nervous system 3
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
ion channels
specialised membrane proteins that select the type of ion (or ions) that they allow to pass through the membrane.
types of ion channels
leak channels and gated ion channels
leakage channels: always open
called leakage channels due to them being consistently open.
said to open and close randomly, they can essentially be treated as permanently open.
enable ions to leak into or out of the cell, aligning with their concentration gradient.
gated ion channels
change conformation/shape and open/close in response to specific signals or stimuli.
gated ion channels: ligand-gated ion channels
respond to chemical signals or ligands that bind to them
once bound, triggers the opening of the ion channel, permitting ions to flow into or out of the cell and hence, altering the cells permeability to that particular ion.
abundant in chemical synapses with neurotransmitters acting as ligands to initiate ion channel opening.
gated ion channels: mechanically gated ion channels.
prevalent in sensory receptors, especially those associated with touch in the skin.
mechanical stimuli activate these channels
ion channel opens upon mechanical stimulation, allowing ions to move in or out of the cell according to their concentration gradients.
gated ion channels: voltage-gated ion channels
sensitive to changes in membrane voltage
in excitable cells (e.g. neurons and muscles) there is a difference in eletrical charge across the cell membrane
voltage channels can trigger the opening of voltage gated ion channels.
these channels play a pivotal role in generating and propagating action potentials in neurons and excitable cells.
found along neuron exons and plasma membrane of muscle cells.
