3.3.6.3 infra-red spectroscopy
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
why is infra-red spectrometry used by organic chemists?
its often used to help them identify the type of bonds in a compound
describe how infra-red spectroscopy works
each bonds vibrates at the same frequency as IR
the frequency depends on the mass of the atoms in the bond, the bond strength, + the type of vibration
observing which frequencies of radiation are absorbed allows bonds to be identified
describe what happens in an IR spectrometer
a beam of IR radiation containing a spread of frequencies is passed through a sample
bonds in a molecule absorb infrared radiation at characteristics wavenumber
the radiation that emerges is missing the frequencies that are absorbed by the bonds found in the sample
the instruments plots a graph of the intensity of the radiation emerging from the sample, called the transmittance, against the frequency of radiation
how is the frequency of radiation measured?
as a wavenumber, measured in cm⁻¹
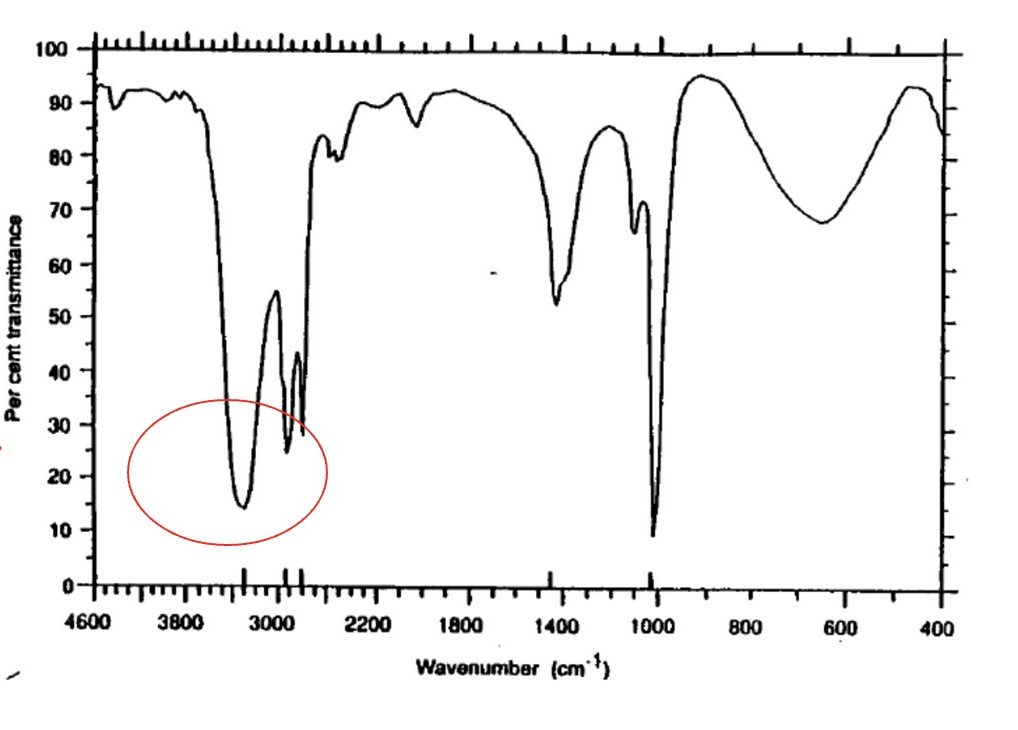
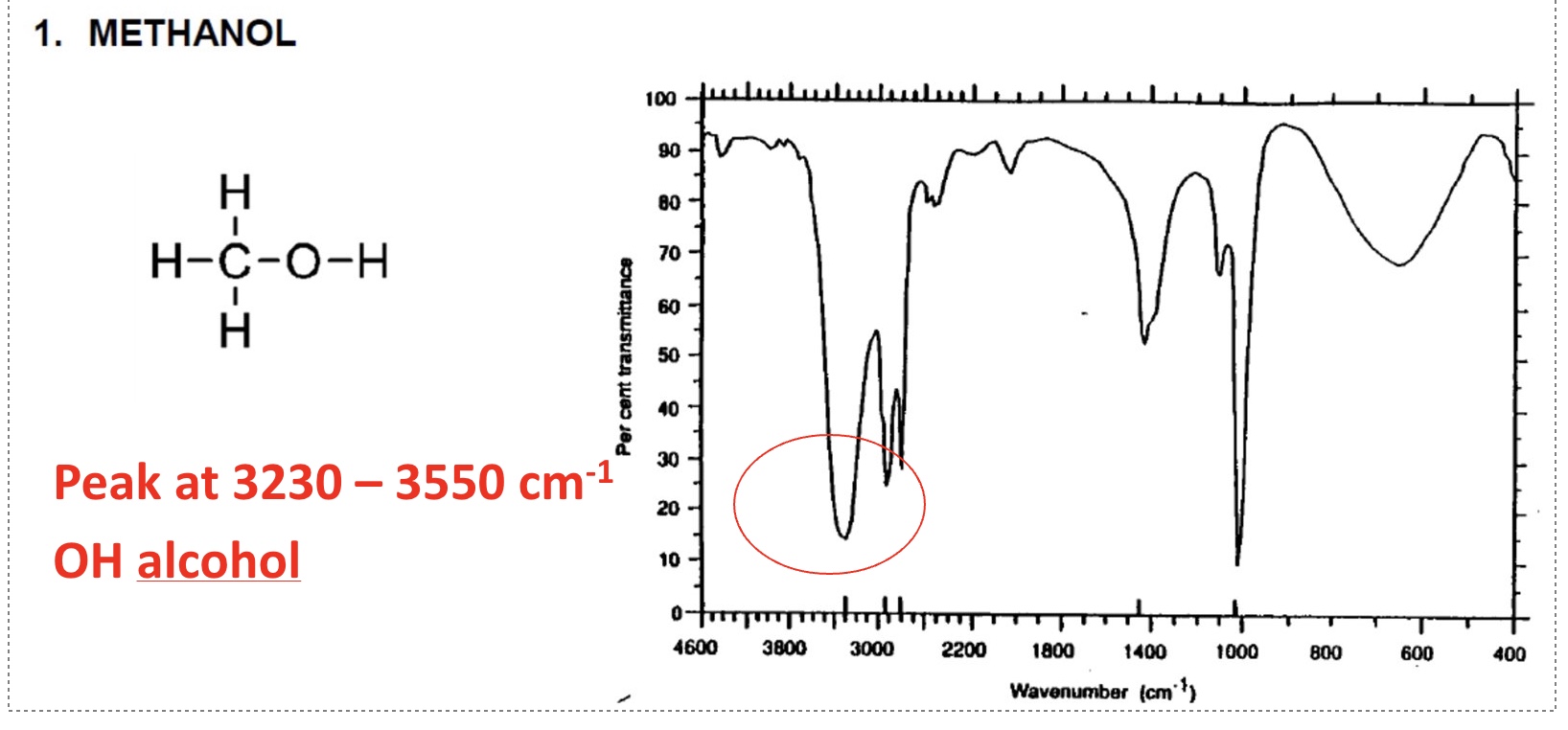
for the spectrum of methanol, label the bonds that are responsible for each stretch + give the wavenumber for each one
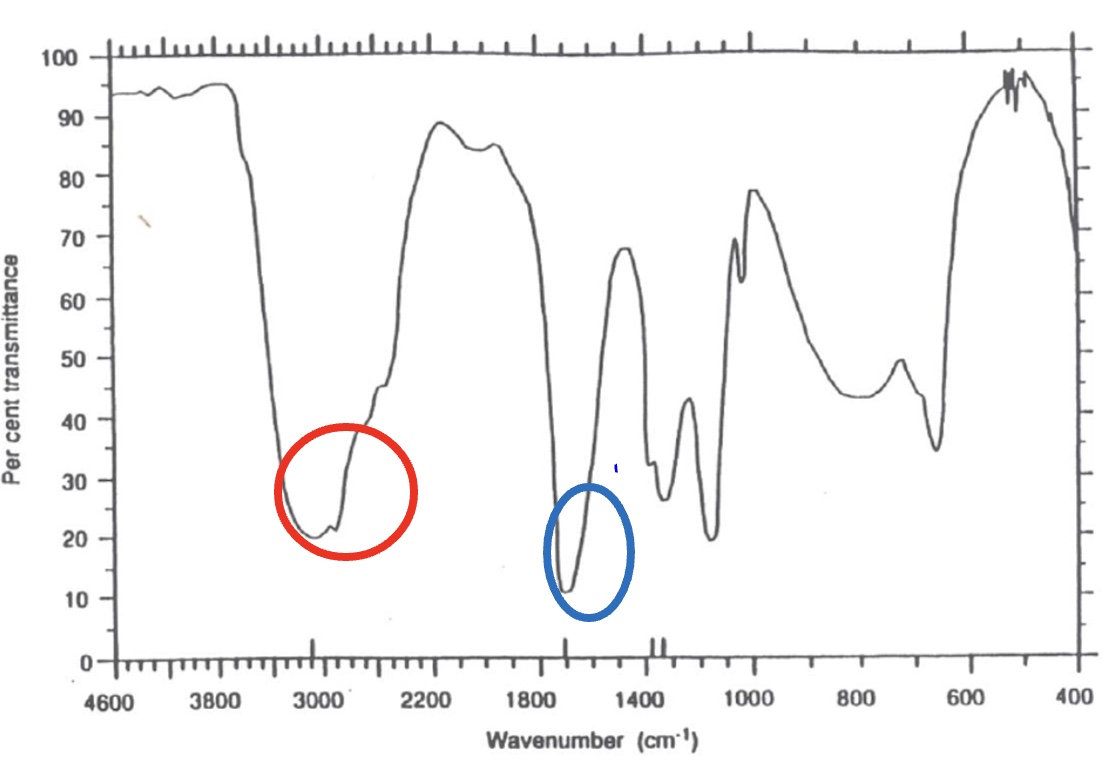
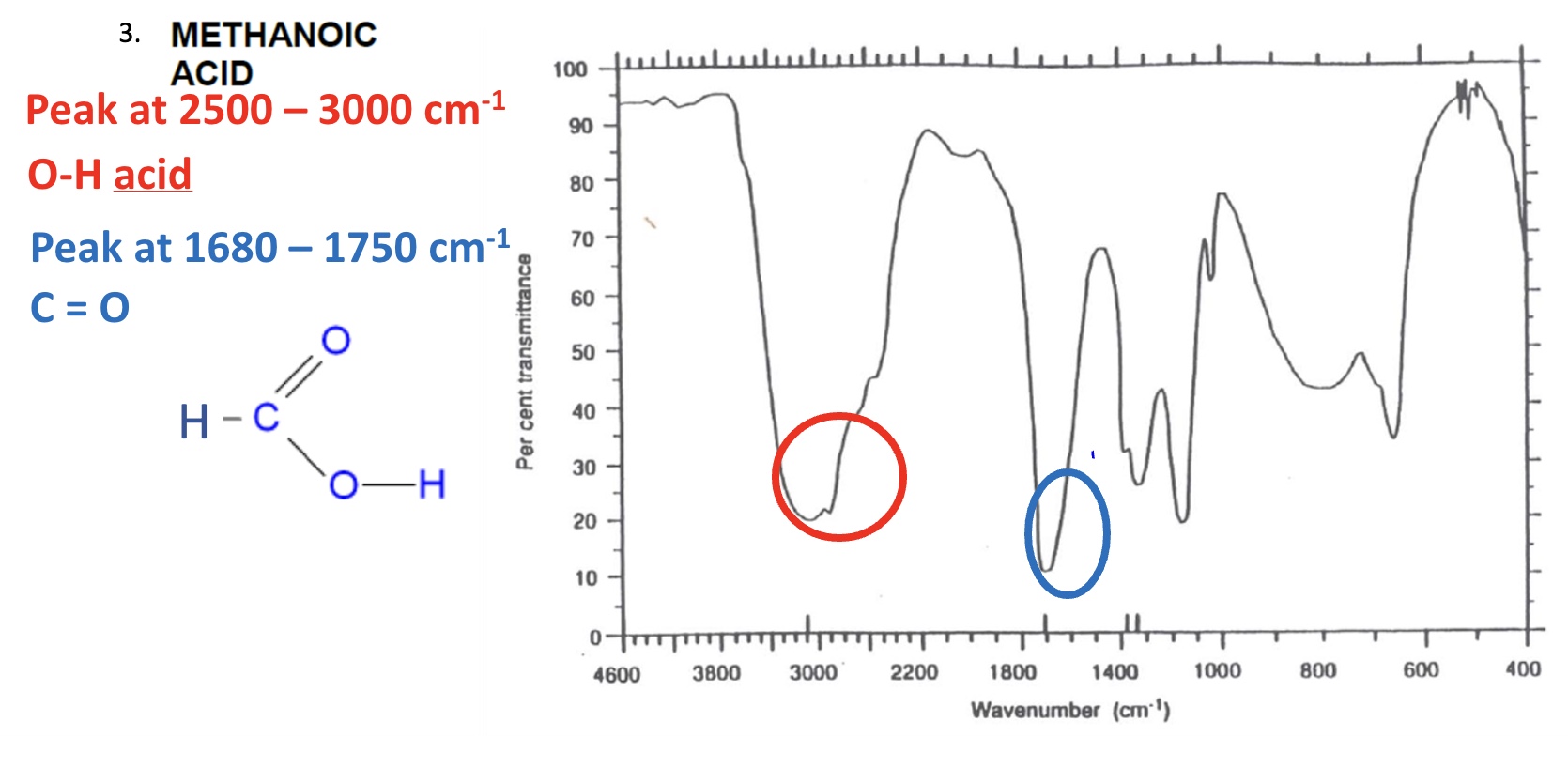
for the spectrum of methanoic acid, label the bonds that are responsible for each stretch + give the wavenumber for each one
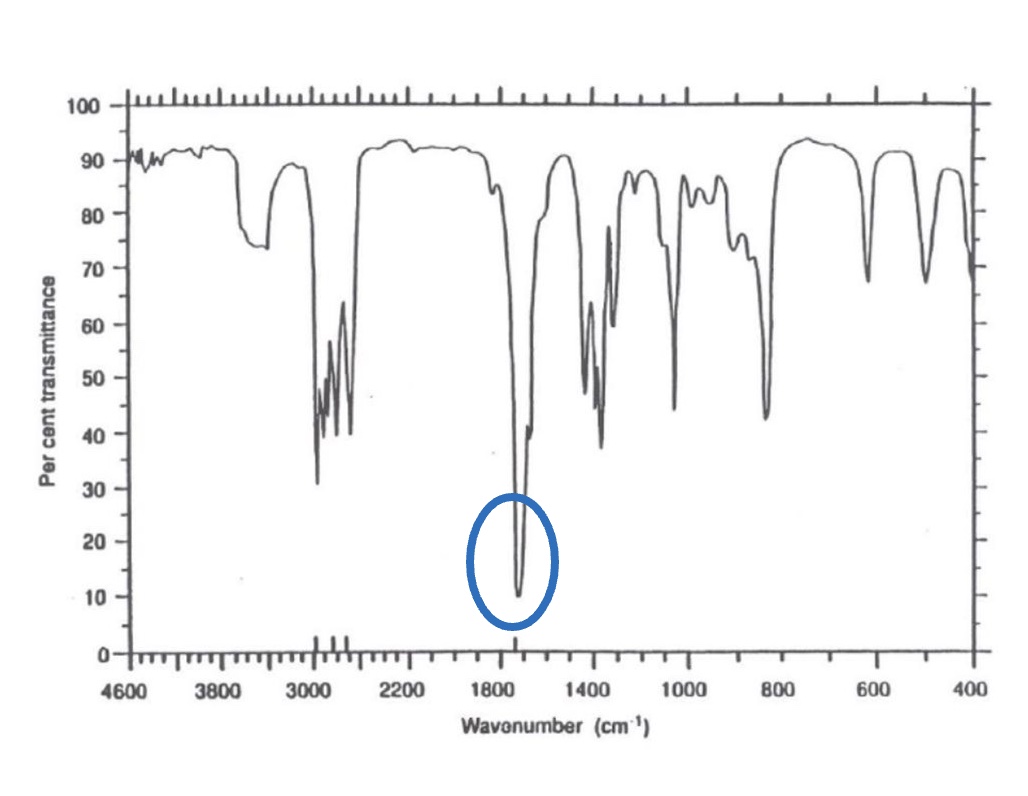
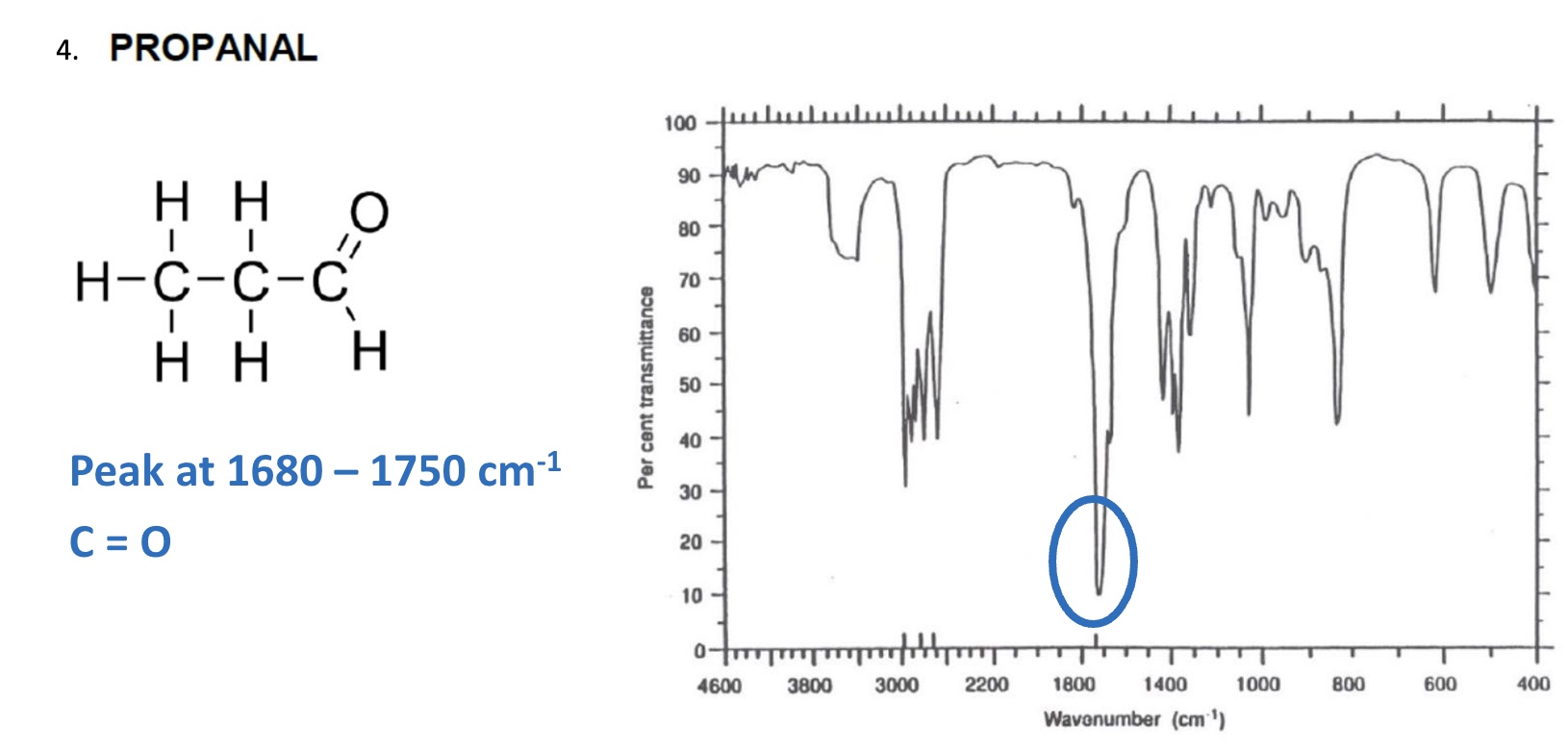
for the spectrum of propanal, label the bonds that are responsible for each stretch + give the wavenumber for each one
