Ionization Energy: Trends, Definitions, and Periodic Table Analysis
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
What is ionization energy?
The energy required to remove an electron from a neutral atom in its gaseous phase.
Is ionization energy always positive or negative?
Ionization energy is always positive.
What is the first ionization energy?
The energy required to remove the outermost, or highest energy, valence electron.
What is the second ionization energy?
The energy required to remove the next highest energy valence electron from a gaseous cation.
Why do subsequent ionization energies require significantly more energy?
Because the preceding configuration may be in a stable octet formation, requiring more energy to ionize the valence electron.
How does ionization energy trend across the periodic table?
Ionization energy generally increases from left to right across a period.
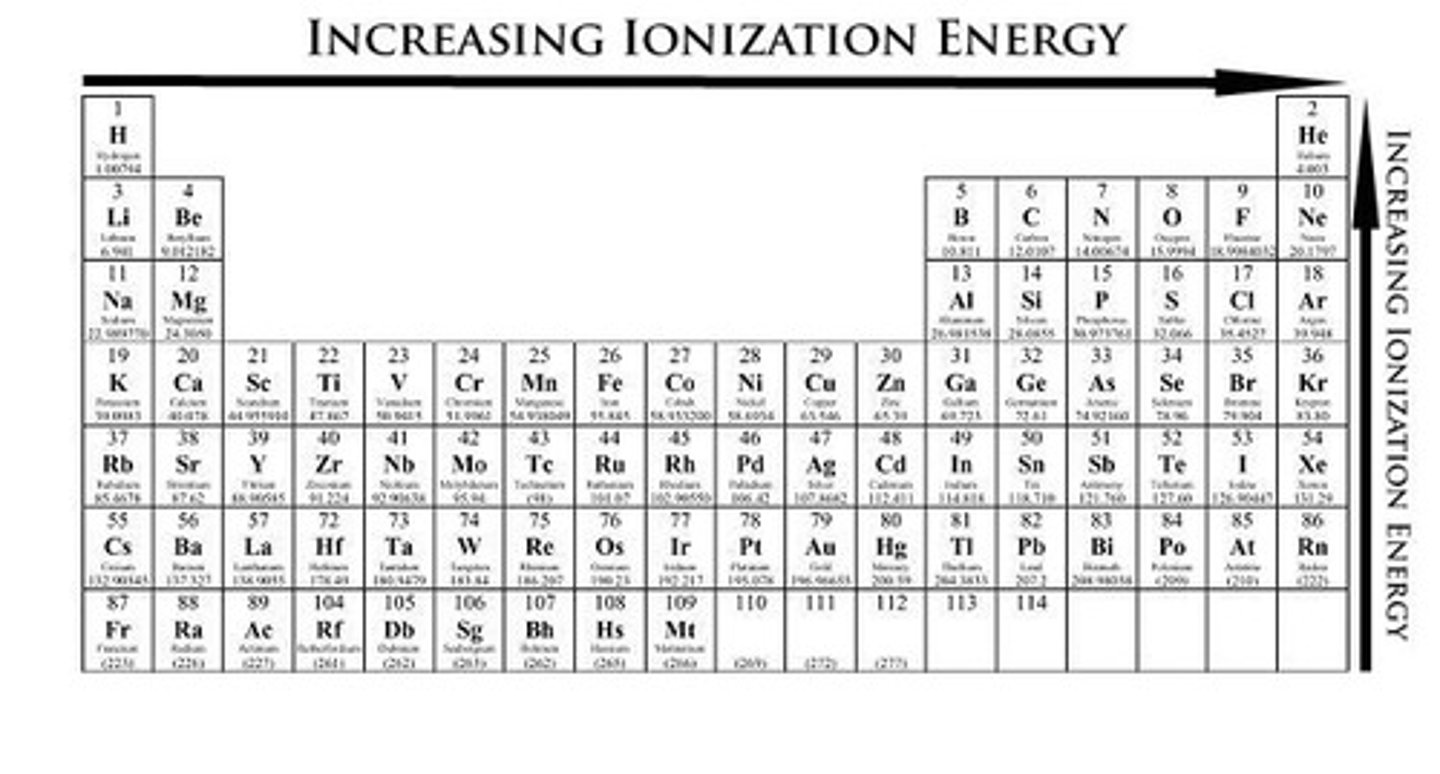
How does ionization energy trend down a group in the periodic table?
Ionization energy generally decreases from top to bottom within a group.
