AQA A Level Chemistry: Inorganic
1/148
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
149 Terms
Metallic (elements)
Sodium, magnesium and aluminium
Semi-metal (metalloid) (elements)
Silicon
Non-metals (elements)
Phosphorus, sulfur and chlorine
Noble gas (elements)
Argon
Metallic (Description)
Shiny, conduct electricity, react with dilute acids to give hydrogen and salts
Semi-metal (metalloid) (Description)
Conducts electricity to some extent, useful form making semiconductor devices
Non-metals (Description)
Don't conduct electricity and have low melting and boiling points
Noble gas (Description)
Chemically unreactive and exists as separate atoms
2Na + 2H2O --> 2NaOH + H2
Sodium + water reaction
Mg + 2H2O --> Mg(OH)2 + H2
Magnesium + water reaction
Mg + H2O --> MgO + H2
Magnesium + steam reaction
4Na + 2O2 --> 2Na2O
Sodium + oxygen reaction
2Mg + O2 --> 2MgO
Magnesium + oxygen reaction
4Al + 3O2 --> Al2O3
Aluminium + oxygen reaction
Si + O2 --> SiO2
Silicon + oxygen reaction
4P + 5O2 --> P4O10
Phosphorus + oxygen reaction
S + O2 --> SO2
Sulfur + oxygen reaction
Allotropes
Pure elements which can exist in different physical forms in which their atoms are arranged differently.
Na2O (Name)
Sodium oxide
Na2O (Bonding)
Ionic bonding
Na2O (Structure)
Giant ionic structure
Na2O (pH)
Strongly alkaline (pH 13-14)
Na2O (Acid or base)
Basic
MgO (Name)
Magnesium oxide
