Chemistry Study Guide Test 11
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
Rates of reactions can be determined by monitoring the change in…
CONCENTRATION of reactants or products and a change in time. ( Δ[ ]/ ΔTime)
The average rate of the reaction over each interval is..
the change in concentration divided by the change in time
↓Concentration = ↓Rate of reaction
Less collisions = less of reaction
Collision Theory/Model
Molecules CANNOT react unless they “bump” into each other
Not all collisions are effective
Collisions must have sufficient energy and proper orientation to collide
Activation energy
minimum amount of energy required for a reaction to occur
A reaction cannot occur UNLESS…
molecules possess enough energy to prevail the activation energy border
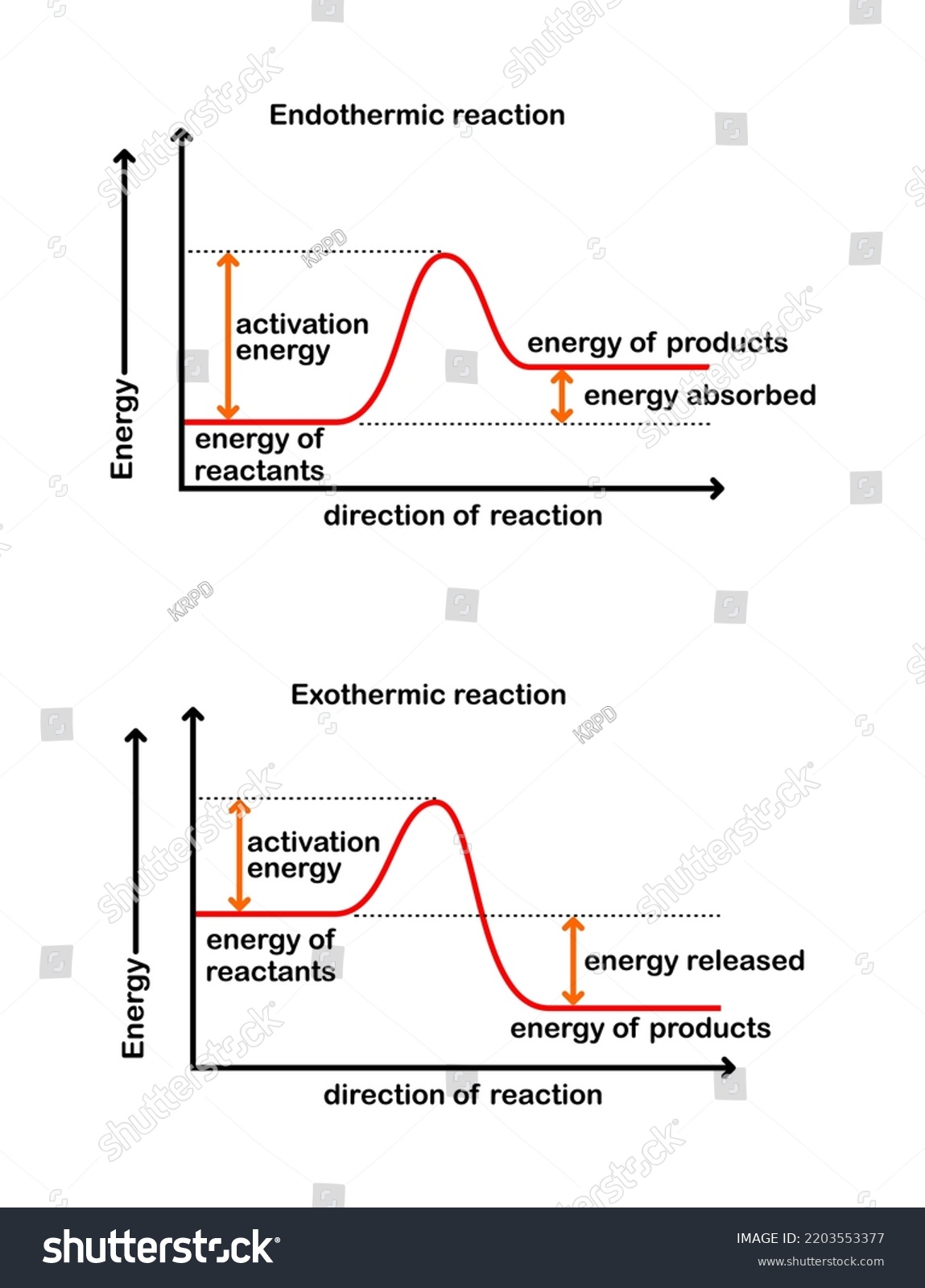
Reaction Coordinate Diagram
An activated complex is intermediate product of the reaction
Activation energy goes over the energy barrier
ΔH is depicted between the reactant and products
+ΔH is endo, -ΔH is exo, EXOTHERMIC REACTION NEEDS LESS ACTIVATION ENERGY
Factors of Reaction Rate
⭡Reaction rate = ⭡effective collisions
⭡amount of reactants
⭡surface area
Stirring
Adding a catalyst
⭡Temperature (more kinetic energy, particles move faster)
Opposite of catalyst
inhibitors
Homogeneous catalysts: same state of matter
Heterogeneous catalysts: different state of matter
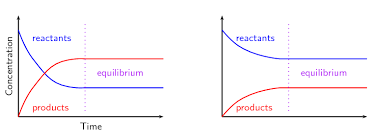
Equilibrium
reversible processes, rate of the forward reaction=rate of reverse reaction
Equilibrium Phase Diagram
Reactants begin to decrease when products begin to increase (get used up)
Opposite of forward reaction
Equilibrium reached when lines flatten out
At equilibrium: Rate of forward reaction = rate of reverse reaction, concentration of reactants and products is CONSTANT
Equilibrium in chemistry: balance of opposite processes (movement)
Equilibrium equation
K = equilibrium constant: K = [concentration of products]/[concentration of reactants]
Solids and pure liquids are NOT included
Coefficients become exponents
Nothing BUT TEMPERATURE changes equilibrium constant
Le Chatelier’s Principle
An equilibrium system subjected to a stress will shift to counteract it.
Types of stresses: change in temperature, change in concentration, change in volume or pressure (for gases)
Catalyst is NOT a stressor
Law of Mass of Action
ratio of concentration products/ratio of concentration reactants = constant
Haber Process
N2 + 3H2 ↔ 2NH3
If H2 is added, N2 will be consumed and the reagents will form MORE NH3
Pressure is proportional ONLY to moles of gas in the system
Mass Action Expression
Q: any point in reaction (reaction quotient)
K: @ equilibrium (equilibrium constant)
Q vs. K
Q>K: equilibrium shifts to reactants
Q≅K: equilibrium
Q<K: equilibrium shifts to products