resting membrane potential
nerve and muscle cells can change their potential in a precise, regulated way.
forms the basis of how nerve cells communicate with one another.
transient changes in the membrane potential from its resting level produce electrical signals.
graded potentials
signal over short distances
action potentials
signal over long distances
to get an electrical signal to start, the membrane potential has to change
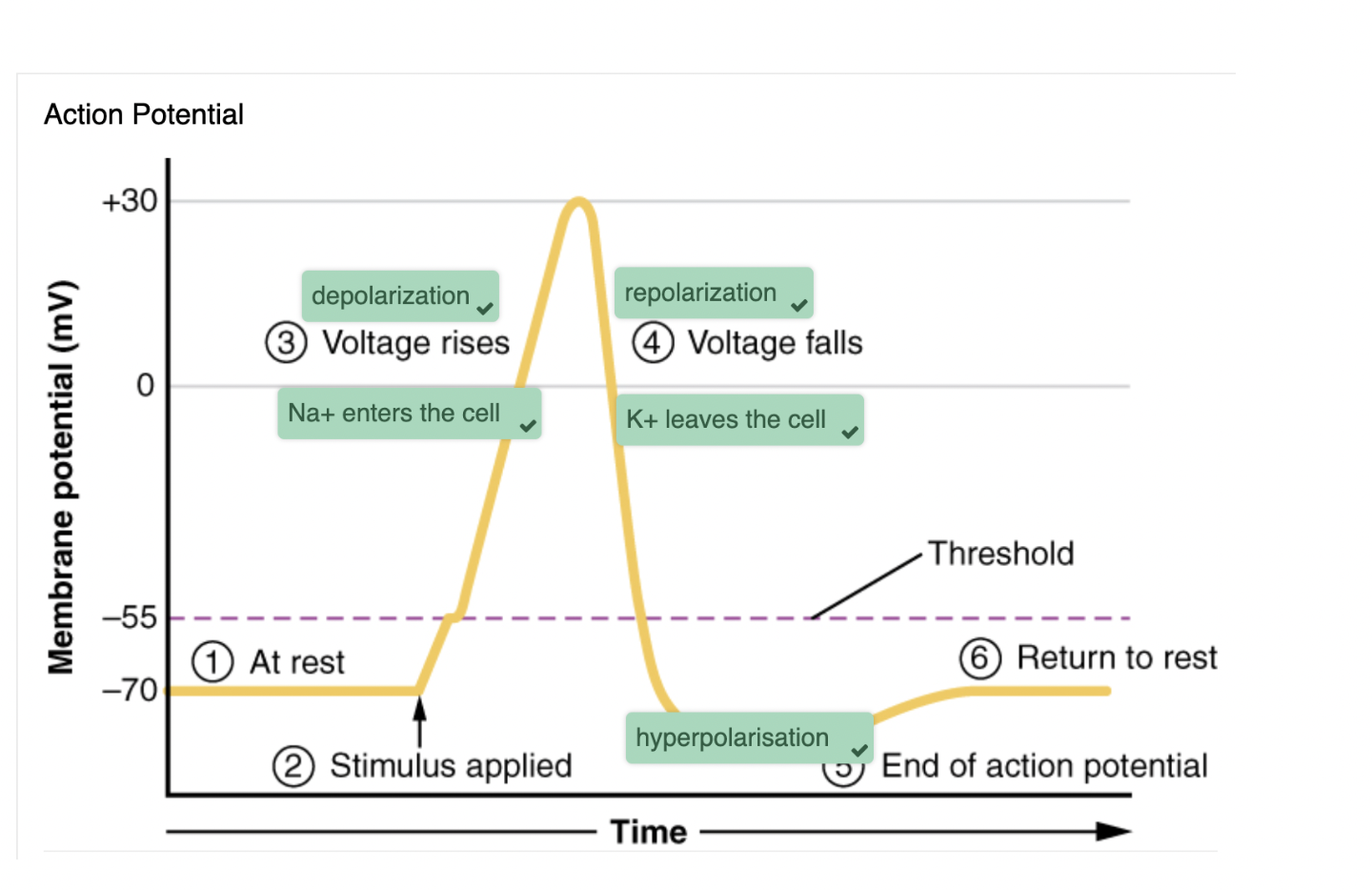
steps of action potentials
membrane potential stays at resting voltage until something changes
channels that start depolarizing the membrane because of a stimulus help the cell depolarise from -70mV to -55mV
once the membrane reaches -55mV, voltage gated Na+ channels open and an action potential starts
voltage-gated channels open in response to changes in charge distributed across the membrane during repolarisation
K+ ions leave the cell
hyperpolarization
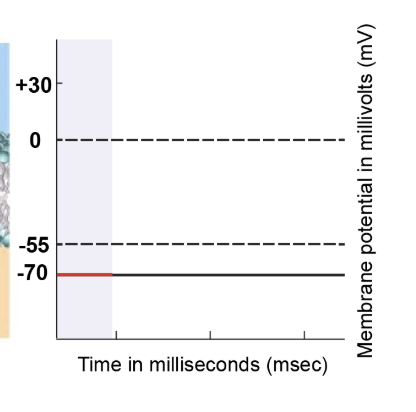
resting state
At the resting state, the cell's membrane potential is -70 millivolts. Ion movement primarily occurs through leak channels and sodium-potassium pumps. All voltage-gated channels, including sodium and potassium, remain closed. This state sets the baseline for the subsequent action potential process.
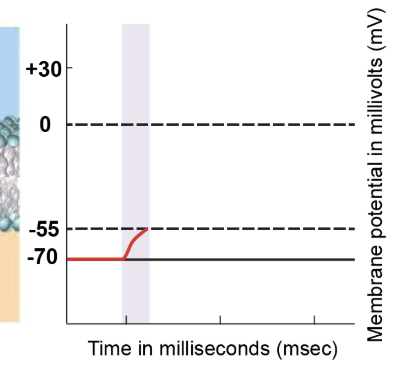
stimulus depolarises membrane to threshold
Graded potentials are initiated by external stimuli and reach the axon hillock. These graded potentials cause depolarisation of the membrane at the axon hillock, pushing it closer to the threshold value. The axon hillock accumulates these graded potentials, and their summation can elevate the membrane potential to -55 millivolts, known as the threshold potential.