MgO (Bonding)
Ionic bonding
MgO (Structure)
Giant ionic structure
MgO (pH)
Weaker alkaline (pH 10)
MgO (Acid or base)
Basic
Al2O3 (Name)
Aluminium oxide
Al2O3 (Bonding)
Ionic/covalent bonding
Al2O3 (Structure)
Giant ionic structure
Al2O3 (Acid or base)
Amphoteric
SiO2 (Name)
Silicon dioxide
SiO2 (Bonding)
Covalent bonding
SiO2 (Structure)
Giant covalent (macromolecular) structure
SiO2 (Acid or base)
Acidic
P4O10 (Name)
Phosphorus pentoxide (Phosphorus (V) oxide)
P4O10 (Bonding)
Covalent bonding
P4O10 (Structure)
Molecular structure
P4O10 (pH)
Fairly strong acid (pH 1-2)
P4O10 (Acid or base)
Acidic
SO3 (Name)
Sulfur trioxide
SO3 (Bonding)
Covalent bonding
SO3 (Structure)
Molecular structure
SO3 (pH)
Strong acid (pH 0-1)
SO3 (Acid or base)
Acidic
SO2 (Name)
Sulfur dioxide
SO2 (Bonding)
Covalent bonding
SO2 (Structure)
Molecular structure
SO2 (pH)
Weak acid (pH 2-3)
SO2 (Acid or base)
Acidic
Na2O + H2O --> 2NaOH
Sodium oxide + water
MgO + H20 --> Mg(OH)2
Magnesium oxide + water
P4O10 + H2O --> 4H3PO4
Phosphorus pentoxide + water
SO2 + H2O --> H2SO3
Sulfur dioxide + water
SO3 + H2O --> H2SO4
Sulfur trioxide + water
Na2O + HCl --> 2NaCl + H2O
Sodium oxide + HCl
MgO + 2HCl --> MgCl2 + H2O
Magnesium oxide + HCl
Al2O3 + 6HCl --> 2AlCl3 + 3H2O
Aluminium oxide (as a base) + HCl
Al2O3 + 2NaOH + 3H2O --> 2Na+[Al(OH)4]-
Aluminium oxide (as an acid) + NaOH