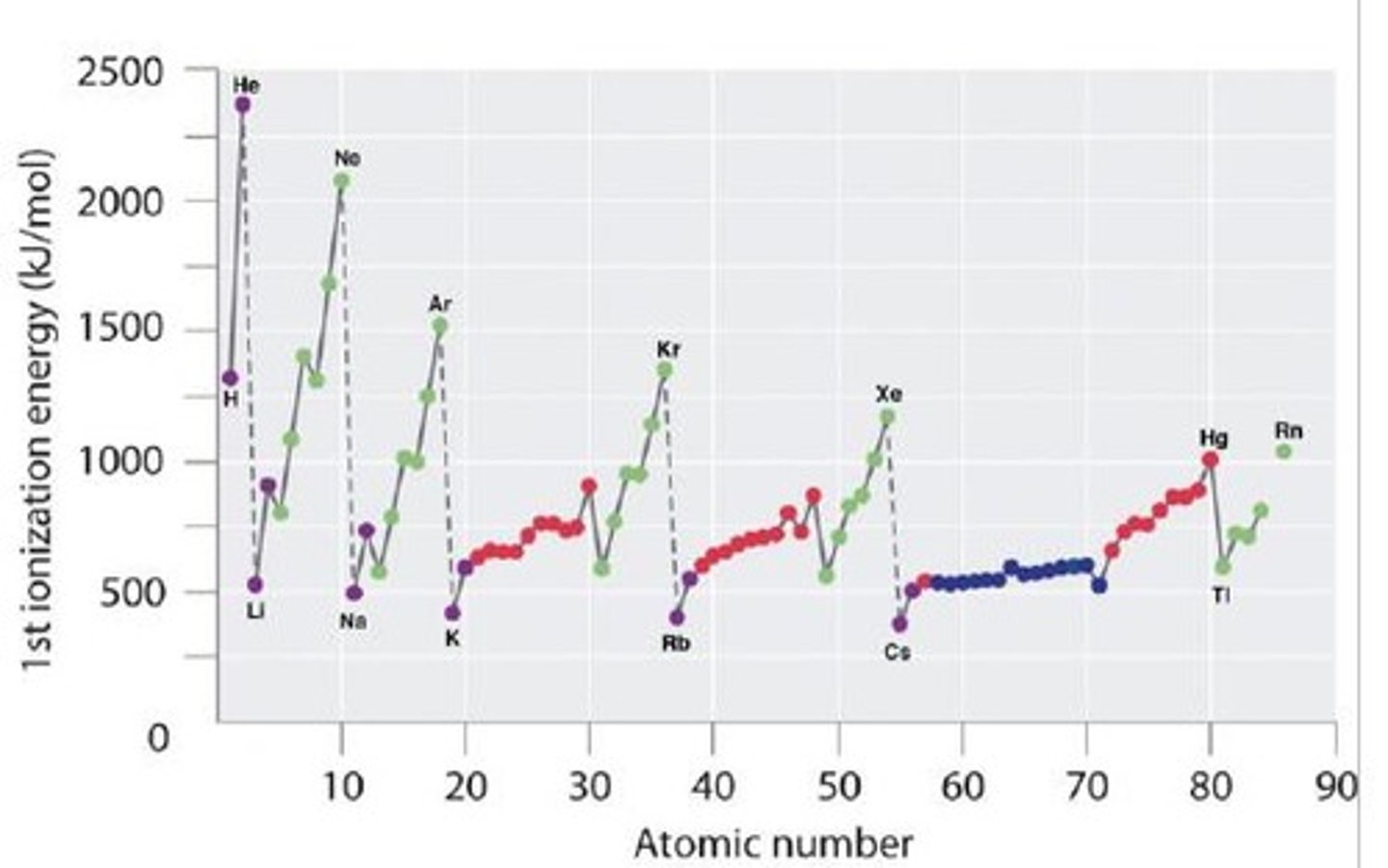
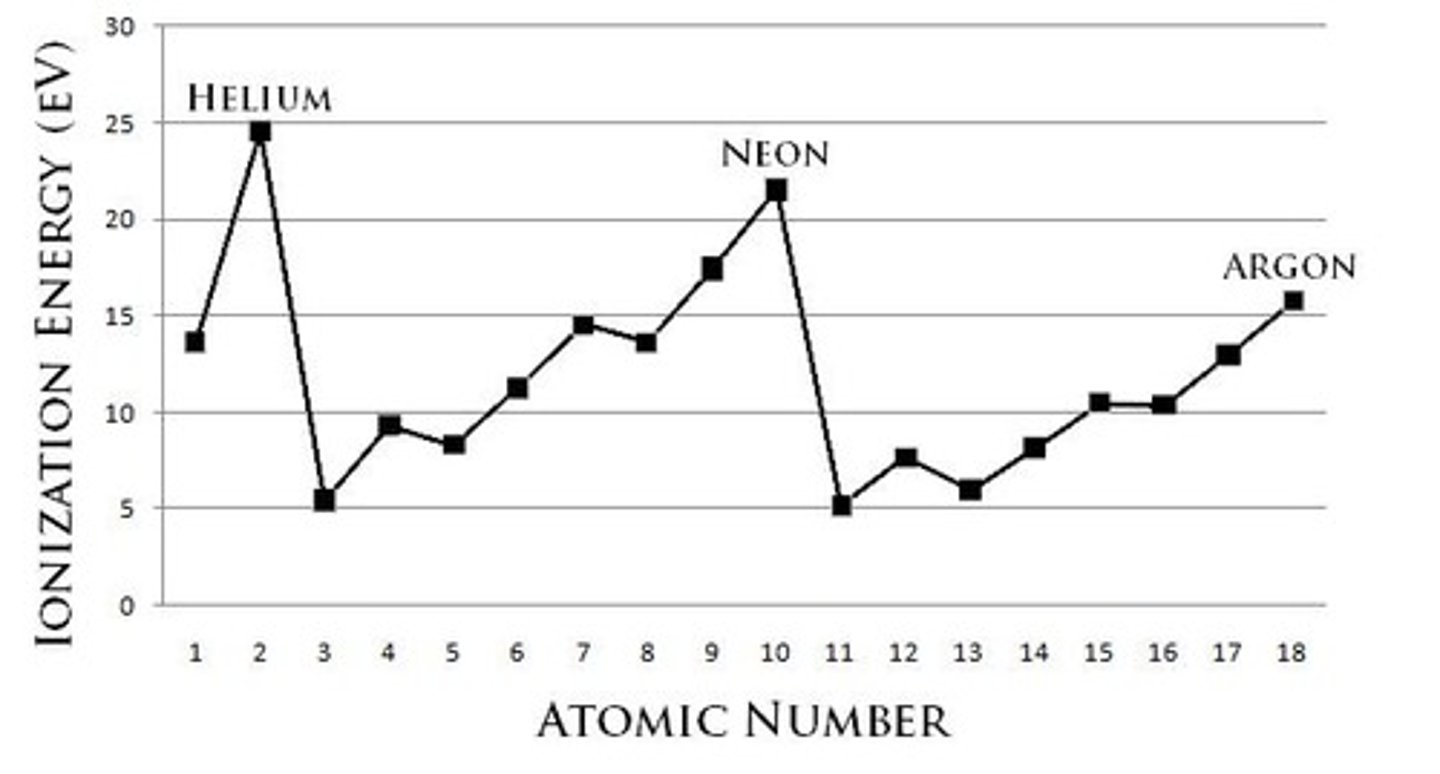
Why do noble gases have high ionization energies?
Because they have full valence shells, resulting in high effective nuclear charge.
What is the relationship between ionization energy and the stability of valence shells?
Higher ionization energy indicates greater stability of the valence shell.
What happens to ionization energy as you move from left to right in a period?
It increases due to the increase in effective nuclear charge.
What is the effect of increasing electron energy shells on ionization energy?
Ionization energy becomes smaller as electrons are added to higher energy shells, increasing the distance from the nucleus.
What is a notable exception in ionization energy trends within the periodic table?
Group 13 elements have lower ionization energy than group 2 elements due to stability of filled and half-filled subshells.
How does the filling of d and f orbitals affect ionization energy trends?
The increase in ionization energy across periods is less pronounced in d-block and f-block elements.
What is the significance of the effective nuclear charge in ionization energy?
It increases from left to right, making it harder to remove electrons as protons are added.
Which element has the highest ionization energy: Fluorine, Nitrogen, or Helium?
Helium (He) has the highest ionization energy.
What is the first ionization energy of Sodium (Na)?
496 kJ/mol.
What is the second ionization energy of Magnesium (Mg)?
1451 kJ/mol.
What is the third ionization energy of Aluminum (Al)?
2745 kJ/mol.
What is the fourth ionization energy of Silicon (Si)?
4356 kJ/mol.
What is the trend for ionization energy in the context of group 16 and group 15 elements?
Group 16 elements have lower ionization energy than group 15 elements due to stability of half-filled subshells.
What is the ionization energy of Argon (Ar)?
12000 kJ/mol.
How does ionization energy relate to the formation of cations?
Lower ionization energy means the atom more readily becomes a cation.
What is the ionization energy of Phosphorus (P)?
1060 kJ/mol for the first ionization energy.
What is the ionization energy of Sulfur (S)?
999.6 kJ/mol for the first ionization energy.