what is the fingerprint region?
the area of an infra-red spectrum below 1500cm⁻¹
how does the fingerprint region differ for different substances?
its shape is unique for any particular substance
what can the fingerprint region be used to do?
to identify the compound
how can the fingerprint region be used to identify compounds?
we can use a computer to match the fingerprint region of a sample with those on a database of known compounds → an exact match confirms identification of the sample
for example, spectra of propan-1-ol and propane-2-ol are both similar, why is that? what makes them identifiable then?
both spectra have absorption in the 3230-3350cm⁻¹ region which is due to the O-H bond in alcohols
but they have different pattern of peaks in the fingerprint region
what can IR spectra also be used to show?
the presence of impurities
how may impurities be revealed?
these may be revealed in absorptions that should not be there in the pure compound
eg the oxidation of an alcohol to a carboxylic acid:
if an IR spectrum is recorded during the reaction + a broad stretch is found at 3400cm⁻¹, which impurity is present?
O-H (alcohol) peak between 3230-3550cm⁻¹
unreacted alcohol purity
eg the oxidation of an alcohol to a carboxylic acid
why might it be difficult to see if an aldehyde impurity is present in the final product?
C=O peak between 1680-1750cm⁻¹
both aldehydes + carboxylic acids have C=O in this range so peaks may overlap
give 3 examples of greenhouse gases
water vapour
carbon dioxide
methane
how do greenhouse gases lead to global warming?
they absorb radiation from the sun + eventually re-emit it as long-wavelength radiation that increases the temperature of the earth → the trap heat in the earth’s atmosphere
what is the link between infrared radiation + global warming? how is this found?
the infrared spectra of water vapour, methane + of carbon dioxide shows this link
all three bonds, C=O, C-H + O-H vibrate in the same frequency as infrared light (although slightly different frequencies within IR region)
what is the criteria for the ‘best greenhouse gas’?
the best greenhouse gas absorbs the most infrared radiation + traps the heat in the earth’s atmosphere
the more ____ the bond, the more IR radiation the bonds absorb
polar
the bigger the area of a peak, the more…
IR radiation absorbed by that bond