated Lipids…(butter, beeswax)
no 3D kinks, packed very tightly
compact, less fluid
form solids @room temp
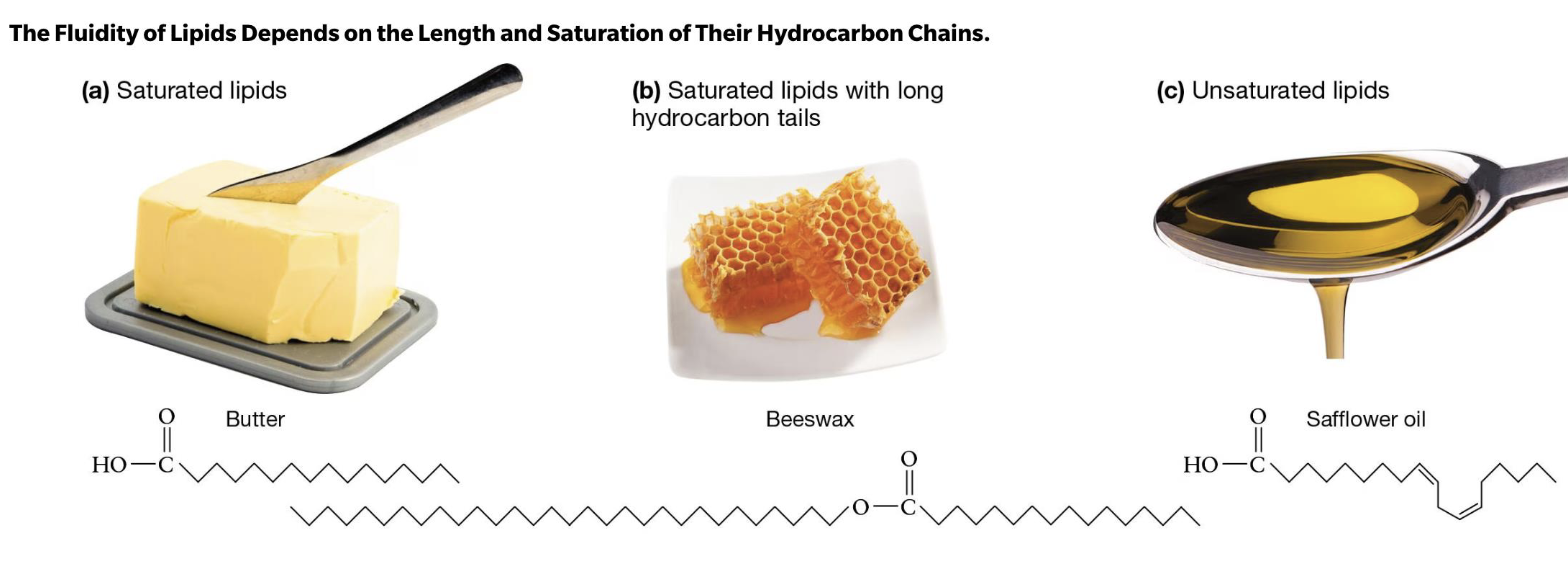
Unsaturated lipids (oil!)
liquid @ room temp bc of kinks! (in backbone)
less compact, more fluidity
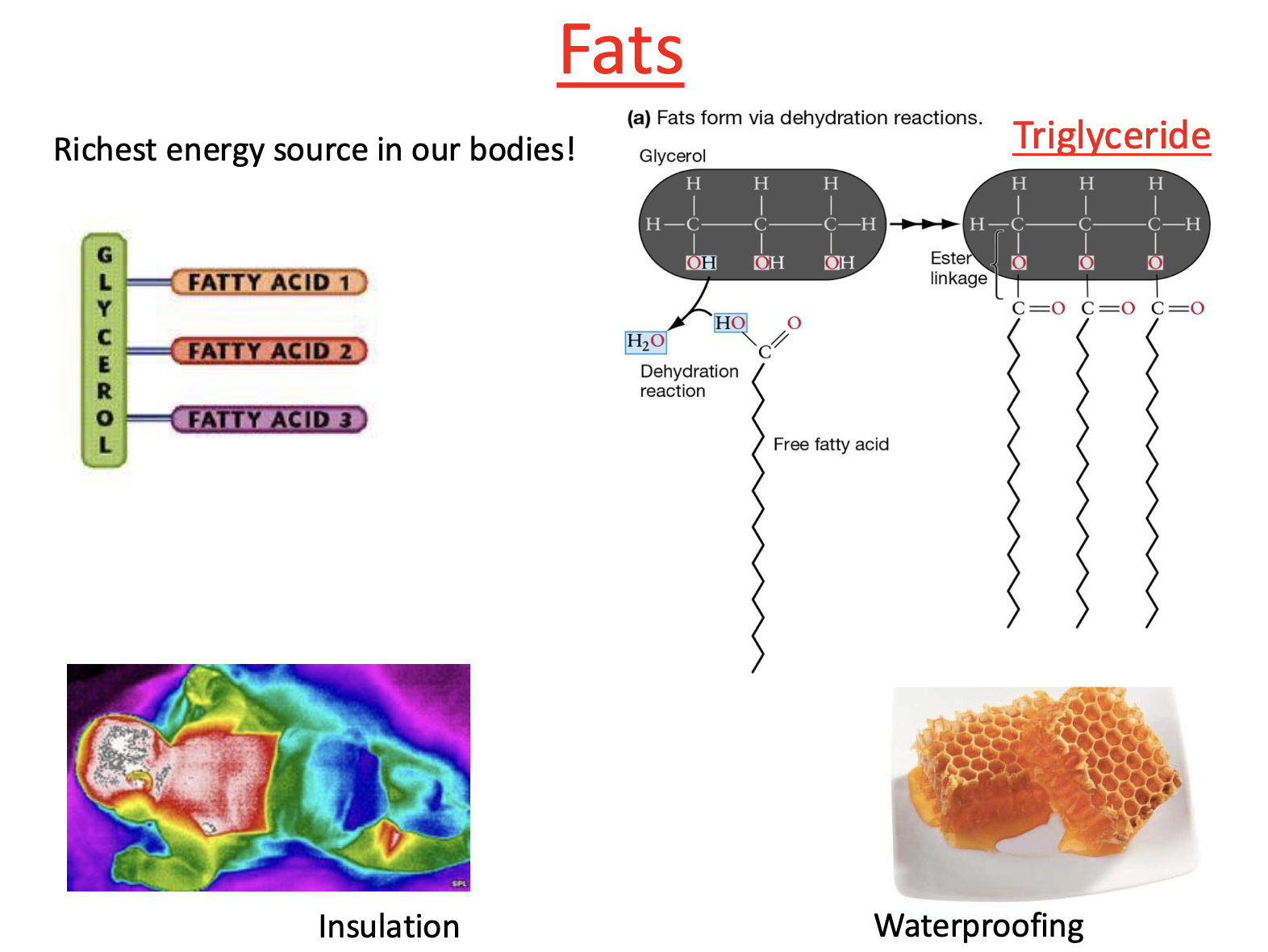
Type of Lipids: Fats
richest energy source in our bodies!
Insulation: highest amount of fat near major organs b/c vital organs require a lot of energy
