Physics paper 1
1/422
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
423 Terms
What is the law of conservation of energy?
Energy cannot be created or destroyed, only transformed from one form to another.
What is kinetic energy?
the energy an object has due to its motion
What is gravitational potential energy?
Energy an object has depends on its height, the work done against the force of gravity
What is mass?
the amount of matter in an object
What is weight?
A measure of the force of gravity on an object
What is the gravitational field strength on Earth?
9.8 N/kg
What is elastic potential energy?
Energy stored in stretched or compressed objects
What is chemical energy?
Energy stored in chemical bonds between atoms
What is thermal energy?
the total kinetic energy of all particles in a substance
What is magnetic energy?
the energy stored within a magnetic field, generated by moving electric charges (like currents) or permanent magnets,
What is electrostatic energy?
- the energy stored in objects due to the forces between electric charges (attraction/repulsion).
- It's built up when like charges are pushed together or opposite charges are pulled apart
What is nuclear energy?
Energy stored in the nucleus of an atom
How is nuclear energy released?
by atoms fusing together or splitting apart
Example of kinetic energy
a moving car
Example of GPE
Book on a shelf, Rollercoaster at the highest point
Example of elastic potential energy
stretched rubber band
Example of chemical energy
food, batteries, fuel
Example of thermal energy
fire, the sun
example of magnetic energy
Magnets attracting or repelling each other
Example of electrostatic energy
static electricity on clothes
Example of nuclear energy
nuclear fusion in sun, nuclear power plants use fission
What is internal energy?
the sum of the kinetic and potential energies of all particles in the system
What is potential energy?
-stored energy an object has due to its position, arrangement, or state
-It's energy waiting to be released, not currently in motion.
What are the four energy transfers?
Mechanically, electrically, heating, radiation
What is mechanical transfer?
the transfer of energy when a force moves an object over a distance, causing it to change its motion or position
What is electrical transfer?
energy transferred when a charge flows
What is transfer by heating?
when energy moves from a hotter object to a cooler one
What is transfer through radiation?
energy transferred as waves (light, sound or infrared)
Example of energy transfer
A rollercoaster at the top of a track has GPE. As it moves down, the energy is transferred mechanically into kinetic energy. some energy is lost as thermal energy due to friction
Example of mechanical energy transfer
A person kicking a ball
chemical→kinetic
Example of electrical energy transfer
A phone charging
chemical→electrical→thermal
Example of energy transfer through heating
A sausepan on a hob
Example of energy transfer through radiation
The sun heating the Earth
nuclear→light→thermal
Example of an energy transfer
A rollercoaster at the top of a track has GPE. As it moves down, energy is transferred mechanically into kinetic energy. Some energy is lost as thermal energy due to friction
How does friction generate heat?
by converting an object's kinetic energy (movement energy) into thermal energy (heat energy) when two surfaces rub together
What is mechanical energy?
- the energy an object has due to its motion (kinetic energy) and position (potential energy)
- like a falling ball converting stored gravitational energy into kinetic energy
What is efficiency?
The percentage of useful energy
Formula for efficiency?
Useful power output / total power input x 100
What is work done?
The energy transferred when a force moves an object over a distance
Formula for work done?
Work done = force x distance
Example of work done
- To raise an object, you need to apply a force to overcome the force of gravity on it
- If the work you do is 20J, the energy transferred to it must be 20J, so its GPE incresessby 20J
What is power?
The rate at which work is done or energy is transferred.
Formula for power
Power = work/time
Power = energy transferred/time
What is work done measured in?
Joules
What is power measured in?
Watts
How is thermal energy transferred?
From the hotter object to the cooler one through conduction, convection and radiation
What is conduction?
transfer of heat through direct contact
In what state does conduction happen in?
solids
How does conduction work?
- Particles in a solid are tightly packed together
- When heated, these particles gain energy and vibrate more
- These vibrations cause the particles to collide and pass energy to nearby particles, transferring heat
Example of conduction?
A pot sitting on a hot burner.
Why are metals good conductors of heat?
because of their "sea" of free, delocalized electrons that can rapidly move through the structure and transfer kinetic energy throughout the material, alongside normal atomic vibrations
Why are plastics and woods bad conductors?
their molecules aren't tightly packed and they lack free electrons
What are conductors?
materials which transfer energy well
What are insulators?
materials that resist the flow of energy, like electricity, heat, or sound, because their electrons are tightly bound and don't move freely, unlike in conductors
What is thermal conductivity?
