Honors Bio: Unit 4 (1-52) 5 (53-
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
Thylakoid
flattened, disc-like membrane-bound sacs inside chloroplasts (or cyanobacteria) that serve as the site for the light-dependent reactions of photosynthesis
Plasma membrane
Without it the cells of living things would die because they are unable to maintain homeostasis
Are the cell membrane and and plasma membrane the same structure?
Yes, the cell membrane and plasma membrane generally the same structure and the terms are used interchangeably. They both refer to the semipermeable lipid bilayer that surrounds all living cells
Cell membrane structure
lipid bilayer (mainly phospholipids), along with proteins and carbohydrates. It acts as a semipermeable, fluid barrier that protects the cell
What is a phospholipid
Phospholipid, “phospho” standing for phosphate, and “lipid” standing for the lipid (fatty acids) this represents its phosphate head and fatty acid tail. Its head being polar and its tail being non polar. it being referred to as the Bilayer (as in phospholipid bilayer) means it includes two layers, composed of two phospholipids together in a long string with their tails facing one another because the heads are hydrophilic and the tails hydrophobic
How do charged molecules enter through the plasma membrane?
Charged particles must pass through transmembrane proteins, such as ion channels or pump proteins, which provide a hydrophilic path through the hydrophobic layer
Why can’t a charged molecule enter the plasma membrane?
because of its hydrophobic core. The membrane consists of a phospholipid bilayer with nonpolar fatty acid tails that repel charged substances. These particles are strongly attracted to water and cannot overcome the high energy barrier required to cross the oily, hydrophobic interior.
How does water enter the plasma membrane?
through specialized channel proteins called aquaporins via facilitated diffusion. While water is polar, it is small enough to occasionally pass directly through the lipid bilayer, but the majority passes through these water-selective channels. This movement is driven by osmosis to balance solute concentrations.
What is osmosis?
the passive movement of water molecules across a semipermeable membrane from a region of higher water concentration (lower solute concentration) to a region of lower water concentration (higher solute concentration) (Energy free process)
What is the difference between polar and non-polar molecules?
Polar molecules have an uneven distribution of electrons, creating partial positive and negative ends (poles), similar to a tiny magnet, such as water. Nonpolar molecules have an even distribution of electrons, resulting in a balanced, symmetrical structure with no charged poles, like oil or methane.
How does protein contribute to the phospholipid bilayer?
Proteins are embedded in the membrane, in the form of integral proteins, channel proteins, and glycoprotein.
How does colesterol contribute to the phospholipid bilayer?
Keeps membrane fluid and flexible
How do carbohydrates contribute to the phospholipid bilayer?
Contribute to protection and cell recognition.
Permeable
the capacity of the phospholipid bilayer and its embedded proteins to allow specific molecules, ions, and substances to pass into or out of the cell
Impermeable
Can NOT enter
Is the cell membrane the only organelle that allows for the diffusion of molecules?
No! Additionally to the plasma membrane is the Mitochondria & Chloroplasts, Endoplasmic Reticulum (ER), and the Golgi Apparatus
Simple diffusion
Movement of molecules from an area of high concentration to an area of low concentration through the lipid bilayer until equilibrium is reached
Facilitated diffusion
When channel proteins help large molecules or charged molecules pass through the membrane (ex. Glucose and water). Osmosis being a popular example of facilitated diffusion
Tonicity
the measure of an extracellular solution's ability to change a cell's volume by affecting osmosis (water movement) across its membrane
how can some molecules pass through the plasma membrane without a channel?
pass through the plasma membrane without a channel via simple diffusion. They move directly through the hydrophobic phospholipid bilayer down their concentration gradient.
Hypotonic
have a higher concentration of solutes inside the cell compared to the surrounding solution. This causes water to move into the cell through osmosis, making it swell
Hypertonic
exists in an environment with a higher solute concentration (lower water concentration) than its cytoplasm, causing water to leave the cell via osmosis
Isotonic
exists in an environment with the same solute concentration as its cytoplasm, resulting in no net water movement across the membrane
Passive transport
The movement of materials across a cell membrane without the use of ATP/ energy. The types are simple diffusion and facilitated diffusion, osmosis being a popular example of facilitated diffusion
Active transport
the energy-dependent process (using ATP) of moving molecules or ions across a cell membrane. Ex. protein pump, and built transport
What happened if a cell wants to move a substance from an area of low concentration to one of high concentration?