Waterproofing: honeycombs made up of triglycerides —> structure waterproofs
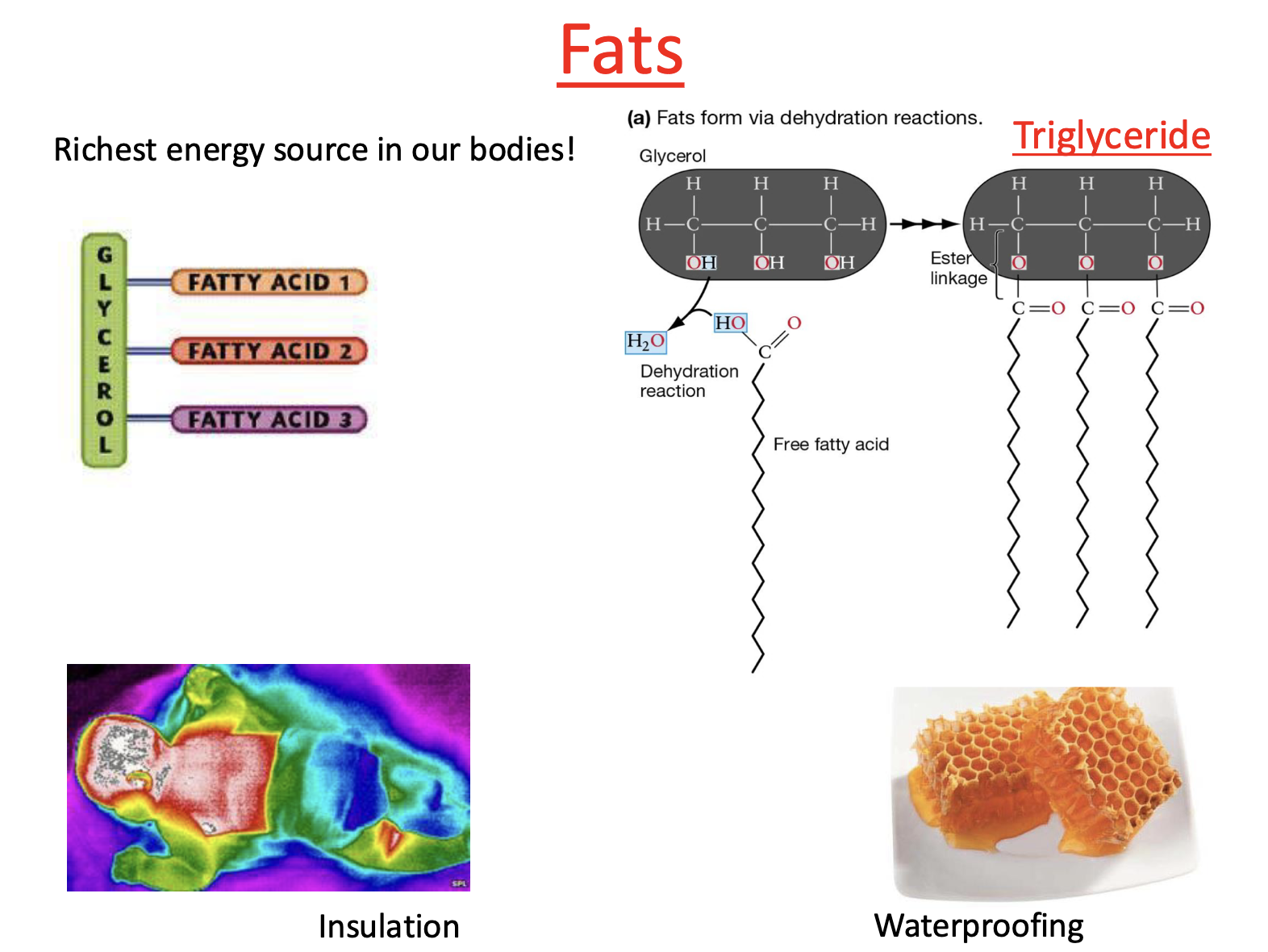
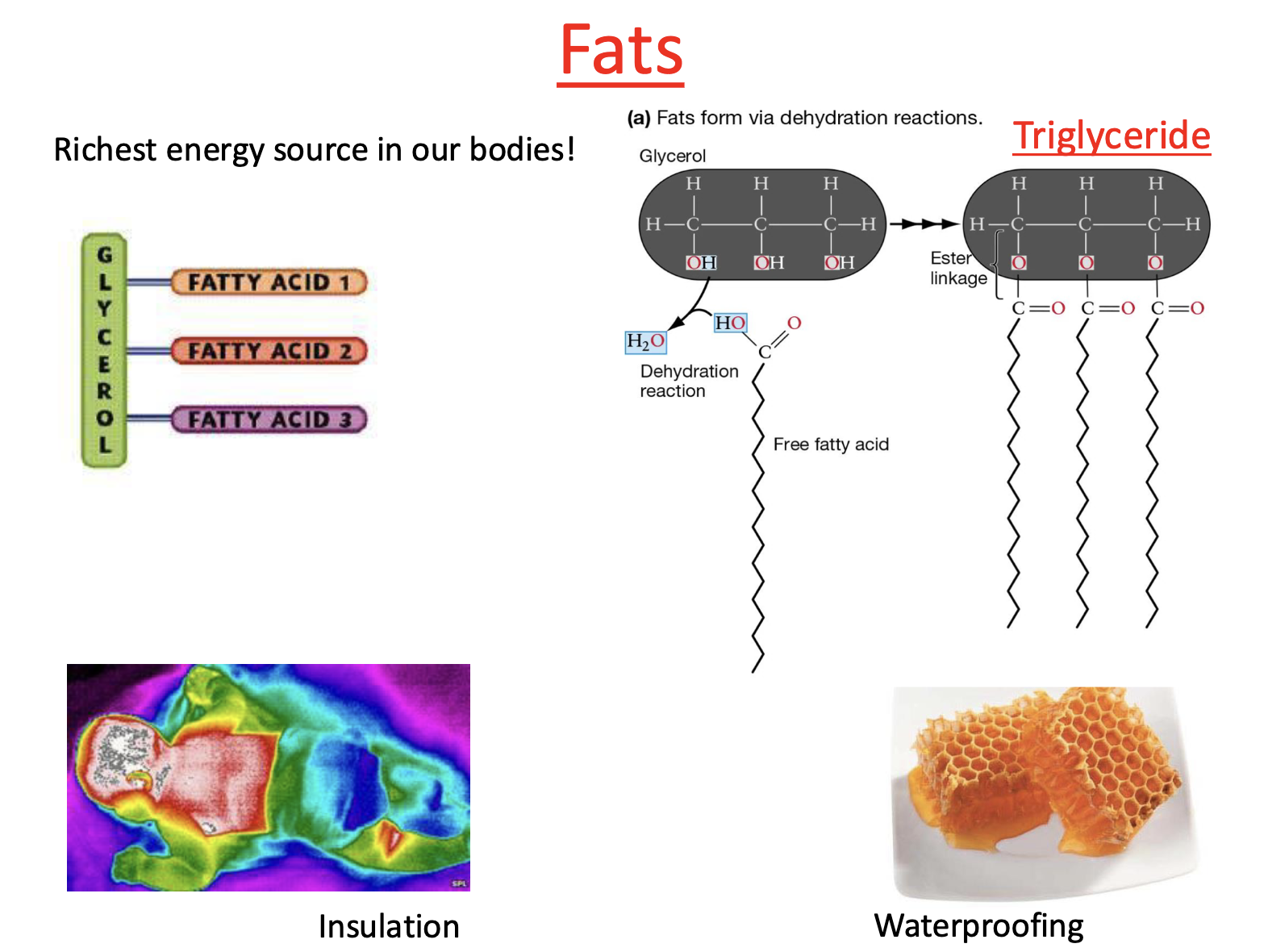
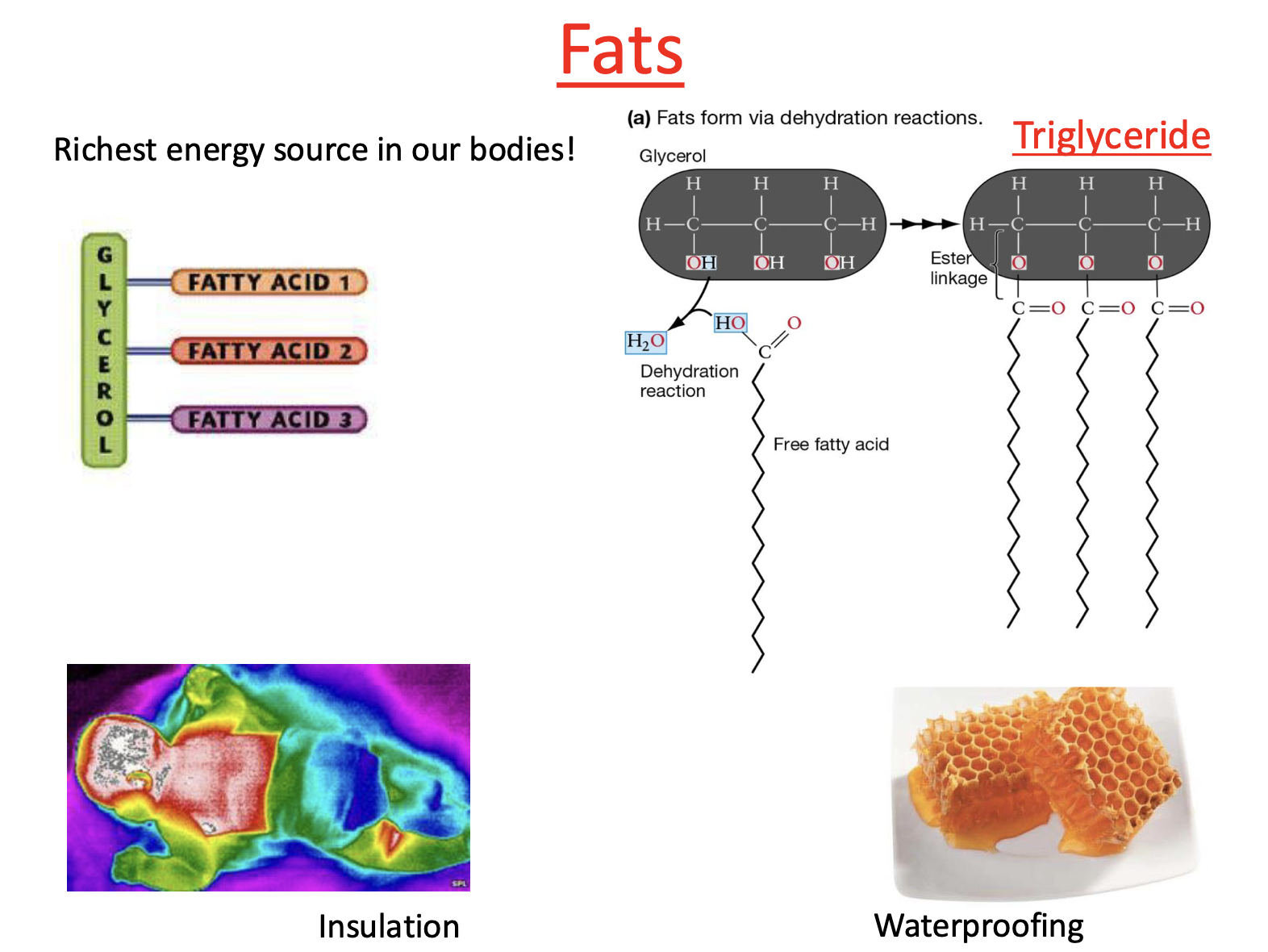
Type of Fat: Triglyceride
Triglycerides form through condensation reaction
Free fatty acids attach to glycerol
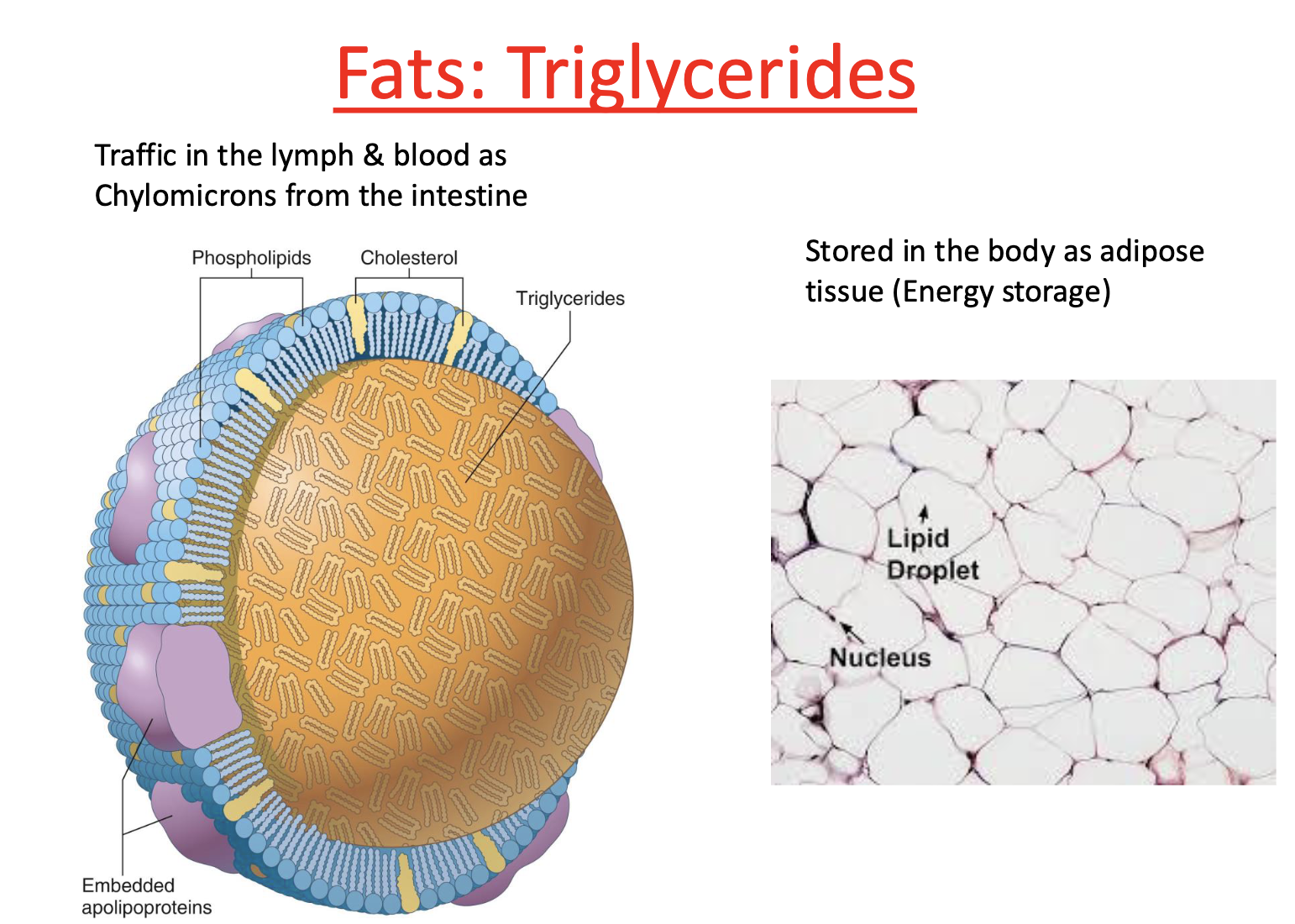
What are triglycerides used for?