SiO2 + 2NaOH --> Na2SiO3 + H2O
Silicon dioxide + NaOH
P4O10 + 12NaOH --> 4Na3PO4 + 6H2O
Phosphorus pentoxide + NaOH
SO2 + 2NaOH --> Na2SO3 + H2O
Sulfur dioxide + NaOH
SO3 + 2NaOH --> Na2SO4 + H2O
Sulfur trioxide + NaOH
Amphoteric
A substance that can act as both an acid and a base
2SO2 + O2 <--> 2SO3
The contact process (V2O5 catalyst)
Transition element
An element that forms at least one stable ion with a part filled d-orbital of electrons
Co-ordinate bond
A covalent bond in which both of the electrons in the bond come from one of the atoms forming the bond
Dative bond
A covalent bond in which both of the electrons in the bond come from one of the atoms forming the bond
Ligand
An atom, ion or molecule that forms a co-ordinate (dative) bond with a transition metal ion using a lone pair of electrons
Co-ordination number
The number of ligand molecules bonded to a metal ion
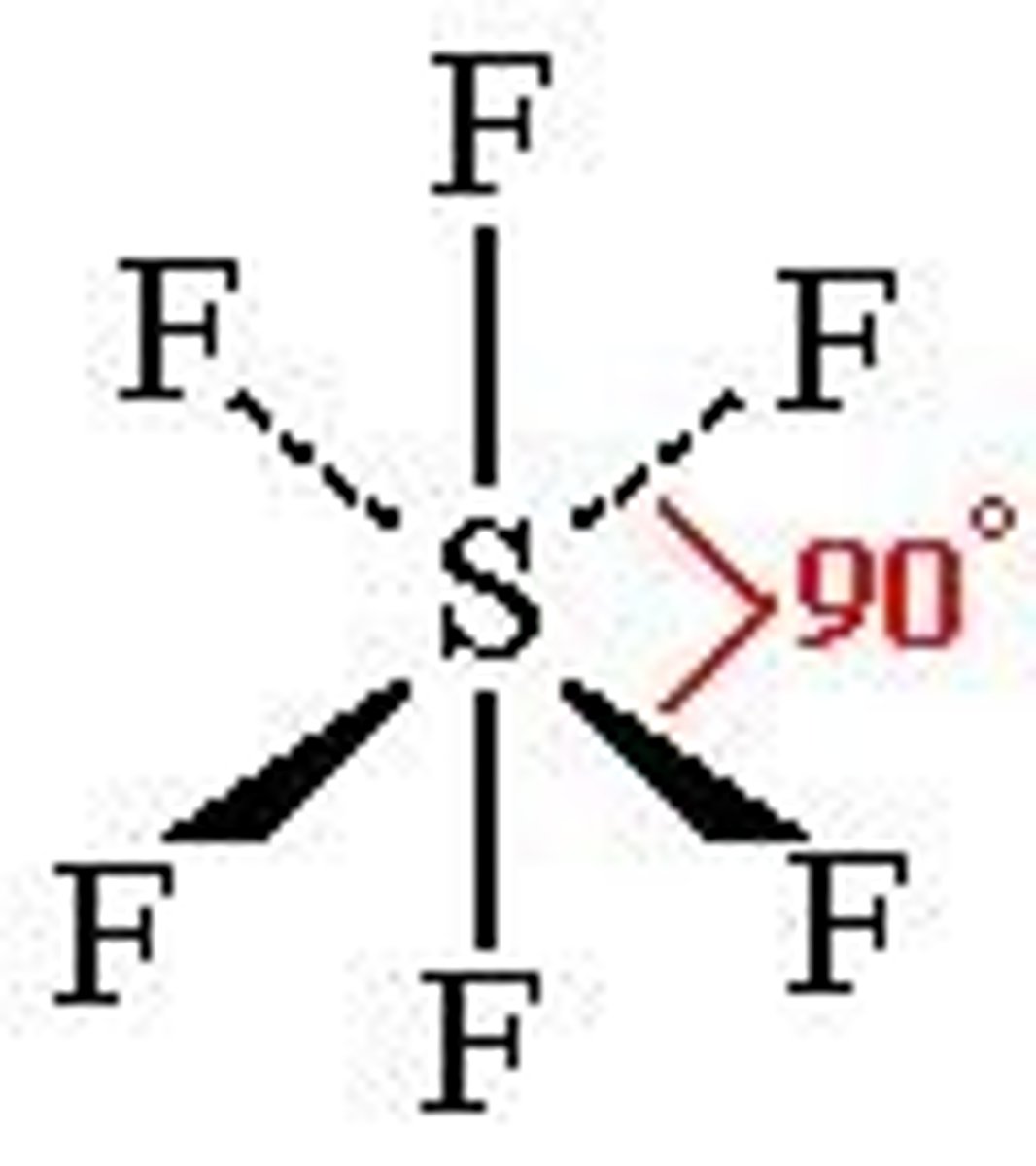
Octahedral
90 degrees
Co-ordination number: 6
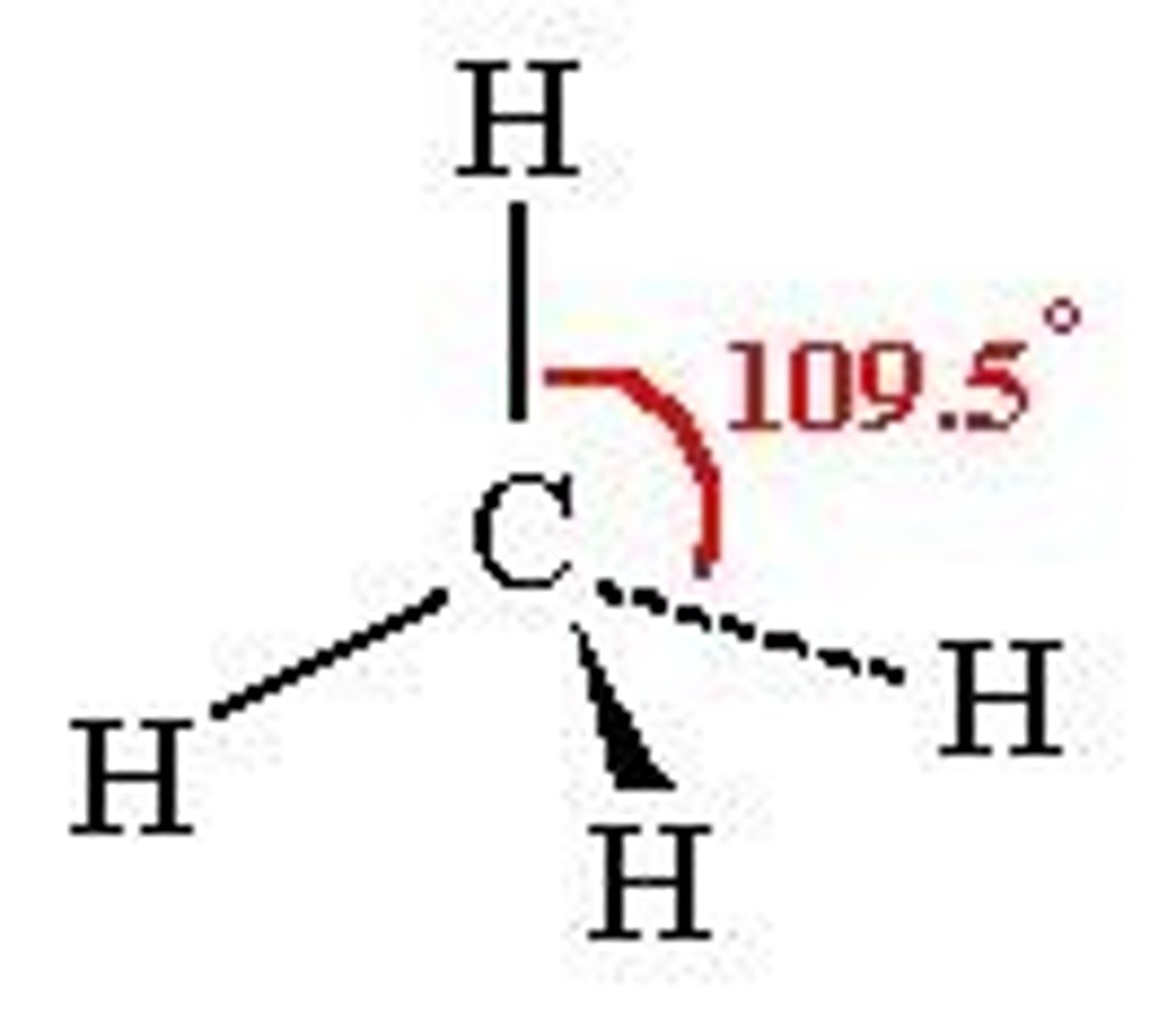
Tetrahedral
109.5 degrees
Co-ordination number: 4
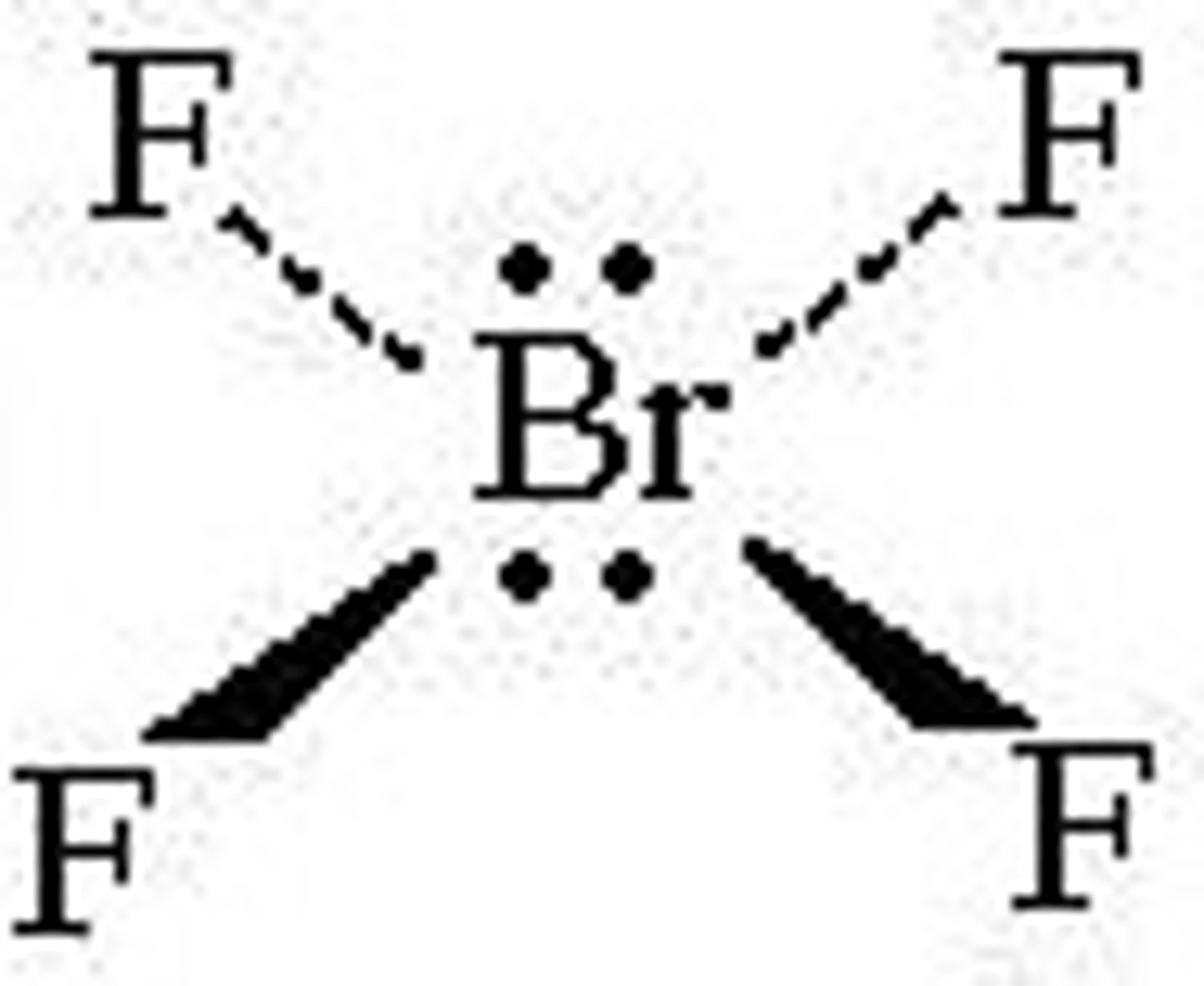
Square planar
90 degrees
Co-ordination number: 4
Linear
180 degrees
Co-ordination number: 2

Aqua ions
A transition metal ion surrounded by (usually 6) water molecules acting as ligands
Multidentate ligand
A ligand that has more than one atom with a lone pair of electrons which can bond to a transition metal ion