Active transport! Sometimes cells need high concentrations of certain substances and low concentrations of others. When a cell wants to do this it needs to use energy to pump the substance against its concentration gradient (going against the flow).
Exocytosis
an active transport process where cells expel molecules, such as neurotransmitters, hormones, and waste, by fusing intracellular vesicles with the plasma membrane
What type of transport does not require energy?
Passive transport
What are the two main components of the cell membrane?
Phospholipids and proteins
How does oxygen pass into the cell?
Simple diffusion
How does glucose cross into the cell?
Facilitated diffusion/ active transport
What is the purpose of channel protein?
To allow the transport of specific molecules through the membrane examples being Ion Channels, and Aquaporins
Solute
A compound that can dissolve in water, often sugar or salt, to create a solution
Hypertonic solutions have…
More solute because they are defined by having a greater solute-to-solvent ratio, causing a higher osmotic pressure. This high concentration gradient forces water to move out of the cell to balance the environment, often causing cells to shrivel.
Hypotonic solutions have…
Less solute and a higher water concentration than the inside of a cell, which causes water to move into the cell through osmosis often making it swell.
Endocytosis
an active transport mechanism where cells ingest extracellular fluid, molecules, and particles by engulfing them in a plasma membrane vesicle that buds off into the cytoplasm
Solvent
a substance, usually a liquid, capable of dissolving, suspending, or extracting other materials (solutes) without changing their chemical makeup, commonly used to create solutions, clean, or dilute substances.
Stroma
the colorless, fluid-filled space within the inner membrane of a chloroplast that surrounds the thylakoid membranes
Concentration gradient
the gradual difference in the concentration of a dissolved substance (solute) between two areas, typically separated by a membrane. It represents an unequal distribution of particles, where movement occurs from high to low concentration (down the gradient) to achieve equilibrium.
Light Dependent
the initial stage of photosynthesis, occurring within the chloroplast's thylakoid membranes, where chlorophyll absorbs solar energy to convert water, ADP, and NADP+ into chemical energy (ATP and NADPH) and oxygen (O2).
Calvin cycle
light-independent, second stage of photosynthesis occurring in the chloroplast stroma, where ATP and NADPH are used to fix atmospheric CO2 into carbohydrates
chloroplast
specialized, double-membrane organelles in plant and algal cells that function as the site of photosynthesis, converting solar energy into chemical energy (glucose) using chlorophyll.
Glycolysis
a 10-step, cytosol-based metabolic pathway that splits one 6-carbon glucose molecule into two 3-carbon pyruvate molecules
Kreb’s Cycle
a mitochondrial matrix process that oxidizes acetyl-CoA to generate energy (ATP, NADH, \FADH2) and precursors for biosynthesis. It occurs in eight steps: citrate formation, isomerization, decarboxylation/oxidation, succinyl-CoA formation, phosphorylation, oxidation, hydration, and regeneration of oxaloacetate
Electron transport chain
a series of protein complexes and electron carriers embedded in the inner mitochondrial membrane (or prokaryotic plasma membrane) that generates ATP through oxidative phosphorylation
aerobic cellular respiration
the metabolic process of breaking down glucose in the presence of oxygen (O2) to produce ATP, water, and carbon dioxide. It is the primary energy-generating mechanism for eukaryotic cells.
Eukaryotic cell
complex, membrane-bound cellular structures containing a distinct nucleus that houses DNA, along with specialized organelles like mitochondria and the Golgi apparatus
Prokaryotic cell
Prokaryotic cells are small, single-celled organisms from the Bacteria and Archaea domains that lack a nucleus and membrane-bound organelles. They feature a nucleoid region containing circular DNA, a rigid cell wall, ribosomes, and often flagella or pilli. They reproduce via binary fission and are found globally
mitochondria
generate over 90% of cellular energy as ATP via aerobic respiration. They are vital for metabolism (breaking down fats and sugars), calcium signaling, iron-sulfur cluster synthesis, and regulating cell death (apoptosis)
Anaerobic cellular respiration
a metabolic process that breaks down glucose to produce energy (ATP) in the absence of oxygen
Fermentation
an anaerobic (oxygen-absent) pathway that follows glycolysis to produce energy
Lactic Acid
an organic compound produced by muscles and red blood cells to break down carbohydrates for energy, particularly during intense exercise or low-oxygen conditions (C3H6O3)
Somatic Cells
all non-reproductive cells forming the body of a multicellular organism
Interphase
the longest part of the cell cycle, where a cell grows, replicates its DNA, and prepares for division. It consists of three stages: G1 phase (growth and normal metabolism), S phase (DNA synthesis/replication), and G2 phase (final growth and preparation), leading to the M phase (mitosis).