Energy storage
stored in body as adipose tissue
What are triglycerides made up of?
Glycerol
3 free fatty acids
What is glycerol?
3 carbon structure
Triglycerides are transported by
molecules covered in protein & phospholipids
triglycerides on inside! (orange)
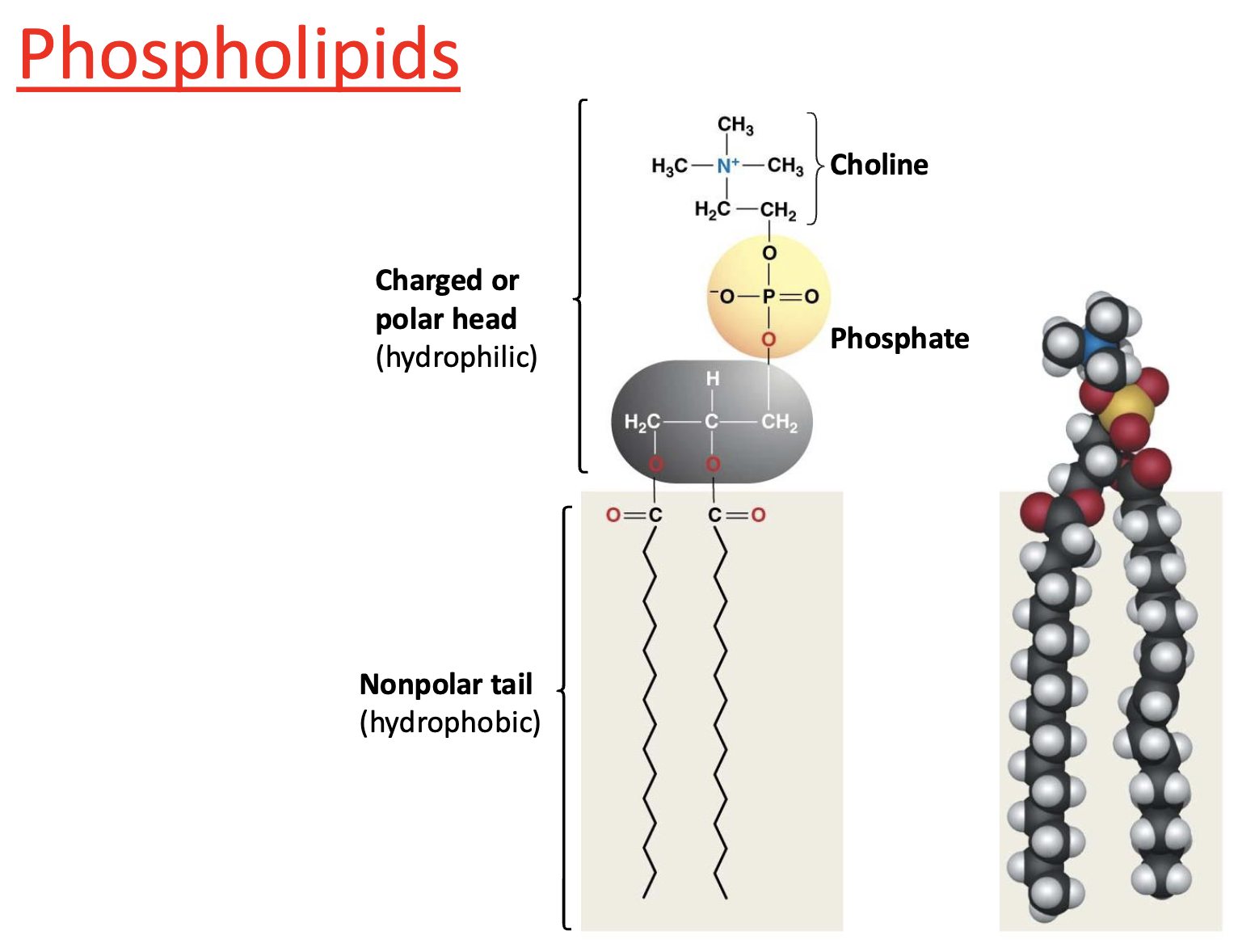
Type of Lipid: Phospholipids
are amphipathic
charged/polar head (hydrophilic)
nonpolar tail (hydrophobic)
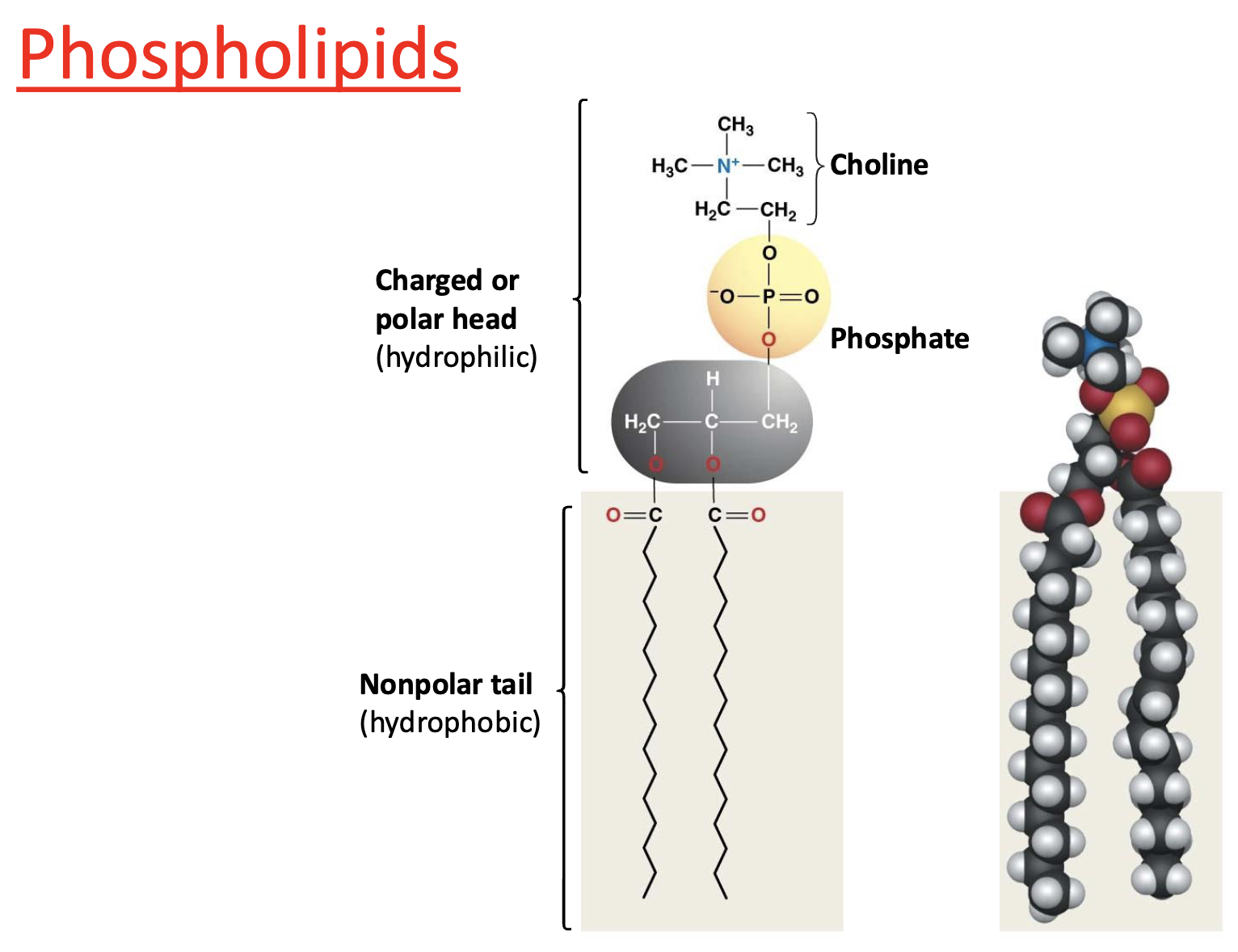
What are phospholipids made of?
Glycerol + 2 fatty acids + phosphate group
Why are phospholipids important?
They form cell membranes
more polarity, more ability to interact w/aqueous environment
What does amphipathic mean?
Has both hydrophilic and hydrophobic regions
Why are phospholipids amphipathic?