Chelates
Complex ions with polydentate ligands
Chelate effect
Chelate complexes with polydentate ligands are favoured over complexes with monodentate ligands
Geometrical isomer
Ligands differ in their position in space relative to one another
Optical isomer
Two isomers are non-superimposable mirror images of each other
Chiral
A molecule that exists in two mirror image forms that are not superimposable
Green
[Fe(H2O)6]2+ colour
Pale brown
[Fe(H2O)6]3+ colour
Blue
[Cr(H2O)6]2+ colour
Red-violet
[Cr(H2O)6]3+ colour
Brown
[Co(NH3)6]2+ colour
Yellow
[Co(NH3)6]3+ colour
5Fe2+ + MnO4- + 8H+ --> 5Fe3+ + Mn2+ + 4H2O
Potassium manganate + iron (II)
Heterogeneous
Catalyst in a reaction in a different phase than the reactants
N2 + 3H2 <--> 2NH3 (iron catalyst)
Haber process
Iron
Catalyst in Haber process
SO2 + V2O5 --> SO3 + V2O4
First step of the Contact process
2V2O4 + O2 --> 2V2O5
Second step of the Contact process (regeneration of the catalyst)
V2O5
Catalyst for the Contact process
V2O5
Vanadium(V) oxide
Homogeneous
Catalyst in reaction in the same phase as the reactants, intermediate species formed
Autocatalysis
When one of the products of a reaction is the catalyst for that reaction
2MnO4- + 5(C2O4)2- + 16H --> 2Mn2+ + 8H2O + 10CO2
Oxidation of ethanoic acid by manganate (VII) ions
[Fe(H2O)6]3+ <--> [Fe(H2O)5(OH)]2+ + H+
Hydrolysis with Fe3+