Mitosis
a type of cell division that results in two daughter cells each having the same number and kind of chromosomes as the parent nucleus, typical of ordinary tissue growth. Prophase, Metaphase, Anaphase, and Telophase.
Cytokinesis
the final stage of cell division, following mitosis or meiosis, where the cytoplasm of a parental cell physically divides into two, generally equal, daughter cells.
Chromatin vs chromosome
Chromatin is the relaxed, thread-like complex of DNA and histone proteins present during interphase for gene expression. Conversely, chromosomes are highly condensed, distinct structures formed from chromatin specifically during cell division for DNA segregation. Essentially, chromatin is the material, and chromosomes are its organized, compact form.
Sister Chromatids
two genetically identical, replicated DNA molecules joined at a centromere, forming a single chromosome.
Nucleotide
the essential building blocks of nucleic acids (DNA and RNA), consisting of a nitrogenous base, a five-carbon sugar, and a phosphate group.
Phosphate group
Phosphate groups create covalent linkages between sugar molecules, forming the sugar-phosphate backbone of nucleic acids.
deoxyribose sugar
a 5-carbon monosaccharide sugar, forming the structural backbone of DNA by alternating with phosphate groups. It is derived from ribose, lacking one oxygen atom, which makes DNA more chemically stable than RNA.
bases ATGC
the four nitrogenous nucleotides—Adenine (A), Thymine (T), Cytosine (C), and Guanine (G)—that form the genetic alphabet of DNA. They pair specifically (A-T and G-C) to form the rungs of the DNA double helix, coding for all genetic information.
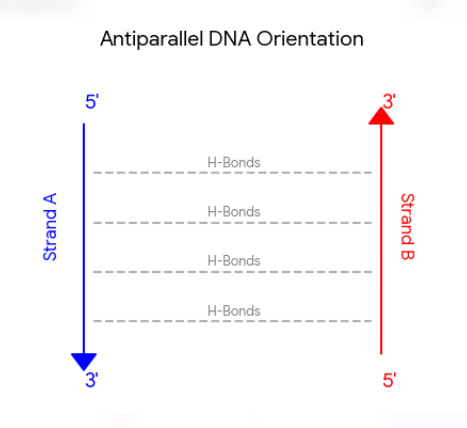
Antiparallel (5’ and 3’)
One strand runs in the 5’ to 3’ direction, while the complementary strand runs in the 3’ to 5’ direction. This orientation is essential for stable hydrogen bonding between nitrogenous bases and dictates how enzymes interact with the genetic code.
Semiconservative
the mechanism by which DNA copies itself, resulting in two molecules each containing one original parent strand and one newly synthesized strand
Hydrogen bonds
weak, non-covalent forces that connect complementary nitrogenous bases on opposite strands, crucial for stabilizing the double helix
Purine vs. Pyrimidines
Purines (Adenine, Guanine) are larger, double-ring nitrogenous bases found in DNA/RNA, while Pyrimidines (Cytosine, Thymine, Uracil) are smaller, single-ring bases
Pedigrees
a diagram or record using standardized symbols to map family members, their relationships, and the inheritance of specific traits, genetic variants, or diseases across generations.
Autosomal
refers to the 22 pairs of numbered, non-sex chromosomes found in human cells (chromosomes 1-22), which are inherited equally from both parents.
Complete Dominance
a form of inheritance in biology where the dominant allele completely masks the expression of the recessive allele in a heterozygous organism
Dihybrid Crosses
tracks the inheritance of two different traits simultaneously, examining how alleles for two separate genes segregate independently