Polar head + nonpolar tails
Lipid Micelles
Most simple arrangement of phospholipids
Hydrophilic heads interact w/water
hydrophobic tails interact w/one another
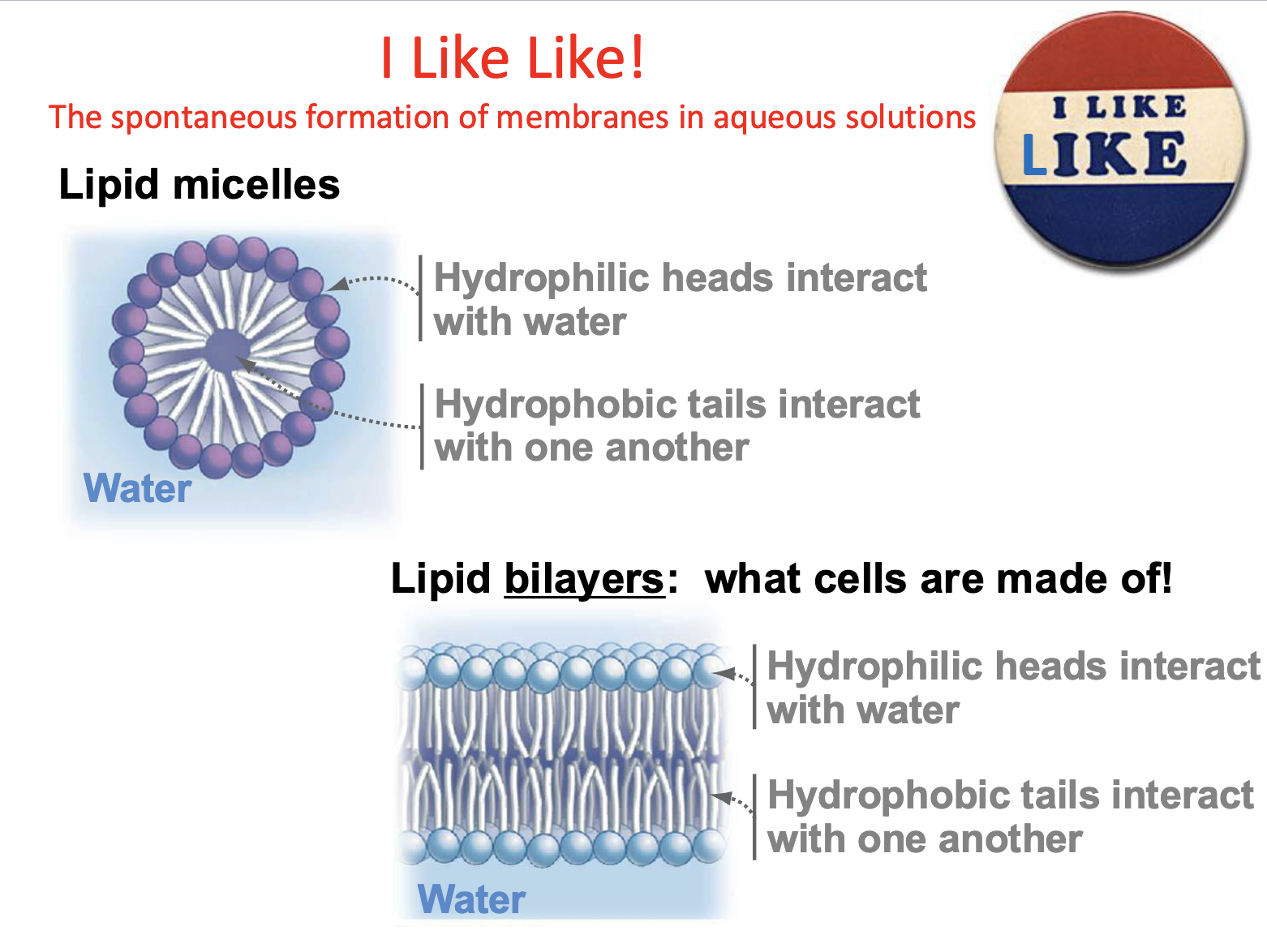
Formation of membranes in aqueous solution are
spontaneous
Lipid Bilayers
what cells are made of!
2 layers form inverted to each other spontaneously
can compartmentalize
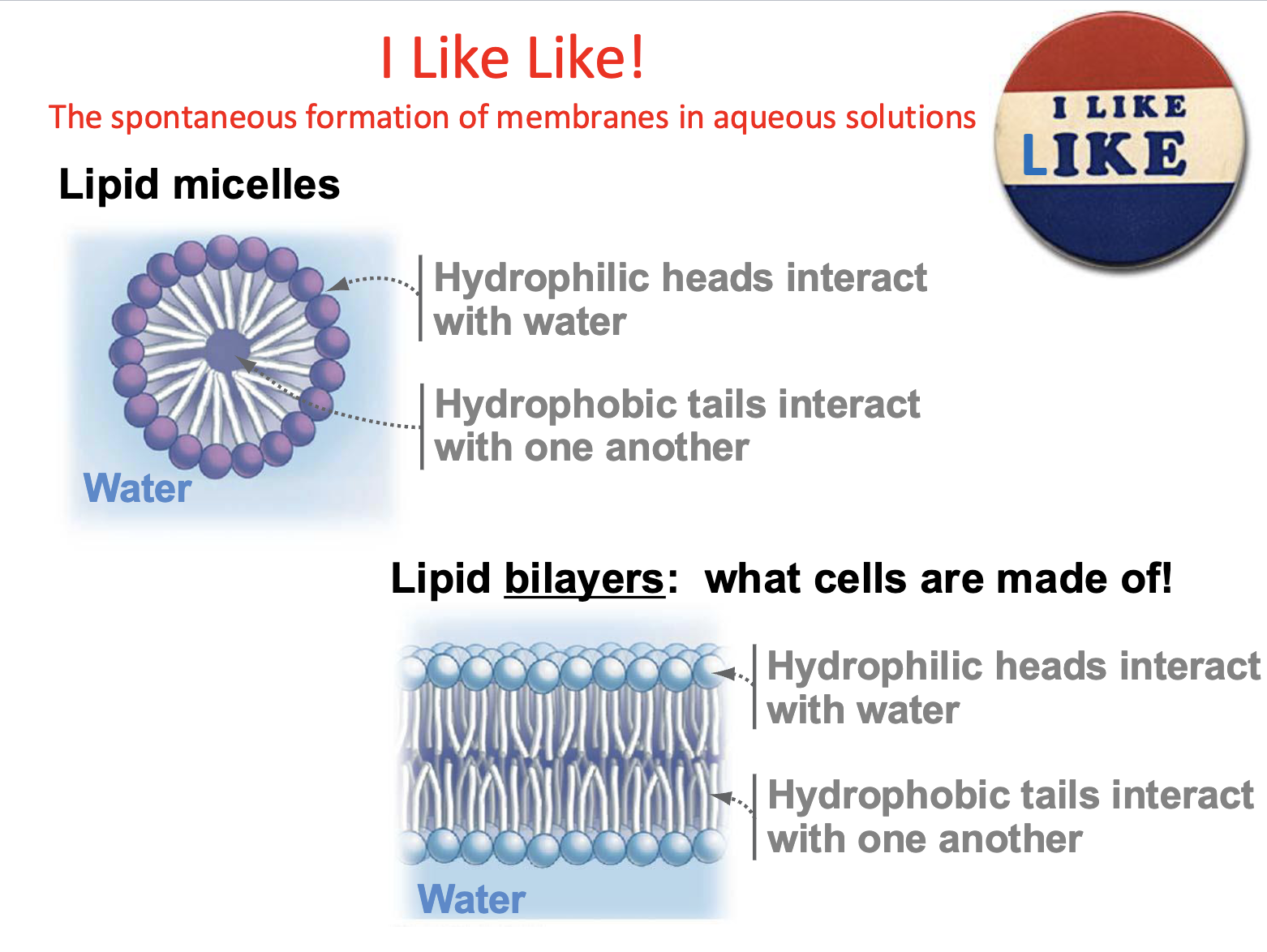
Why do lipid bilayers form?
Hydrophobic effect (tails avoid water)
