Structure + Physical properties
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
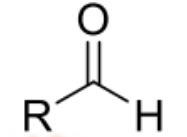
general structure and nomenclature of aldehyde
RCHO where R= H, alkyl or aryl(benzaldehyde)
name ends with ‘al’ eg methanal, ethanal
Carbon in -CHO is numbered as carbon 1
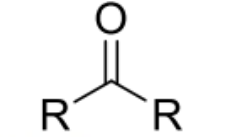
general structure and nomenclature of ketones
RCOR’ where R/R’=alkyl or aryl
name ends with -one eg propanone (there needs to be at least 3 carbons so methanone and ethanone do not exist)
eg pentan-2-one —> ketone group is in the position 2
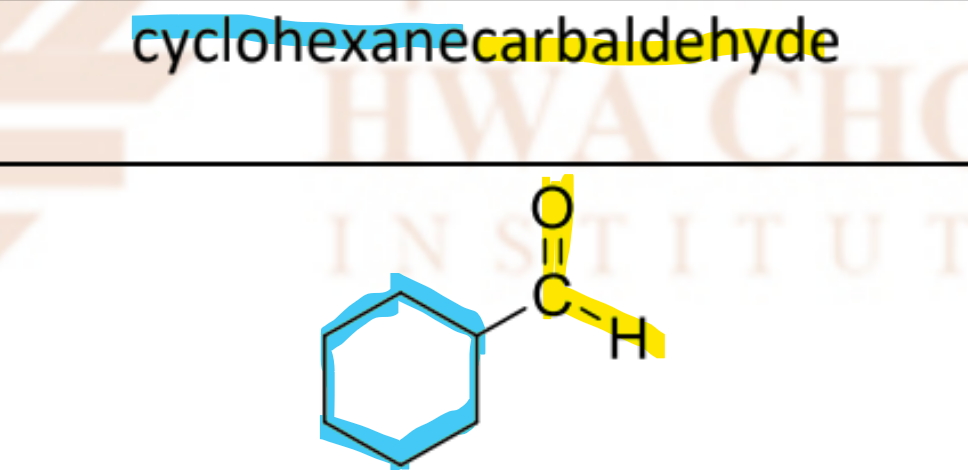
cyclohexanecarbaldehyde
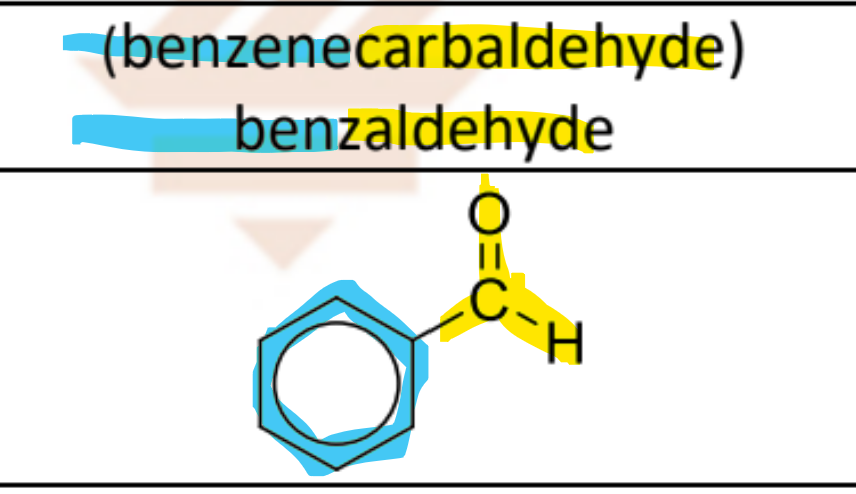
benzenecarbaldeyde OR benzaldehyde
aldehydes and ketones are _______ isomers
constitutional isomers
Order the following in increasing boiling point:
aldehydes and ketones
hydrocarbons
alcohols and carboxylic acis
(assuming all have similar no. of e'-)
hydrocarbons > aldehydes and ketones > alcohols and carboxylic acids
Why do aldehydes and ketones have higher bp than corresponding hydrocarbons?
carbonyl group is polar
more energy required to overcome the stronger permanent dipole-permanent dipole interactions exist between the molecules in aldehydes/ketones
than the weaker disperson forces between hydrocarbon molecules
Why do aldehydes and ketones have lower bp than alcohols and carboxylic acids?
less energy is required to overcome the relatively weaker permanent dipole-permanent dipole interactions between molecules in aldehydes/ketones
as compared to the stronger hydrogen bonds between molecules in alcohol/carobxylic acids
What kinds of carbonyl compounds are soluble and why?
smaller aliphatic(no ring structure) aldehydes and ketones
smaller non-polar groups which result in weaker dispersion forces and less disruption in hydrogen bonding
energy released from the formation of stronger hydrogen bonds between aldehyde/ketone and water molecules
more able to overcome the permanent dipole-permanent dipole interactions as well as weaker dispersion forces between aldeyde/ketone molecules and hydrogen bonds between water molecules
What kinds carbonyls compounds are virtually insoluble and why?
aldehydes/ketones containing alkyl chains of more than 5 carbons or an aromatic ring
this results in stronger dispersion forces and more disrupted hydrogen bonding with water
energy released from the formation of weaker hydrogen bonds between aldehyde/ketone and water molecules
less able to overcome the permanent dipole-permanent dipole interactions as well as stronger dispersion forces between aldeyde/ketone molecules and hydrogen bonds between water molecules