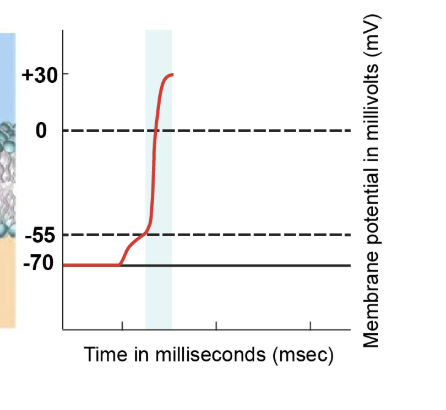
depolarisation phase
Upon reaching the threshold potential of -55 millivolts, voltage-gated sodium channels rapidly open. Sodium ions rush into the cell due to the higher concentration outside. The influx of positively charged sodium ions leads to significant depolarisation. This rapid depolarisation phase sees the membrane potential increase from -55 millivolts to approximately +30 millivolts.
repolarisation phase
As the membrane potential reaches its peak positive value, inactivation gates of voltage-gated sodium channels close. Simultaneously, voltage-gated potassium channels start opening. These channels, though slower to open, permit potassium ions to exit the cell. The efflux of positively charged potassium ions initiates repolarisation, causing the membrane potential to become more negative.
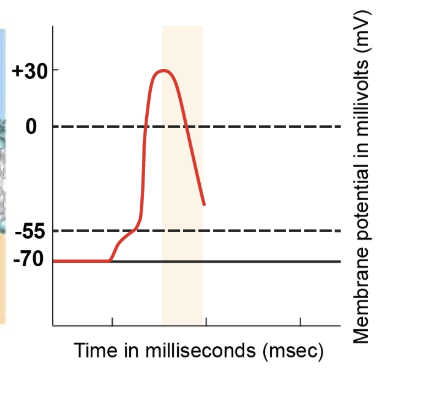
hyperpolarisation phase
Voltage-gated potassium channels continue to remain open, allowing more potassium ions to leave the cell. As a result, the cell loses a positive charge, making the internal environment more negative than the resting potential. This brief hyperpolarisation phase showcases the membrane potential dropping below the resting value of -70 millivolts.
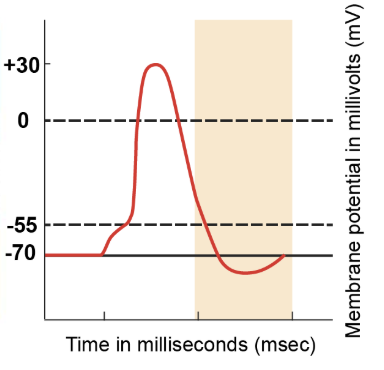
return to resting state
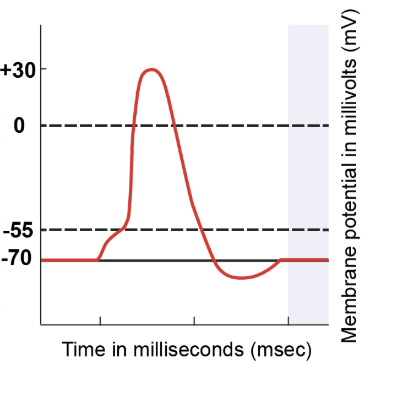
Even as the membrane potential returns to -70 millivolts, some voltage-gated potassium channels remain open. This ongoing outflow of potassium ions leads to a more negative membrane potential than the resting state. However, these voltage-gated potassium channels eventually close, leaving only leak channels and the sodium-potassium pumps to maintain ion balance. Regardless of the starting point, when only leak channels and pumps are active, the membrane potential returns to the resting value of -70 millivolts. This sequence marks the completion of the action potential process.
chemical synapses
neurotransmitter is released from one cell and it affects the other cell
involve the transmission of chemical information from one cell to the next
transmits signals (action potentials) between neurons or neurons and muscles or glands
neurotransmitter
chemical signal that is released from the synaptic end bulb of a neiron to cause a change in the target cell
chemical synapse
connection between two neurons, or bewteen a neuron and its target, where a neurotransmitter diffuses across a very short distance.
synaptic cleft
small gap between cells in a chemical synapse where neurotransmitter diffuses from the presynaptic element to the postsynaptic element.