A measure of how quickly energy is transferred through a material via conduction
What is thermal conductivity measured in?
W/m°C
What is convection?
The transfer of heat by the movement of a fluid
In what state does convection happen?
liquids and gases
How does convection work?
1. The fluid near the heat source expands, becomes less dense and rises
2. cooler, denser fluid moves to take its place
3. This cycle continues and creates a convection current
What is thermal radiation?
The transfer of heat by the emission of infrared radiation
How does thermal radiation work?
- All objects emit and absorb infrared radiation
- Dark, matte surfaces absorb and emit more heat
- Shiny, light surfaces reflect heat
In what state does thermal radiation happen?
any, it doesn't require any particles
What is infrared radiation?
a type of electromagnetic radiation
What do hotter objects emit?
they emit more infrared radiation in a given time
What do objects at a constant temperature do?
emit infrared radiation at the same rate that they are absorbing it
Why do hotter objects emit shorter wavelengths?
They have a higher thermal energy, which makes their particles vibrate faster, producing higher-frequency electromagnetic waves
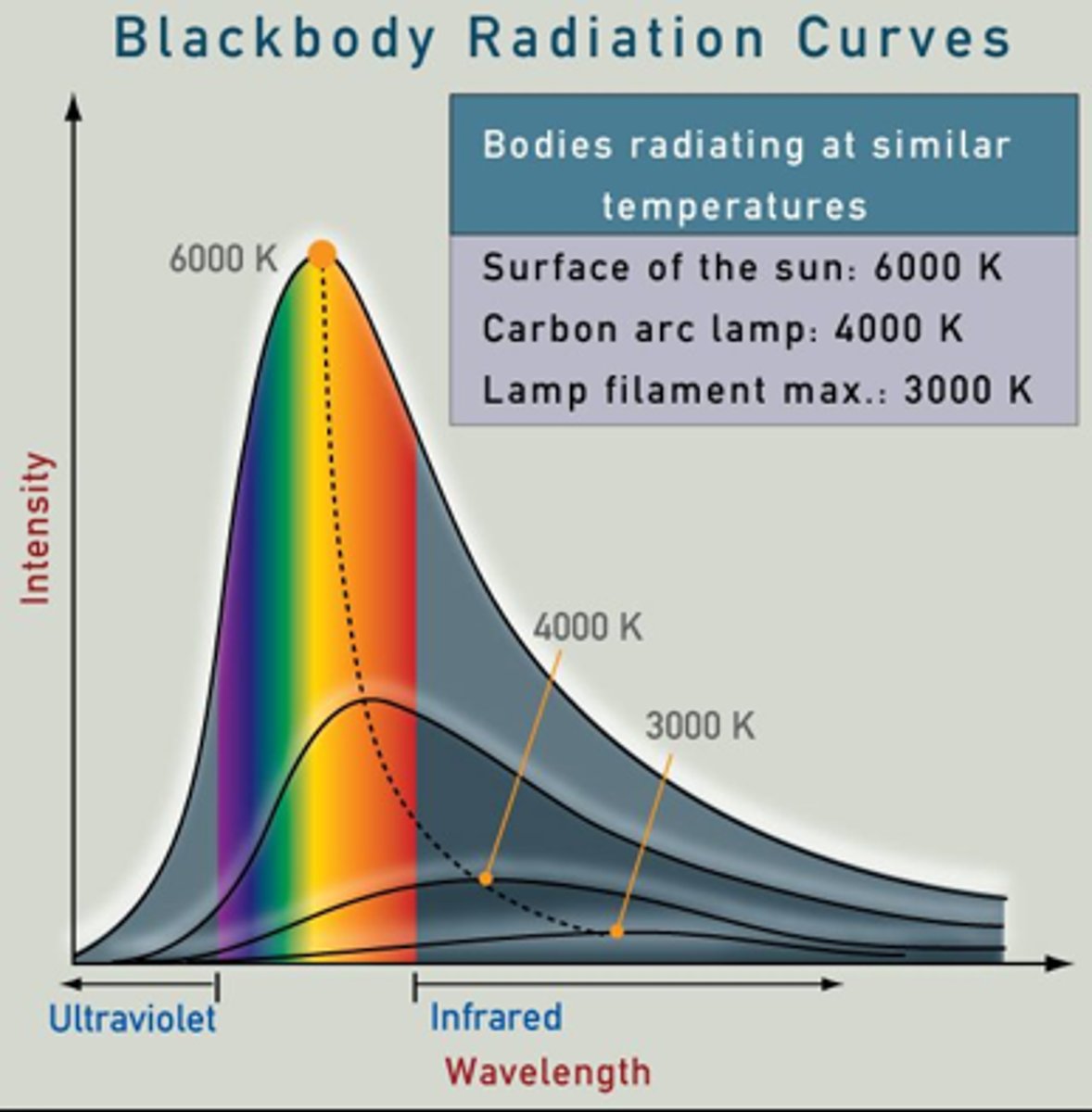
Why do all objects absorb and emit infrared?