Phospholipid bilayer membrane allow…
for life as we know it!
can compartmentalize cell’s inside/outside
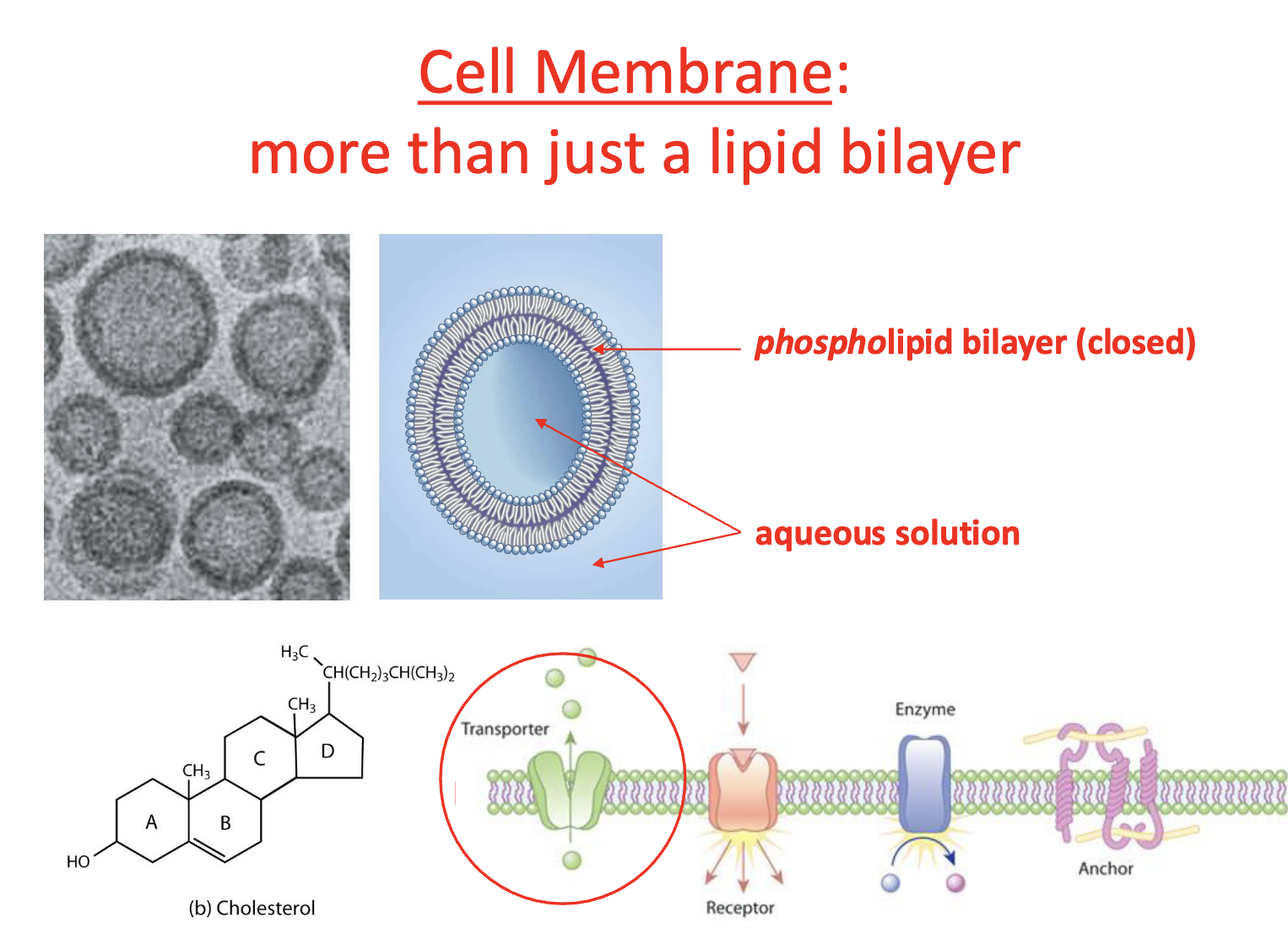
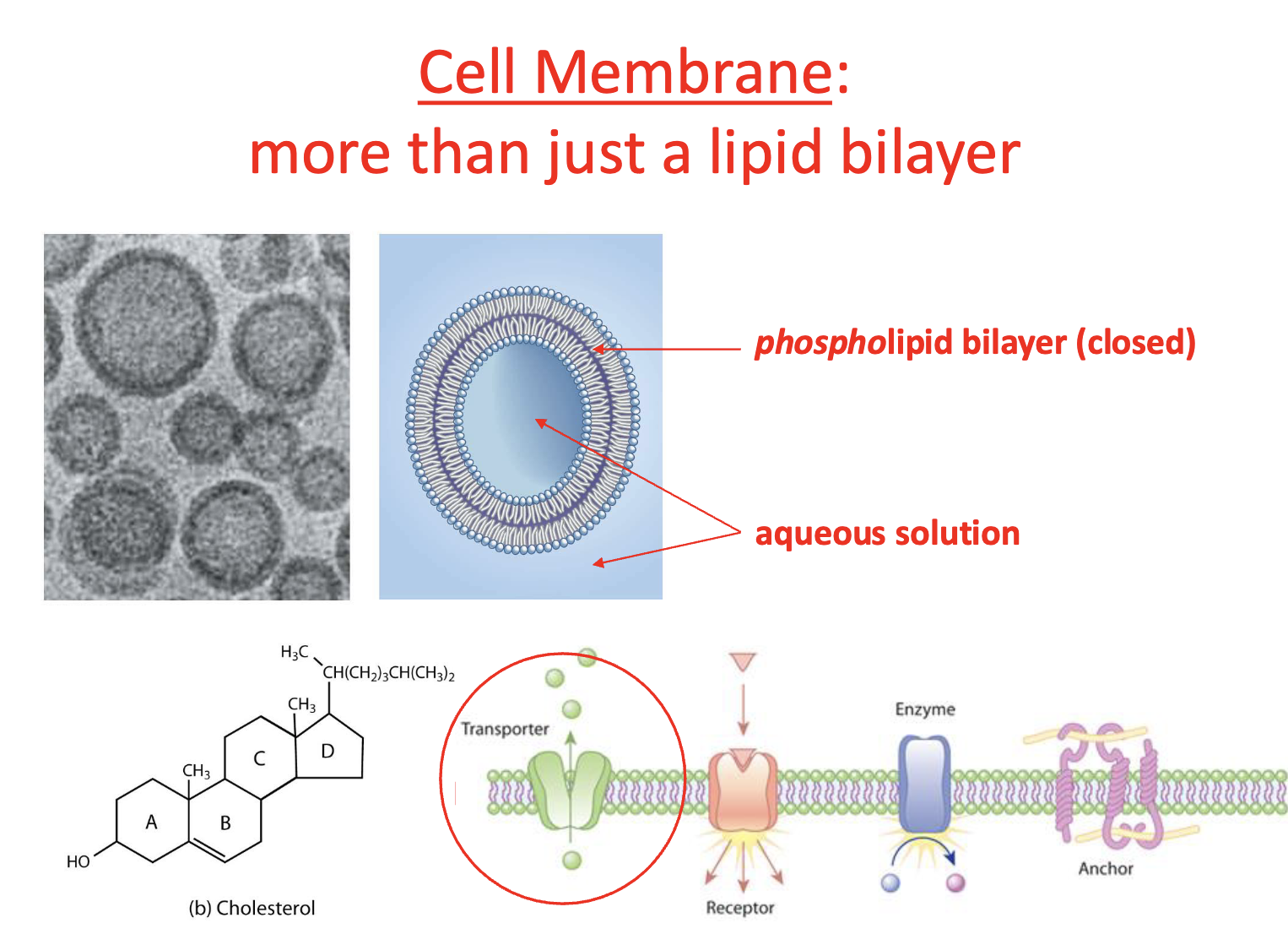
In the membrane proteins are
conduits in the membrane
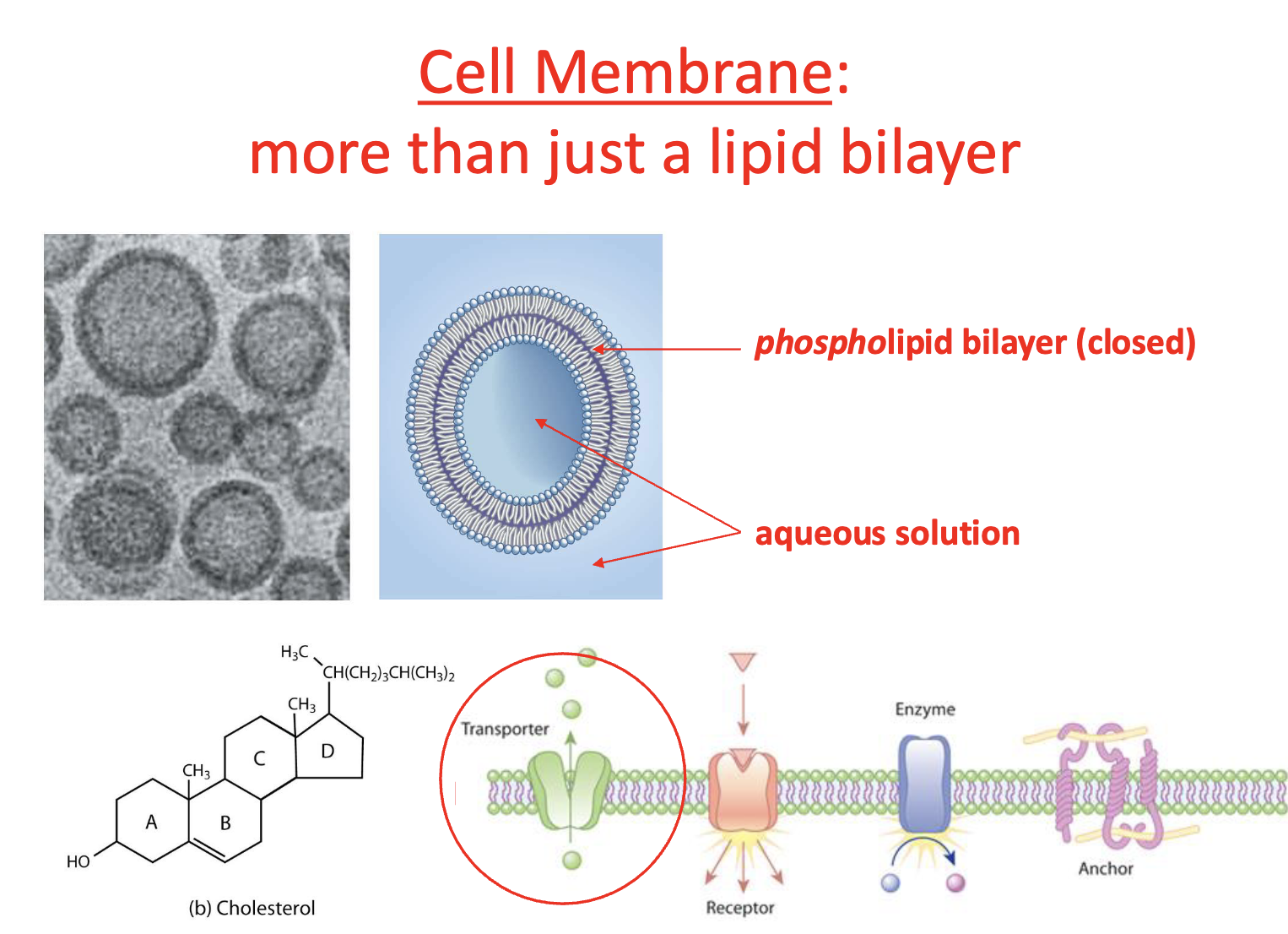
Cholesterol & saturation of hydrocarbons of fatty acids (membrane wise)
influence attachment of proteins + fluidity
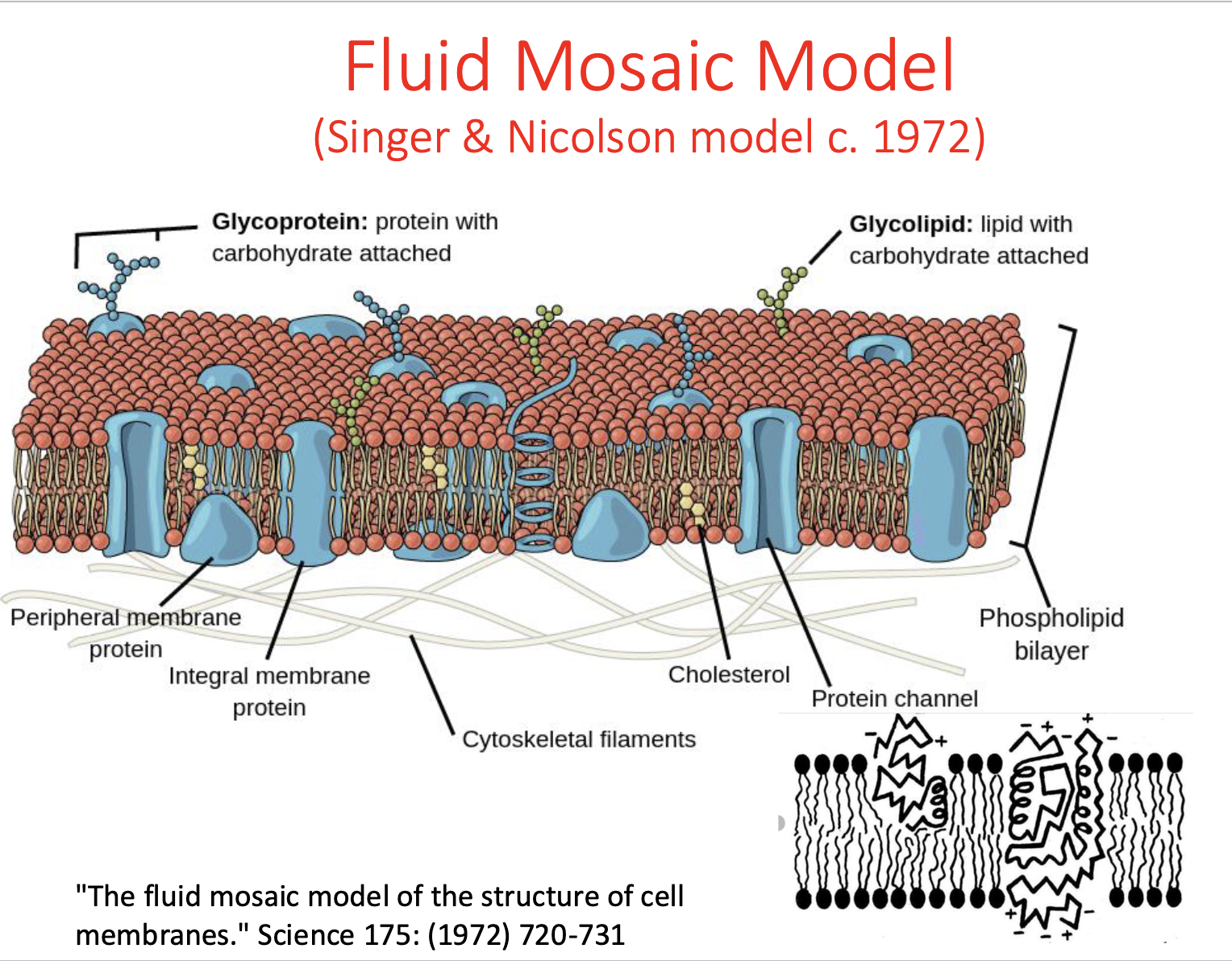
What is the Fluid Mosaic Model?