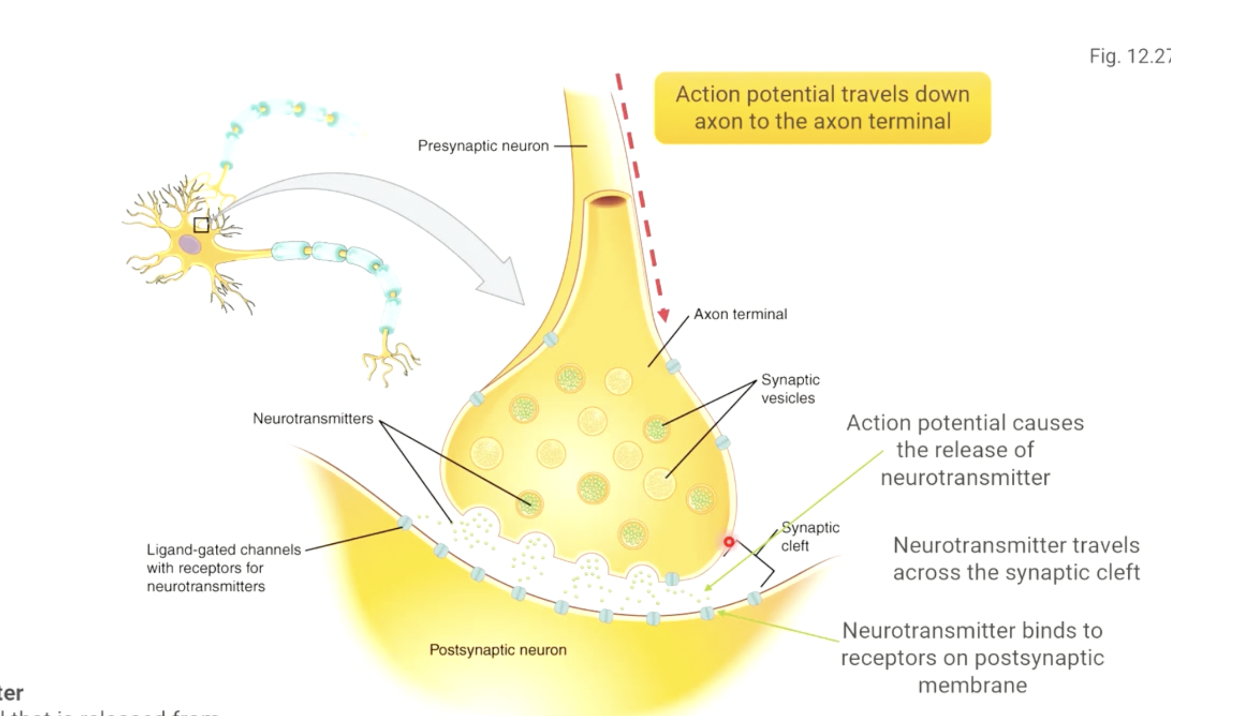
steps of neurotransmission
the action potential travels down the axon to the axon terminal, one of the branches at the very end of the axon.
the electrical signal causes the axon terminal to release the neurotransmitter it has stored.
neurotransmitter travels across the space between the two neurons (synaptic cleft)
neurotransmitter binds to receptors on the postsynaptic membrane, and binding triggers a change in the cell
impact of drugs, neurotoxins and diseases can be grouped by…
Where they act
- CNS, PNS, neuromuscular junction
- Ion channels, receptors, enzymes, synapses
What they do (excite, inhibit, block, mimic, disrupt)
Which neurotransmitter system they influence
Their clinical/behavioural effect
Their mechanism of action
mechanism of action: nervous system functioning can be altered by affecting…
ion channels
neurotransmitter release
neurotransmitter binding to postsynaptic receptors
neurotransmitter degradation (preventing removal of neurotransmitter)
ion channels
blocking or disrupting various ion channels to disrupt normal functioning
Na+ channels
K+ channels
Ca2+ channels
Cl- channels
e.g. numerous toxins from animal venoms (snake, spider)
neurotransmitter release: inhibiting
botulinum toxin (botox)
tetanus toxin
neurotransmitter release: increasing
red back spider venom
receptor binding: activating receptors (‘mimicking neurotransmitter’)
nicotine
opioids (e.g. morphine, heroin)
benzodiazepines (e.g. valium)
receptor binding: blocking receptors (antagonists)
some venom/toxins
curare
ketamine
caffeine
preventing neurotransmitter removal: preventing re-uptake
cocaine
SSRI’s (antidepressants)
preventing neurotransmitter removal: preventing breakdown
organophosphate insecticides
nerve agents