because all objects with a temperature above absolute zero has vibrating atoms and molecules, which constantly release and absorb infrared
Why do objects emit infrared?
because all objects with a temperature above absolute zero have internal energy in their atoms and molecules, causing them to vibrate and release this energy as electromagnetic waves
Why do objects absorb infrared?
because it's a form of electromagnetic energy that makes the particles (atoms/molecules) in the object's surface vibrate and gain kinetic energy, causing the object to heat up
What is a black body?
A theoretical object that absorbs 100% of the radiation that falls on it. It does not reflect or transmit any radiation and is the best emitter
What is the greenhouse effect?
Greenhouse gases (like water vapour, methane and carbon dioxide) absorb long-wavelength radiation that gets reflected back off the Earth. They then re-radiate it in all directions, including back towards Earth. The longwave radiation is thermal radiation, so it results in warming of the Earth's surface
What is specific heat capacity?
The amount of energy required to increase the temperature of 1kg of a substance by 1°C
What does a higher specific heat capacity mean?
It takes more energy to heat up 1kg by 1°C and it stores heat well
What does a low specific heat capacity mean?
It heats up and cools down quickly
Why do materials with a low specific heat capacity cool down quickly?
They require less energy to change their temperature, meaning they release their stored heat energy to the surroundings more rapidly, causing a faster drop in temperature
What is the SHC of water?
4200
What is SHC measured in?
J/kg°C
What is the SHC of iron?
450
Example of SHC
Oceans absorb and store large amounts of heat, helping to regulate the Earth's temperature
What is loft insulation?
Material such as fibreglass is put into the loft space because it is a good insulator, so it reduces the rate of energy transfer by conduction. Thicker insulation will be a better insulator.
What is cavity wall insulation?
Insulation material that is used to fill the cavity between the two brick layers of an external house wall to reduce unwanted energy transfer
What are double-glazed windows?
Two glass panes with dry air or a vacuum between them to prevent heat loss
How can you reduce energy loss in the home?
- make walls from an insulating material
- use cavity wall insulation
- double-glazed windows
- Insulate walls and the attic
- Insulate hot water systems
- aluminium foil between a radiator panel and the wall to reflect heat away from the walls
How are particles arranged in solids?
Particles are closely packed in fixed positions by strong forces of attraction
How are particles arranged in liquids?
The particles of a liquid are close together but can move around one another freely. The forces of attraction are weaker than in solids
How are particles arranged in gases?
Particles are far apart and move freely. There are almost no forces of attraction
How do particles move in solids?
Vibrate about a fixed position
How do particles move in liquids?
Particles are free to move and slide past each other randomly
How do particles move in gases?
High speed, random directions
What are the properties of solids?
High density, fixed volume and fixed shape unless deformed by an external force. They can't be compressed or squashed.
What are the properties of liquids?
They have a fixed volume and take shape of a container, they flow and move randomly. They cannot be compressed or squashed.
What are the properties of gases?
-low density
-no fixed volume
-They can flow and move randomly
-they spread out to fill all available space
-They can be compressed or squashed.
What is solid to liquid called?
melting
What is liquid to gas called?
evaporation
What is gas to liquid called?
condensation
What is liquid to solid called?
freezing
What is solid to gas called?
sublimation
What is gas to solid called?
deposition
What is the kinetic theory of matter?
- All atoms are in constant motion, so all atoms have kinetic energy.
- For a given amount of a substance, its particles have the most energy in the gas state and the least energy in the solid state
What happens when a change in state takes place?
The temperature stays the same because the added heat energy it's used to break the strong intermolecular bonds holding the substance in its current state, allowing it to transition to the next state