proteins & cholesterol embedded in membrane
good model, still stands today BUT doesn’t specify micro domains present in membranes
Lipid Raft Model
membrane micro-domain
clustering of cholesterol & specific transmembrane proteins
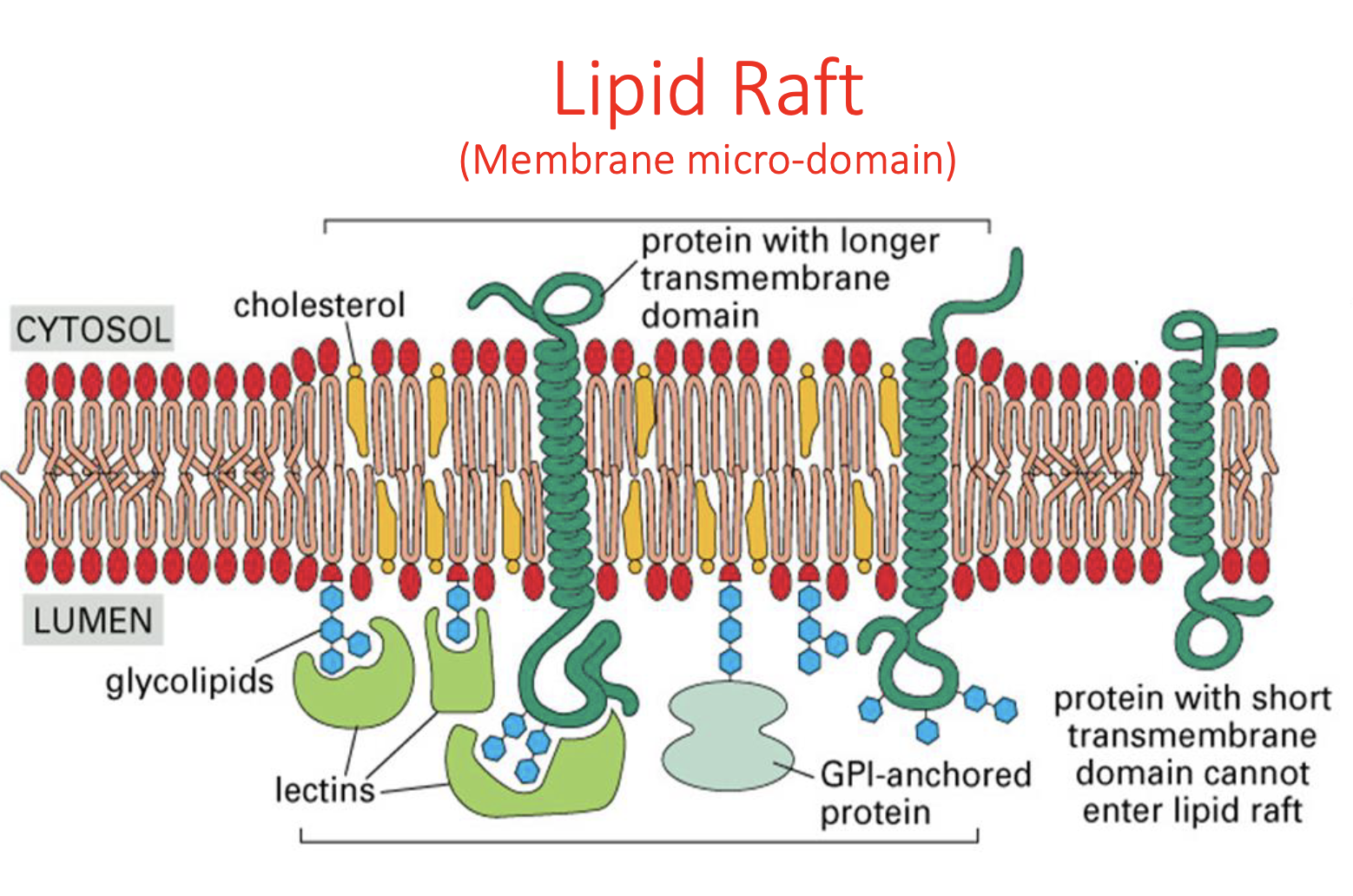
Cells use lipid rafts for
Regulatory function
—> being able to separate signaling molecules/cascades so they aren’t turned on all the time
Cholesterol exists in two main states in the body
transported (packaged) within lipoproteins in the blood
structurally embedded within cell phospholipid membranes.
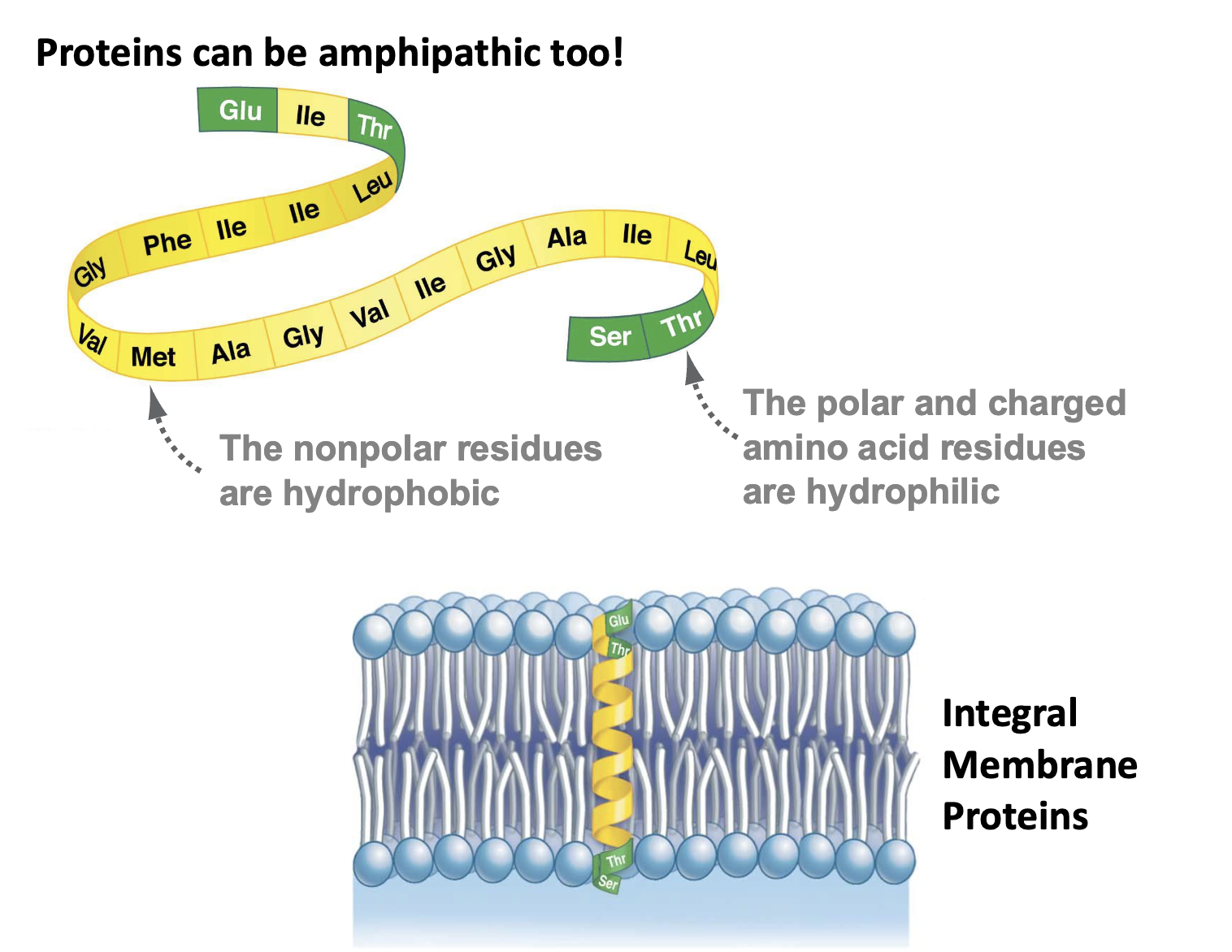
Proteins can be amphipathic too…
can be polar or nonpolar so they can embed themselves in membranes w/non polar amino acids & polar amino acids on outside
The lipid raft model is considered a "better" or more accurate refinement of the Fluid Mosaic Model because
it acknowledges that the cell membrane is not a homogeneous, random sea of lipids and proteins
shows that membranes contain specialized, ordered, and functional microdomains that act as signaling platforms, providing a more complex and biologically realistic view.
essentially a more advanced version
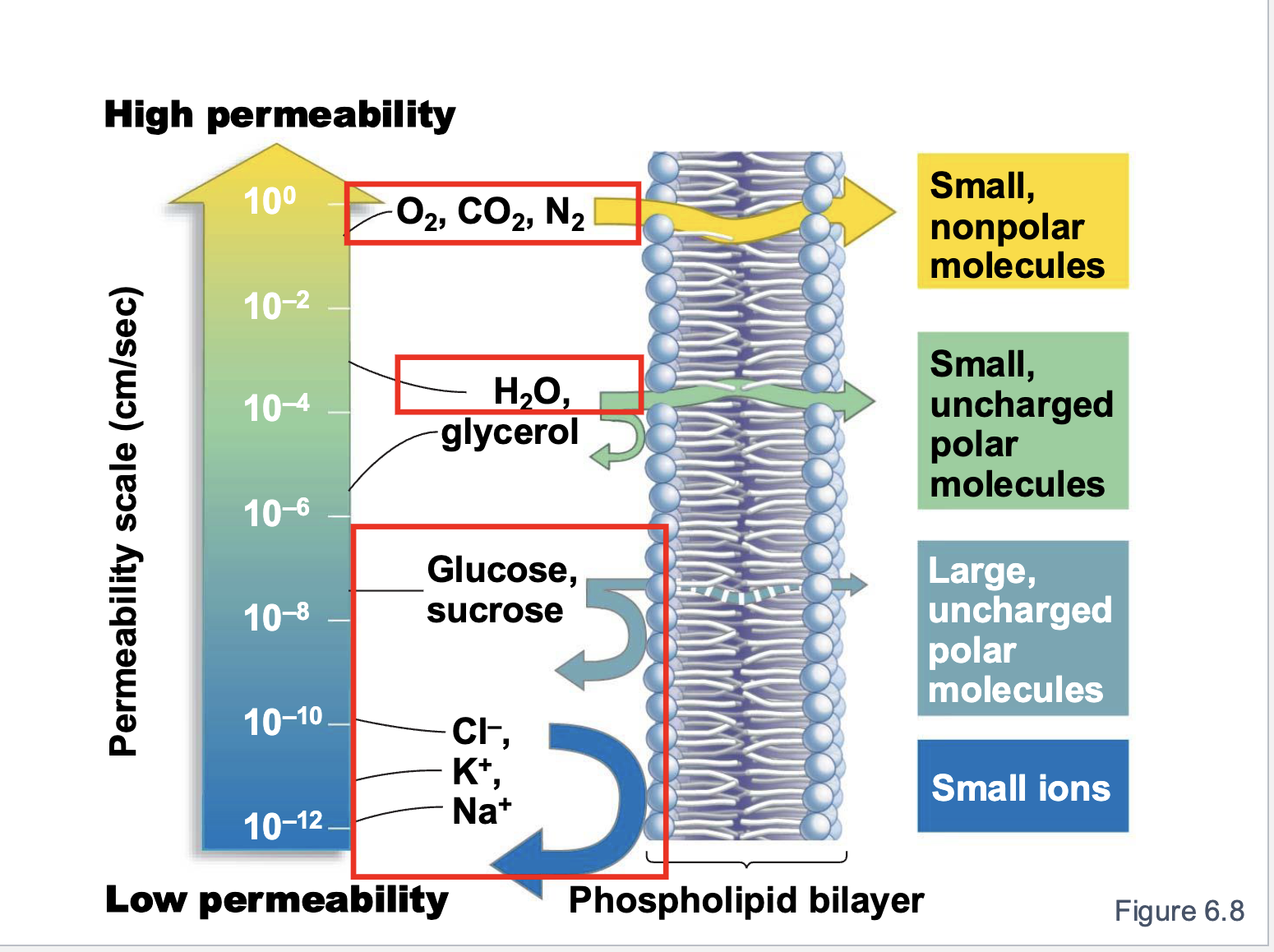
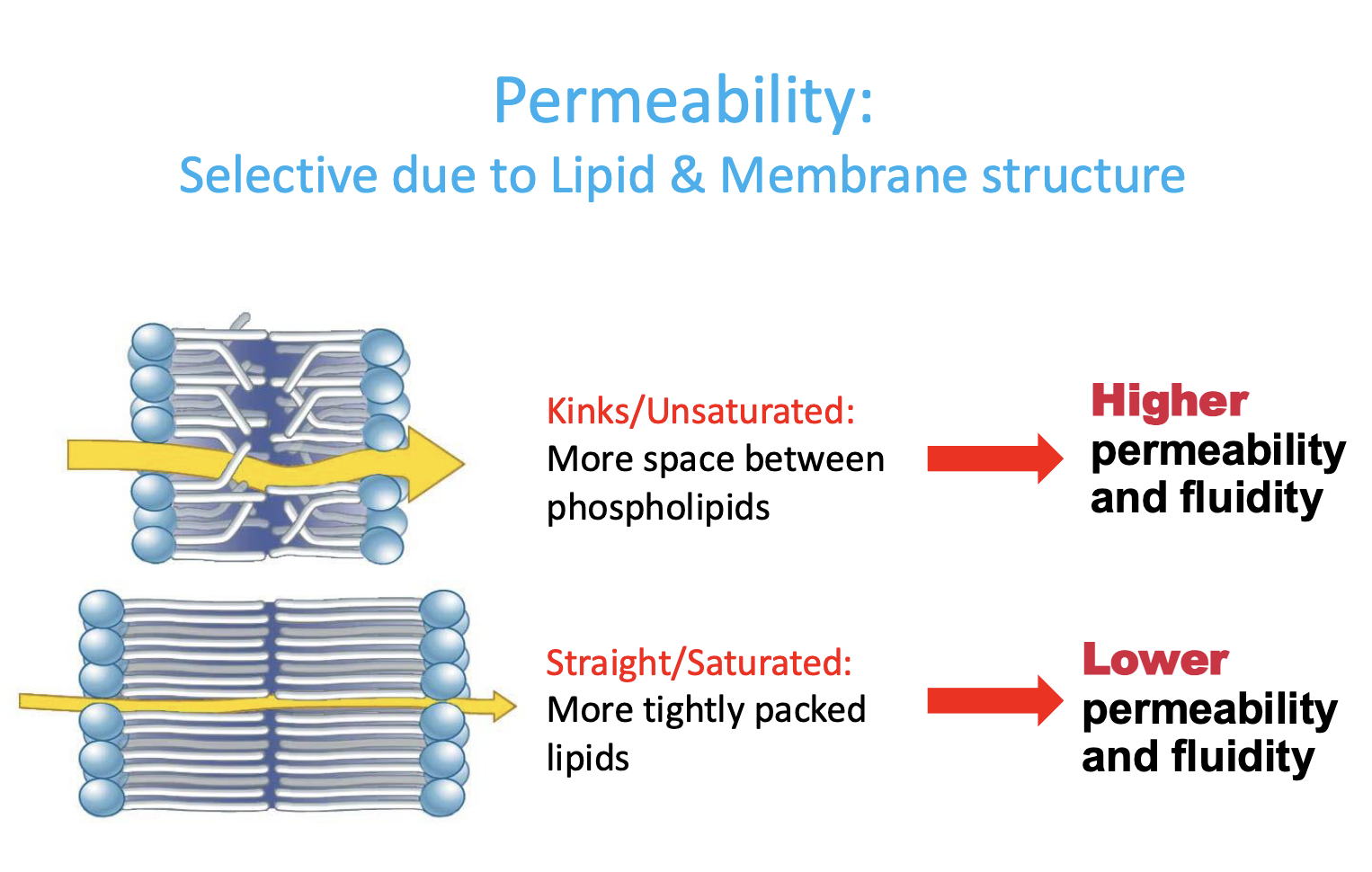
permeability
selective due to lipid & membrane structure
based on fatty acid saturation/unsaturation
Membranes: Kins/Unsatured
more spaces between phospholipids —> HIGHER permeability & fluidity
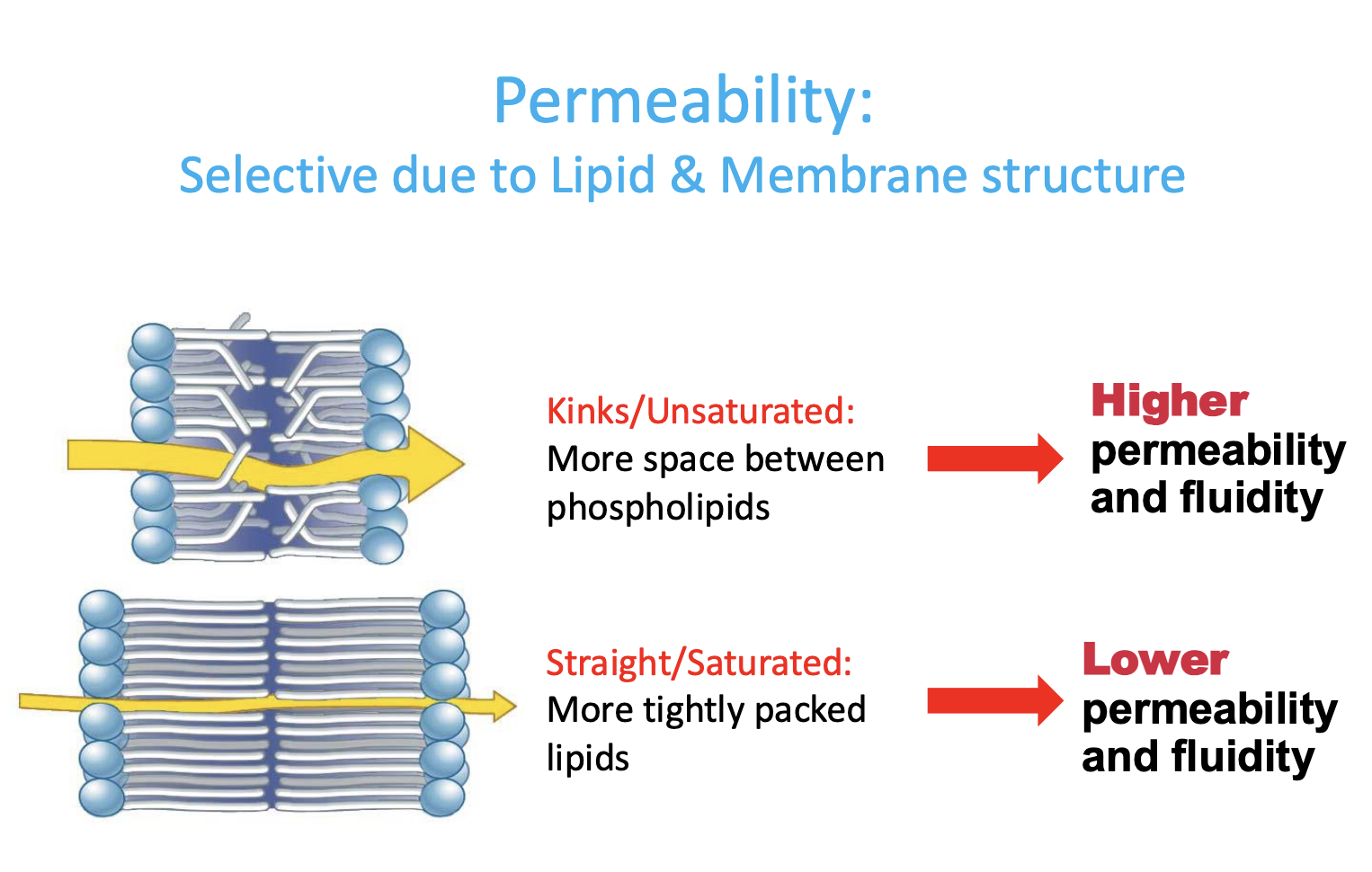
Membranes: Straight/Saturated
more tightly packed lipids —> LOWER permeability & fluidity
Introduction of cholesterol in membrane
decreases fluidity
What is membrane permeability?
What substances can cross the membrane
What is membrane fluidity?
flexible & dynamic nature of the cellular lipid bilayer, allowing lipids and proteins to move laterally within the membrane
Movement of lipids within the bilayer
Are fluidity and permeability the same?
no
What is cholesterol’s role in membranes?
Regulates fluidity and stabilizes membrane
How does cholesterol affect permeability?
Decreases permeability to small molecules
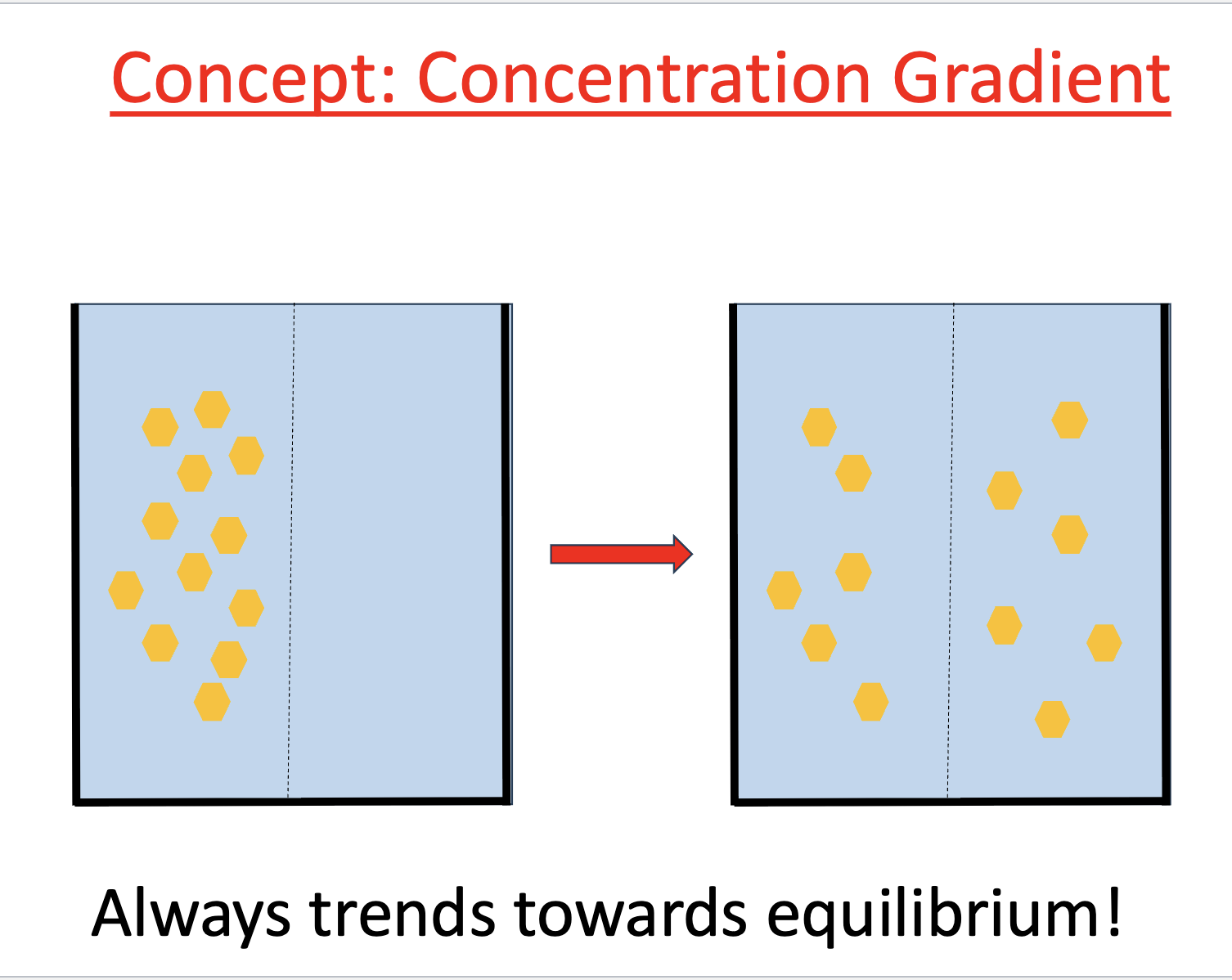
Concentration gradient
always trends toward equilibrium
concentration gradients of molecules on sides of membranes
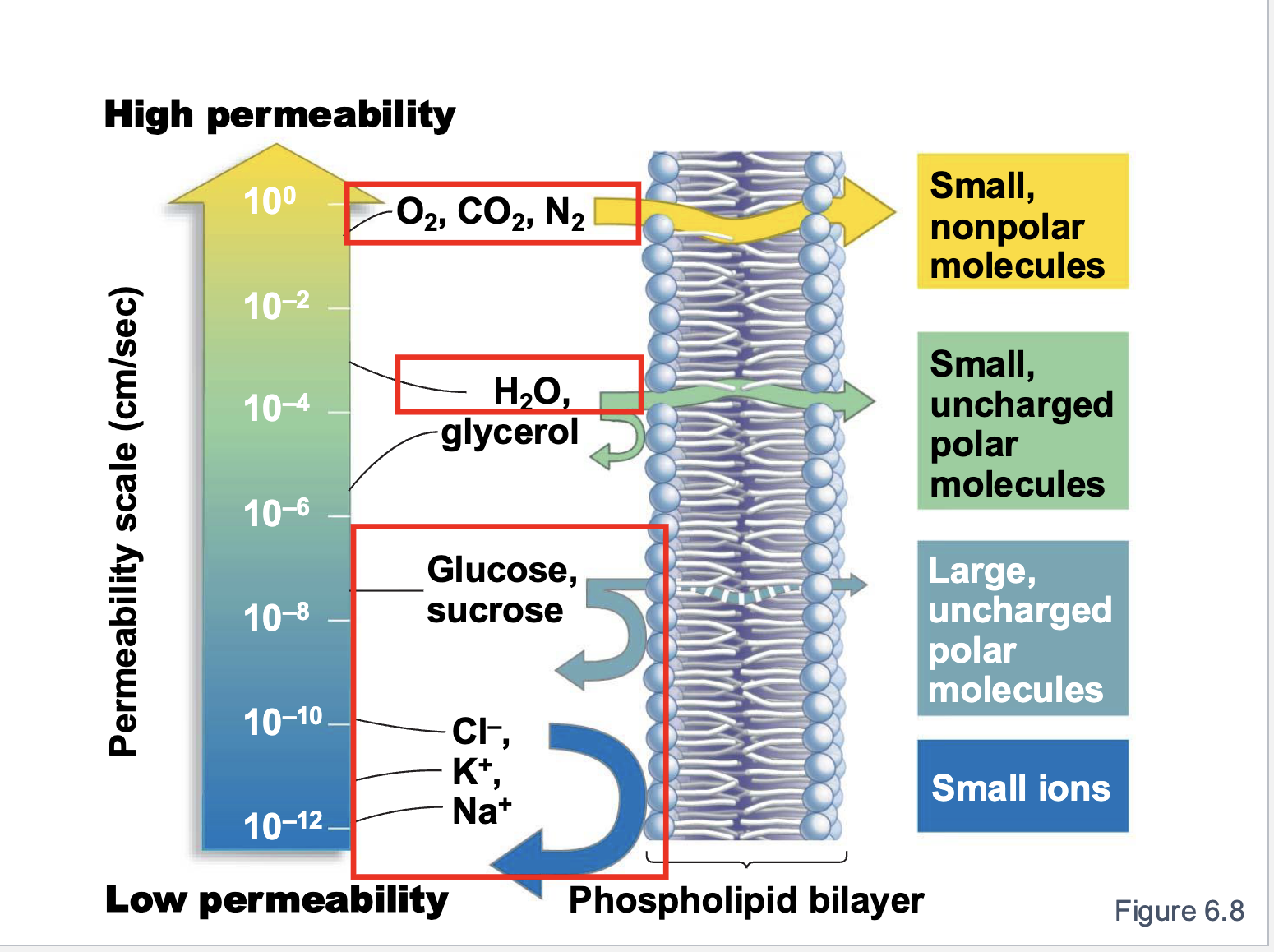
What has the highest permeability?
small, nonpolar molecules
What has the lowest permeability?
small ions
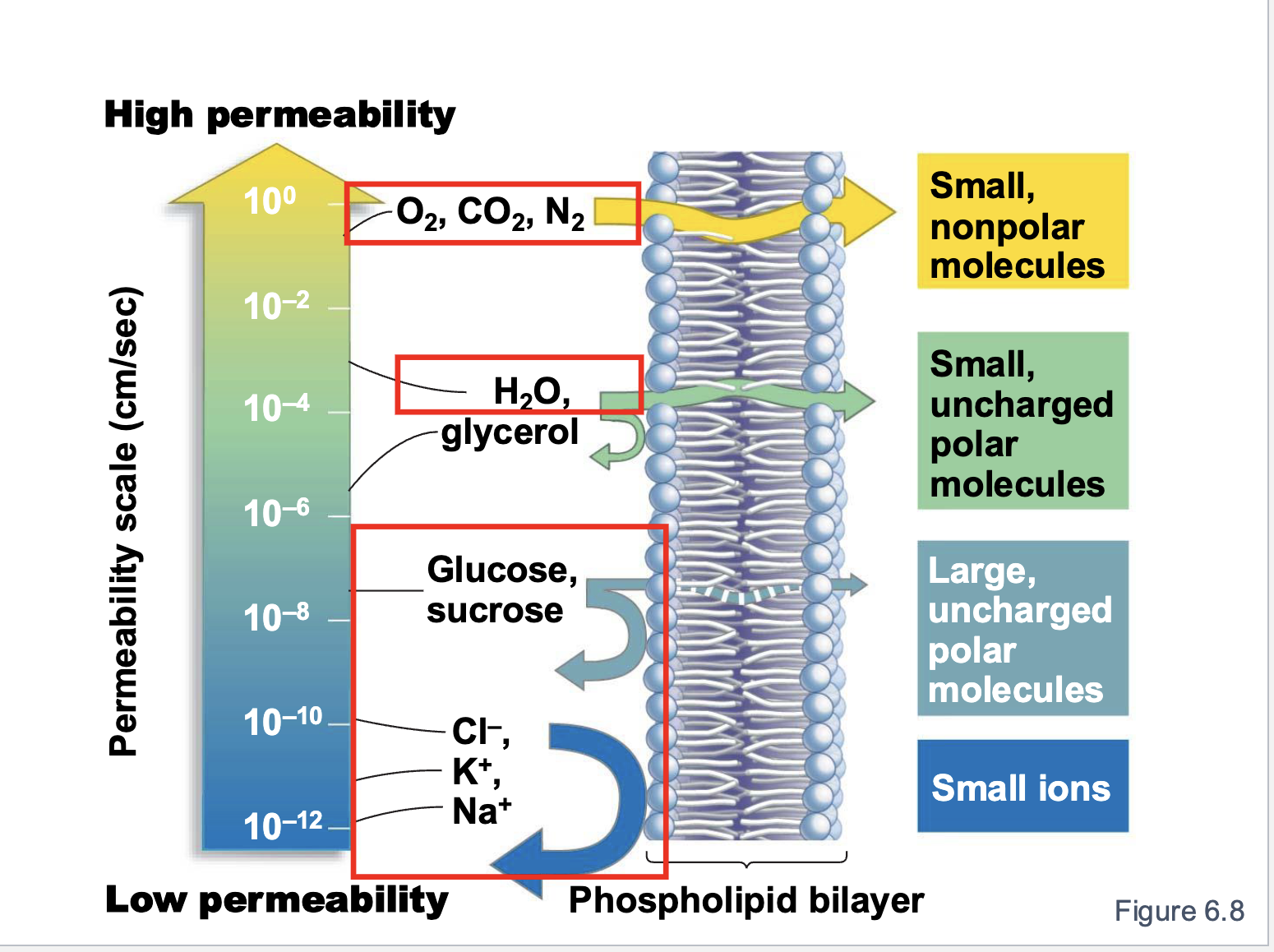
Order of permeability
small non polar molecules —> small uncharged polar molecules —> large uncharged polar molecules —> small ions